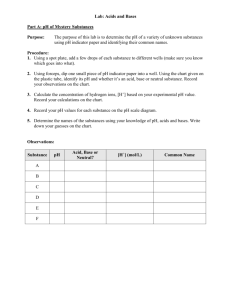
Types of Chemical Reactions Lab

Part B:
Part A: Reactants: Magnesium and Oxygen
2. Light the Bunsen burner.
3. Observe the magnesium and record your observations in your table
4. Get a strip of magnesium out of the bag and hold the magnesium in the flame with the tongs until it lights. When it starts to burn on its own, hold it over the aluminum foil. Turn off the
Bunsen burner. Record your observations, noting the changes it goes through.
5. Classify the type of reaction and predict the products based on the type of reaction.
5. Leave the magnesium “scraps” on the aluminum foil, but wipe off the tongs with a paper towel and throw the paper towel away!
Reactant: Peroxide (H
2
O
2
)
1. Place approximately 5 mL of H
2. Add a SMALL scoop of MnO
2
.
2
O
2
in a test tube. Record your observations.
3. Record your observations. Feel the test tube.
4. Classify the type of reaction and predict the products based on the type of reaction.
5. Wash out the test tube and clean off any materials you used in the sink. Wipe off the table if anything spilled and throw away the paper towels.
Part C: Reactants: Zinc and Hydrochloric Acid
1. Place a piece of zinc into a clean, dry test tube. Record your observations.
2. One person needs to put gloves on. That person will pour approximately 3-4 mL of HCl into the test tube. Cover the end of the test tube for 10-20 seconds with your thumb. Do not allow
the HCl to come into contact with your skin! Record your observations.
3. Have your lab partner light a wooden splint and place the burning splint directly above the test tube. Record your observations.
4. Classify the type of reaction and predict the products based on the type of reaction.
5. Pour the contents of the test tube in the waste container. Wash out the graduated cylinder and throw away the wooden splint after you wet it to make sure it completely out.
Part D: Reactants: lead (II) nitrate and potassium iodide
1. Place 4-5 drops of lead(II) nitrate into the middle test tube.
2. Add 4-5 drops of potassium iodide into the middle test tube. Record your observations.
3. Classify the type of reaction and predict the products based on the type of reaction.
4. Wash out middle test tube with water in the sink.