rcm7291-sup-0001-SI
advertisement

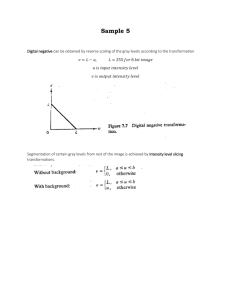
Page 1 Supporting Information Observation and Confirmation of Oxidation Reaction Occurring on Ultra-high Performance Liquid Chromatography Column Caiming Tang 1,2,*, Jianhua Tan 3,4,*, Jiabin Jin 1,2, Shaofeng Xi 4, Huiyong Li 4, Qilai Xie 3, Xianzhi Peng 1 1 State Key Laboratory of Organic Geochemistry, Guangzhou Institute of Geochemistry, Chinese Academy of Sciences, Guangzhou 510640, China 2 University of Chinese Academy of Sciences, Beijing 100049, China 3 College of Natural Resources and Environment, South China Agricultural University, Guangzhou 510642, China 4 Guangzhou Quality Supervision and Testing Institute, Guangzhou 510110, China *Corresponding Author. Caiming Tang Tel: +86-020-85291489; Fax: +86-020-85290009. E-mail: CaimingTang@gig.ac.cn. Jianhua Tan Tel: +86-020-020-83300529; Fax: +86-020-020-83300529. E-mail: tanjianhua0734@aliyun.com. Page 2 Figure Captions. Figure S-1. Workflow of the finding, confirmation and explanation of the redox reaction on UPLC column. Notes: [M-G-H]+ is the product ion of the precursor ion [MH]+ losing a glucoside (G) group, and [M-G-3H]- is the product ion of the precursor ion [M-3H]- losing a G group; VC: ascorbic acid. Figure S-2. High resolution mass spectra of baicalin and its oxidized product caused by on-column oxidation reaction. The precursor ions of baicalein and baicalein oxide were generated during collision induced dissociation (CID). The detection was conducted with ESI-Q-TOF HRMS operated in negative mode. Figure S-3. High resolution mass spectra of baicalin and its oxidized product caused by oxidation reaction on HPLC columns. Column A was Ultimate® AQ-C18 column (4.6×150 mm, 3 µm, Welch materials, Shanghai, China), and column B was Waters XBridgeTM C8 column (4.6×250 mm, 5 µm, Milford, MA, USA). The detection was conducted with ESI-Q-TOF HRMS operated in negative mode. Figure S-4. Extracted diode array detector (DAD) spectra at the retention times of baicalin, baicalein and their on-column oxidized with the wavelength range of 210-400 nm. Figure S-5. Chromatograms of baicalin and its on-column oxidized product baicalein oxide on the UPLC column without inlet frit (left panel) and the column without both the inlet and outlet frits (right panel). The used UPLC column was Waters BEH C18 column (2.1×50 mm, 1.7 µm). Figure S-6. The ratio of the signal intensity of the oxidized product (baicalin oxide/ baicalein oxide) to the summational signal intensity of corresponding oxidized product and unchanged parent analyte (baicalin/baicalein) with different elution flow rates. The used UPLC column was Waters BEH C18 column (2.1×50 mm, 1.7 µm). The ions m/z 269, m/z 271, m/z 445 and m/z 447 correspond to baicalein oxide, baicalein, baicalin oxide and baicalin, respectively. The blue bars link to the precursor ions of baicalein and baicalein oxide which were generated during ESI process by baicalin and baicalin oxide, respectively. The green bars correspond to the intact parent analyte baicalein and the oxidized product baicalein oxide generated on the UPLC column. Figure S-7. Chromatograms of baicalin, baicalein and their on-column oxidized products on the UPLC column used in reversed flow direction. The used UPLC column was Waters BEH C18 column (2.1×50 mm, 1.7 µm). Figure S-8. The ratio of the signal intensity of baicalin oxide (m/z 445) to the summational signal intensity of baicalin oxide and baicalin (m/z 447) with and without Page 3 the post-column infusion of reducing agent solution ((NH4)2S). The concentration of the post-column infused (NH4)2S solution was 1.7 mM. The solution was prepared with ultra-pure water. The flow