Chapter 16 Assignment
advertisement

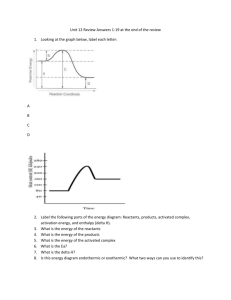
Chemistry 621 Chapter 16 Assignment #1 1. Define and distinguish among the following terms: thermal energy, potential energy, heat, and temperature. 2. Examine the following data collected from a chemistry lab: Mass of empty cup Mass of cup + water Mass of cup + water + metal Initial temperature of water Initial temperature of metal Final temperature of system 2.31 g 180.89 g 780.89 g 17.0°C 52.0°C 27.0°C Using the above data, calculate the following (show all work): (a) Mass of water (b) Mass of metal (c) Temperature change of water (d) Temperature change of metal (e) Heat lost or gained by water (f) The specific heat capacity of the metal 3. (a) Explain why evaporation is a cooling process. (b) If all the molecules in a glass of water at room temperature had the same kinetic energy, would the water evaporate? Explain your answer. 4. a) 5 g of iron at 75.0°C is added to 150.0 g of water at 15.0°C in a calorimeter. What is the final temperature of the iron and the water? (For iron, c = 0.444 J/g°C.) b) A mixture is made by adding 75 g of an unknown liquid at a temperature of 25oC to 60 g of water at a temperature of 90oC. The final temperature of the mixture is 65oC. Calculate the specific heat capacity of the liquid. 5. Consider the following equation: 2C2H2(g) + 5O2(g) 4CO2(g) + 2H2O(g) + energy (a) Does the equation represent an endothermic reaction or an exothermic reaction? Justify your answer. (b) What effect would this reaction have on the temperature of its surroundings? Compare the temperature of the surroundings before the reaction takes place to the temperature of the surroundings after the reaction takes place. (c) What does the equation communicate about the relative energy of the chemical bonds in the reactants and products? 6. Write balanced thermochemical equations for the following word equations. Include the heat term in the equation. (a) In the neutralization of nitric acid solution with aqueous potassium hydroxide to form potassium nitrate and water, 53.4 kJ/mol of heat are produced. (b) When water evaporates, 44.0 kJ/mol of heat is absorbed. (c) Graphite reacts in excess oxygen to form carbon dioxide, releasing 393.5 kJ of heat for each mol of graphite that reacts. 7. 8. Draw enthalpy diagrams for each equation you wrote in question 6. Liquid hydrogen peroxide is an oxidizing agent in many rocket fuel mixtures because it releases oxygen gas on decomposition: 2H2O2(l) 2H2O(l) + O2(g) ∆H = –196.1 kJ How much heat is released when 650 kg H2O2(l) decomposes? 9. A glass containing 140.0 g of water at 22.8°C is placed in a refrigerator. Eventually, the water becomes ice at –10.0 °C. (a) Draw a cooling curve for this process. (b) Calculate the quantity of heat removed from the water. specific heat capacity of water is 4.184 J/g°C specific heat capacity of ice is 2.01 J/g°C molar enthalpy of freezing of water = –6.02 kJ/mol