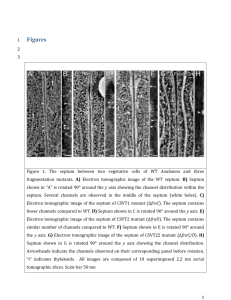
Requirement of FraG for channel formation between vegetative cells
advertisement

1 Requirement of Fra proteins for communication channels between cells in the filamentous 2 nitrogen-fixing cyanobacterium Anabaena sp. PCC 7120 3 4 5 6 Amin Omairi-Nassera, Vicente Mariscalb, Jotham Austin IIa,c, and Robert Haselkorna* 7 8 a 9 University of Chicago, Chicago, Illinois 60637, USA. Department of Molecular Genetics and Cell Biology and cAdvanced Electron Microscopy Facility, The 10 b 11 Universidad de Sevilla, E-41092 Seville, Spain. Instituto de Bioquímica Vegetal y Fotosíntesis, Consejo Superior de Investigaciones Científicas and 12 13 14 A.O.-N. and V.M. contributed equally to this work. 15 *Corresponding author, rh01@uchicago.edu 16 17 18 19 20 21 Author contributions: R.H. designed research and supervised the work; A.O.-N. and V.M. performed research and analyzed the results; J.A. supervised the work and analyzed data; all authors contributed to writing and editing the paper. 22 1 23 Abstract 24 The filamentous nitrogen-fixing cyanobacterium Anabaena sp. PCC 7120 differentiates 25 specialized cells, heterocysts, that fix atmospheric nitrogen and transfer the fixed nitrogen to 26 adjacent vegetative cells. Reciprocally, vegetative cells transfer fixed carbon to heterocysts. 27 Several routes have been described for metabolite exchange within the filament, one of which 28 involves communicating channels that penetrate the septum between adjacent cells. Several fra 29 gene mutants were isolated 25 years ago on the basis of their phenotypes: inability to fix nitrogen 30 and fragmentation of filaments upon transfer from N+ to N- media. Cryopreservation combined 31 with electron tomography, were used to investigate the role of three fra gene products in channel 32 formation. FraC and FraG are clearly involved in channel formation while FraD has a minor part. 33 Additionally, FraG was located close to the cytoplasmic membrane and in the heterocyst neck, 34 using immunogold labeling with antibody raised to the N-terminal domain of the FraG protein. 35 Significance 36 Cellular communication along the filaments of heterocyst-forming, nitrogen-fixing cyanobacteria 37 has been discussed for at least 50 years but how this might be accomplished is not fully 38 understood. We recently showed that the septum between heterocysts and vegetative cells is 39 pierced by channels 12 nm in diameter and 20 nm long. Here, we show that three proteins, FraC, 40 FraD and FraG, participate in the formation of the channels although none of them appears to be 41 a structural component of the channels. Moreover, using gold particle-labeled antibody, FraG was 42 found around the cyanophycin plug as well as associated with the cytoplasmic membrane in the 43 neighborhood of the peptidoglycan that forms the septum. 2 44 Introduction 45 Cyanobacteria are phototrophic microbes that bear a gram-negative cell envelope and are 46 capable of oxygenic photosynthesis. Some cyanobacteria, such as the filamentous Anabaena sp. 47 strain PCC 7120 (hereafter called Anabaena), are capable of fixing atmospheric N2 when grown 48 in media lacking combined nitrogen. Nitrogen fixation occurs in heterocysts, specialized cells 49 that differentiate from vegetative cells along the filaments and provide a micro-oxic environment 50 for the process (1). One long-standing attraction of Anabaena is its beautiful pattern of 51 differentiation: new heterocysts differentiate midway between two heterocysts as the distance 52 between them doubles due to division of the vegetative cells. This organism, which belongs to 53 one of the first prokaryotic groups on earth to have evolved multicellularity, had to develop 54 structures for intercellular communication. Intercellular communication between heterocysts and 55 vegetative cells comprises small molecules, such as sucrose moving from vegetative cells to 56 heterocysts (2–5) and a dipeptide, -aspartyl-arginine, moving from heterocysts to vegetative 57 cells (6, 7). The mechanism of communication between heterocysts and vegetative cells has been 58 debated for the last 50 years. Two pathways have been proposed for such exchanges (1, 8–10). 59 One is through the periplasm, suggested by the continuity of the outer membrane surrounding the 60 entire filament (9, 11, 12). The other proposed means of communication requires structures 61 between adjacent cells in the filament. Several structures connecting vegetative cells and 62 heterocysts and vegetative cells with each other have been observed using freeze-fracture, 63 conventional electron microscopy and cryo fixation with electron tomography (13–17). Different 64 names have been given to these structures: microplasmodesmata, septosomes, septal junctions, or 65 nanopores (12, 13, 18, 19). Using cryopreservation combined with electron tomography, we 66 observed structures we call channels traversing the peptidoglycan layer in Anabaena (19). These 67 channels are 12 nm long with a diameter of 12 nm, in the septa between vegetative cells. Longer 68 channels, 21 nm long with a similar diameter of 12 nm, were seen in the septa between vegetative 69 cells and heterocysts (20). 70 Several Anabaena gene products were proposed to be involved specifically in intercellular 71 communication. Three were characterized initially from a large set of mutants selected on the 72 basis of their inability to fix nitrogen (21). These mutants manifest a fragmentation phenotype, 73 meaning that they fragment into short filaments upon transfer to liquid medium lacking combined 74 nitrogen, after which they die (15, 22, 23). Further characterization of these mutants led to 3 75 uncovering a role for several fra gene products in intercellular molecular transfer (23–25). 