rates of the post-column infusion were set from 5 μL/min to 50 μL/min. Figure S-9. The ratio of the signal intensity of baicalein oxide (m/z 269) to the summational signal intensity of baicalein oxide and baicalein (m/z 271) with and without the post-column infusion of reducing agent solution ((NH4)2S). The concentration of the post-column infused (NH4)2S solution was 1.7 mM. The solution was prepared with ultra-pure water. The flow rates of the post-column infusion were set from 5 μL/min to 50 μL/min. Figure S-10. The ratio of the signal intensity of baicalin oxide (m/z 445) to the summational signal intensity of baicalin oxide and baicalin (m/z 447) with and without the post-column infusion of reducing agent solutions tested in this study. The reducing agent solutions contained the respective agent with the concentration of 10 mM, excepted for (NH4)2S, of which the concentration was 1.7 mM. These solutions were prepared with ultra-pure water. The post-column infusion flow rates of the reducing agents were transformed into nmol/min based on the calculation with the concentrations and flow rates of the post-column infusion. Figure S-11. The ratio of the signal intensity of baicalein oxide (m/z 269) to the summational signal intensity of baicalein oxide and baicalein (m/z 271) with and without the post-column infusion of reducing agent solutions tested in this study. The reducing agent solutions contained the respective agent with the concentration of 10 mM, excepted for (NH4)2S, of which the concentration was 1.7 mM. These solutions were prepared with ultra-pure water. The post-column infusion flow rates of the reducing agents were transformed into nmol/min based on the calculation with the concentrations and flow rates of the post-column infusion. Figure S-12. General illustration of the main findings of this study: oxidation reaction on UPLC column and reduction reaction on ESI source. Page 4 Table captions Table S-1. Chemical information of the investigated model compounds. Table S-2. Gradient elution program I. Table S-3. Gradient elution program II. Table S-4. Mass spectrometry working conditions. Table S-5. Exact mass spectrometry data of the investigated model compounds and their degradation products caused by on-column oxidation reaction. Page 5 Tables Table S-1. Chemical information of the investigated model compounds. Name Baicalin Molecular Exact molecular formula weight (u) C21H18O11 446.0849 CAS No Chemical structure 21967-41-9 OH O HO Baicalein C15H10O5 270.0528 491-67-8 HO O HO Propyl gallate (PG) O C10H12O5 212.0685 121-79-9 HO O HO Rutin C27H30O16 610.1534 153-18-4 C21H20O11 448.1006 522-12-3 C27H32O14 580.1792 10236-47-2 C20H22O9 406.1264 82373-94-2 quercetin-3rhamnoside (QR) Naringin 2,3,5,4’Tetrahydroxy stilbene-2-Οβ-D-glucoside (THS-G) Page 6 Table S-2. Gradient elution program I. Step Total time (min) Flow rate (µL/min) MPA (%) MPB (%) 1 Initial 400 70 30 2 1.5 400 30 70 3 1.6 400 70 30 4 2.5 400 70 30 Table S-3. Gradient elution program II. Step Total time (min) Flow rate (µL/min) MPA (%) MPB (%) 0 Initial 400 70 30 1 3.5 400 30 70 2 3.6 400 70 30 3 5 400 70 30 Table S-4. Mass spectrometry working conditions. Parameter Value Trap Collision Energy 6 Transfer Collision Energy 4 Collision adduced dissociation Medium Capillary Voltage 2500 V Cone voltage 35 V Desolvation temperature 400 oC Ion source temperature 100 oC Cone Gas 1 50 L/h Desolvation gas 600 L/h Mass scan range 100-1000 u Acquisition mode centroid Scan time 0.2 s Lock Mass 554.2615 u Acquisition mode V mode Page 7 Table S-5. Exact mass spectrometry data of the investigated model compounds and their degradation products caused by on-column oxidation reaction. Name Precursor