check list of registration file for vaccine
advertisement

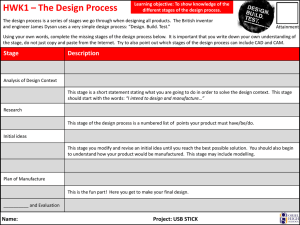
Islamic Republic of Iran Ministry of Health and Medical Education Food and Drug Organization Title: check list of registration file for vaccine Type: √ Finalized Draft No.: Date of Issue: Document No.: 102-62-03 Name of Office: Biologic,Deputy for Drug Product name: Brand name: Drug form (compatible with official index of Iranian drugs): Product strength (dose or potency) Manufacturer name and address: Importer name and address 1 General information regarding the product: Number and volume of packaging Storage and transportation conditions Route of injection (if applicable) Indications and volume of injection compared to available references Necessary cautions about the product Description of cold chain Result of assessment 2 Information regarding the manufacture process Formulation Amount of each constituent per one drug unit Function of each constituent Health certificate of substances used in manufacture for BSE agent, if applicable Health certificate of plasma and albumin analysis sheet, if applicable Formulation amounts for one batch List of substances used during manufacture and purification 1 Manufacture process, including production, isolation and purification - In diagrams - Description of steps Controls during manufacture Method of product sterilization Evaluation of manufacture process with mention of results List of important equipment of production line Information regarding reprocessing Results of tests of production homogeneity Result of assessment 3- Reagents used in productions (Biologic substances used in the manufacture process, e.g. monoclonal antibodies for purification, enzymes etc.) Name of substances Name of purchase site Nature and function of substances in manufacture Results of tests conducted Protocol for re-use of biologic reagents Complete file indicating lack of virus in reagent, if necessary (e.g. monoclonal antibodies for purification) Result assessment 4- Information regarding primary substances: 4-1 viral vaccines Strain used (seed) - Information - History - Registration number in NCA of country of origin - Information of primary cell bank (MSB) - Information of secondary cell bank (WSB) Primary cell culture - Information - History - Registration number in NCCA f country of origin - Results of tests conducted - Information of primary cell bank (MSB) - Information of secondary cell bank (WSB) Origin and results of tests conducted on the serum used in cell culture Health certificate of serum for BSE agent 4-2 Microbial vaccines Primary and secondary seed - Information - History - Results of tests conducted Information of culture medium - Complete composition of culture medium - Analysis sheet of substances used with mention of valid reference - Results of tests conducted on culture medium 2 - If the vaccine consists of different constituents, information for each constituent must be provided separately and completely Result of assessment 4-3 Control of auxiliary substances (including all substances used in production, purification and formulation) Reference of substance control in accordance with international pharmacopeias or submission of SOP and validation Analysis sheet from product manufacturer Health certificate of substances for BSE agent Result of assessment 5- Control of intermediate substances (including primary harvests, purified bulk, absorbed bulk and final bulk) Method of control Mention of valid reference for control or submission of SOP and validation Result of assessment 6 Control of final product Method of control Valid international reference for control Validation of control methods Analysis sheet Result of assessment 7- Information regarding stability Batch numbers tested, alongside manufacture date, site and size of batch Test procedure - Expedited - Periodic Storage conditions of tested samples Packaging type of samples Duration of stability Name and address of laboratory where tests are conducted Test results (numerical results, chromatograms, gel photographs, peptide maps, etc) Result of assessment 8 Information regarding packaging Type and material of packaging Analysis sheet of packaging materials Sample color label Control of packaging instructions in accordance with valid references Sample packaging vessel Sample brochure Result of assessment 3 9 Information regarding clinical and subclinical studies Study methods Formula used in studies Results Reference Result of assessment 10- File of the solvent used (including: formulation, analysis sheet of substances and final product, results of stability tests on solvent, results of stability tests after filling to volume, sample solvent packaging and label) Result of assessment 11- Sample manufacture protocol summary alongside sample signatures of releases authorities Results of assessment Result of final assessment Date of assessment: Name and signature of assessing expert: Name and signature of expert in-charge 4