ion (In ESI+, [M+H]+) Chemical formula Theoretical m/z (u) [1] Precursor ion (In ESI-, [M-H]-) Detected Mass accuracy Chemical Theoretical m/z (u) (ppm) formula m/z (u) [1] Detected Mass accuracy m/z (u) (ppm) Baicalin C21H19O11 447.0927 447.0960 7.4 C21H17O11 445.0776 445.0789 4.0 Baicalin oxide C21H17O11 445.0771 445.0799 6.3 C21H15O11 443.0614 443.0562 -11.7 Baicalein C15H11O5 271.0606 271.0632 9.6 C15H9O5 269.0455 269.0450 -1.9 Baicalein oxide C15H9O5 269.0451 269.0451 0 C15H7O5 267.0294 267.0301 2.6 PG NA NA NA NA C10H11O5 211.0607 211.0616 4.3 PG oxide NA NA NA NA C10H9O5 209.0450 209.0472 10.5 Rutin NA NA NA NA C27H29O16 609.1456 609.1451 -0.8 Rutin oxide NA NA NA NA C27H27O16 607.1299 607.1323 4.0 QR NA NA NA NA C21H19O11 447.0927 447.0915 -2.7 QR oxide NA NA NA NA C21H17O11 445.0771 445.0766 -1.1 Naringin NA NA NA NA C27H31O14 579.1714 579.1691 -4.0 THS-G NA NA NA NA C20H21O9 405.1186 405.1165 -5.2 [1] Patiny, L., Borel, A. ChemCalc: A building block for tomorrow’s chemical infrastructure. Journal of chemical information and modeling, 2013, 53(5), 1223-1228. Page 8 Figures Figure S-1. Workflow of the finding, confirmation and explanation of the redox reaction on UPLC column. Notes: [M-G-H]+ is the product ion of the precursor ion [MH]+ losing a glucuronide (G) group, and [M-G-3H]- is the product ion of the precursor ion [M-3H]- losing a G group; VC: ascorbic acid. Page 9 Figure S-2. High resolution mass spectra of baicalin and its oxidized product caused by on-column oxidation reaction. The precursor ions of baicalein and baicalein oxide were generated during collision induced dissociation (CID). The detection was conducted with ESI-Q-TOF HRMS operated in negative mode. Page 10 Figure S-3. High resolution mass spectra of baicalin and its oxidized product caused by oxidation reaction on HPLC columns. Column A was Ultimate® AQ-C18 column (4.6×150 mm, 3 µm, Welch materials, Shanghai, China), and column B was Waters XBridgeTM C8 column (4.6×250 mm, 5 µm, Milford, MA, USA). The detection was conducted with ESI-Q-TOF HRMS operated in negative mode. Page 11 Figure S-4. Extracted diode array detector (DAD) spectra at the retention times of baicalin, baicalein and their on-column oxidized products with the wavelength range of 210-400 nm. Page 12 Figure S-5. Chromatograms of baicalin and its on-column oxidized product baicalein oxide on the UPLC column without inlet frit (left panel) and the column without both the inlet and outlet frits (right panel). The used UPLC column was Waters BEH C18 column (2.1×50 mm, 1.7 µm). Page 13 Signal intensity of oxidized product/(signal intensity of oxidized product+signal intensity of intact analyte) 1 Ratio 269/(269+271) Ratio 445/(445+447) Ratio 269/(269+271) (Baicalein) 0.9 0.8 0.7 Ratio 0.6 0.5 0.4 0.3 0.2 0.1 0 50 μL/min 200 μL/min 300 μL/min 400 μL/min 600 μL/min Flow rate Figure S-6. The ratio of the signal intensity of the oxidized product (baicalin oxide/baicalein oxide) to the summational signal intensity of corresponding oxidized product and unchanged parent analyte (baicalin/baicalein) with different elution flow rates. The used UPLC column was Waters BEH C18 column (2.1×50 mm, 1.7 µm). The ions m/z 269, m/z 271, m/z 445 and m/z 447 correspond to baicalein oxide, baicalein, baicalin oxide and baicalin, respectively. The blue bars link to the precursor ions of baicalein and baicalein oxide which were generated during ESI process by baicalin and baicalin oxide, respectively. The green bars correspond to the intact parent analyte baicalein and the oxidized product baicalein oxide generated on the UPLC column. Page 14 Figure S-7. Chromatograms of baicalin, baicalein and their on-column oxidized products on the UPLC column used in reversed flow direction. The used UPLC column was Waters BEH C18 column (2.1×50 mm, 1.7 µm). Page 15 Signal intensity of m/z 445/(signal intensity of m/z 445+signal intensity of m/z 447) 0.7 0.6 Ratio 0.5 0.4 0.3 0.2 0.1 0 Non-infusion 5 μL/min 10 μL/min 20 μL/min 30 μL/min 40 μL/min 50 μL/min Flow rate of post-column infusion of (NH4)2S solution (1.7 mM in pure water) Figure S-8. The ratio of the signal intensity of baicalin oxide (m/z 445) to the summational signal intensity of baicalin oxide and baicalin (m/z 447) with and without the post-column infusion of reducing agent solution ((NH4)2S). The concentration of the post-column infused (NH4)2S solution was 1.7 mM. The solution was prepared with ultra-pure water. The flow rates of the post-column infusion were set from 5 μL/min to 50 μL/min. Page 16 Signal intensity of m/z 269/(signal intensity of m/z 269+signal intensity of m/z 271) 1 0.9 0.8 0.7 Ratio 0.6 0.5 0.4 0.3 0.2 0.1 0 Non-infusion 5 μL/min 10 μL/min 20 μL/min 30 μL/min 40 μL/min 50 μL/min Flow rate of post-column infusion of (NH4)2S solution (1.7 mM in pure water) Figure S-9. The ratio of the signal intensity of baicalein oxide (m/z 269) to the summational signal intensity of baicalein oxide and baicalein (m/z 271) with and without the post-column infusion of reducing agent solution ((NH4)2S). The concentration of the post-column infused (NH4)2S solution was 1.7 mM. The solution was prepared with ultra-pure water. The flow rates of the post-column infusion were set from 5 μL/min to 50 μL/min. Page 17 Signal intensity of m/z 445/(signal intensity of m/z 445+signal intensity of m/z 447) 0.9 0.8 0.7 Ratio 0.6 0.5 0.4 0.3 0.2 0.1 0 Flow rate of post-column infusion of reducing agent solutions (disolved in pure water) Figure S-10. The ratio of the signal intensity of baicalin oxide (m/z 445) to the summational signal intensity of baicalin oxide and baicalin (m/z 447) with and without the post-column infusion of reducing agent solutions tested in this study. The reducing agent solutions contained the respective agent with the concentration of 10 mM, excepted for (NH4)2S, of which the concentration was 1.7 mM. These solutions were prepared with ultra-pure water. The post-column infusion flow rates of the reducing agents were transformed into nmol/min based on the calculation with the concentrations and flow rates of the post-column infusion. Page 18 Signal intensity of m/z 269/(signal intensity of m/z 269+ signal intensity of m/z 271) 1.2 1 Ratio 0.8 0.6 0.4 0.2 0 Flow rate of post-column infusion of reducing agent solutions (disolved in pure water) Figure S-11. The ratio of the signal intensity of baicalein oxide (m/z 269) to the summational signal intensity of baicalein oxide and baicalein (m/z 271) with and without the post-column infusion of reducing agent solutions tested in this study. The reducing agent solutions contained the respective agent with the concentration of 10 mM, excepted for (NH4)2S, of which the concentration was 1.7 mM. These solutions were prepared with ultra-pure water. The post-column infusion flow rates of the reducing agents were transformed into nmol/min based on the calculation with the concentrations and flow rates of the post-column infusion. Page 19 Figure S-12. General illustration of the main findings of this study: oxidation reaction on UPLC column and reduction reaction on ESI source. Oxidation reactions of polyphenolic compounds can occur on UPLC columns, which could pose negative impact to the quantitative and qualitative analysis of these compounds. Fortunately, the oxidation products can be reduced back to their parent forms during the ESI process with the presence of reducing agents.