76 fraC encodes a 179-amino acid protein with three predicted trans-membrane segments; 77 fraD encodes a 343-amino acid protein with five predicted trans-membrane segments and a 78 coiled-coil domain; and fraG (also called sepJ) encodes a 751-amino acid protein predicted to 79 have an N-terminal coiled-coil domain, an internal linker domain, and a C-terminal permease-like 80 domain with either 10 trans-membrane segments (22) or 9 or 11 trans-membrane segments (26). 81 fraG deletion prevents heterocyst differentiation and glycolipid layer formation, while the 82 deletion of either fraC or fraD allows heterocyst differentiation, but the heterocysts formed show 83 an aberrant neck and do not fix nitrogen (23, 25). Using GFP tags, FraC, FraD and FraG proteins 84 were shown to be located in the septum between cells (23, 26). FraD was further localized to the 85 septum by immunogold labeling using an antibody raised against the N-terminal coiled-coil part 86 of FraD (25). Fluorescence recovery after photobleaching (FRAP) experiments showed 87 impairment in cell-cell transfer of small molecules such as calcein (622-Da) and 5- 88 carboxyfluorescein (374-Da) in fraC, fraD and fraG mutants, further indicating a role of these 89 gene products in intercellular communication (23–25). 90 In the work reported here, cryopreservation combined with electron tomography was used 91 to investigate the role of these three fra gene products in channel formation. We found that FraC 92 and FraG are clearly required for channel formation, whereas FraD plays a minor role. 93 Immunogold labeling with antibody to the N-terminal coiled-coil domain of FraG yielded an 94 improved localization for FraG. 95 4 96 Results 97 Roles of FraC and FraD in channel formation between vegetative cells 98 In earlier studies, three deletion mutant strains CSVT1 (∆fraC) CSVT2 (∆fraD), and 99 CSVT22 (double mutant ∆fraC/D) revealed the same fragmentation phenotype: upon transfer to 100 N- medium, the filaments fell apart (15, 23, 25). However, in nitrogen-rich media, these mutants 101 did not show any morphological alteration when examined by light microscopy, although transfer 102 of calcein and 5-CFDA between cells was hampered. In addition, GFP fusions to wild-type FraC 103 and FraD allowed localization of both proteins to the septum connecting vegetative cells. 104 Additionally, immunogold labeling of FraD confirmed its location in the septum (23, 25). 105 These observations prompted us to investigate the channels in these mutants under 106 nitrogen-replete conditions. We examined 2.2-nm tomographic sections of septa between 107 vegetative cells of the ∆fraC, ∆fraD and ∆fraC/D mutants in three or four tomographic volumes 108 for each strain, including some tomograms covering the whole septum (serial tomograms). We 109 present here only the middle part of the septum between two vegetative cells (Fig. 1). (For the 110 entire septum see supplement Fig. S1). The septum of CSVT1 (∆fraC) shows fewer and wider 111 channels than the WT (Fig. 1A to 1D). Note that the septum in each tomogram corresponds to a 112 200- to 300-nm section of the entire septum. The dimensions and frequency of channels in the 113 septa of CSVT2 (∆fraD) were similar to those observed in the WT (Fig. 1E and 1F). The septum 114 between vegetative cells in CSVT22 (∆fraC/D) displays only a single channel, this one appearing 115 to be wider than those observed in WT (Fig. 1G and 1H) (other tomograms of CSVT22 show two 116 to three channels in the septum). When the septum is rotated 90° around the y-axis, it is clear that 117 CSVT1 and the double mutant contain about 90% fewer channels than the WT. In ∆fraC, as well 118 as in the double mutant, the length of the channels is 12 nm, which is similar to WT, while the 119 diameter is 21 nm, noticeably wider than WT (Table 1). Based on these observations we conclude 120 that FraC plays a role, possibly structural, in assembly of the channels, while FraD does not. 121 The heterocyst-vegetative cell septa in the ∆fraC/D double mutant 122 Although CSVT22 (∆fraC/D) produces heterocysts in response to nitrogen limitation, it is 123 not able to grow diazotrophically. This phenotype may result from an altered structure of the 124 heterocyst/vegetative cell septa as in the single mutants (25). Four tomograms for the mutant 125 vegetative cell-heterocyst junctions, including one tomogram covering the whole junction (three 5 126 serial tomograms), were analyzed. The cup-like structure typical of the WT heterocyst neck is 127 missing in the CSVT22 strain, consistent with previously reported results (25). In addition, the 128 septum in the ∆fraC/D mutant appears to be three to four times wider than in the WT (82 ±25 nm 129 in the mutant compared to 21 nm in WT; Fig. 2, Table 1). The septum also contains fewer 130 channels than WT, one to four channels per septum, compared to ~20 in WT. These results 131 resemble those for the channels in the septa between vegetative cells of the same mutant, as 132 described in the previous section. 133 134 135 Requirement of FraG for channel formation between vegetative cells 136 tomography. We examined 2.2-nm tomographic sections of septa between vegetative cells of the 137 ∆fraG mutant, grown in complete medium, in eight tomographic reconstructions. Only three of 138 the eight reconstructed tomograms showed a few (3-4) channels in the septum (Fig. 3C), 139 compared to 15-20 channels in each of the five WT tomograms. The channels are seen clearly 140 when the tomographic volume is rotated 90° around the y axis, where they appear as white holes 141 in the dark background of the septum. CSVM34 (∆fraG) shows many fewer channels compared 142 to WT (Fig. 3B and 3D). The dimensions of the channels observed in ∆fraG are not statistically 143 different from those of WT (Table 1). These results suggest that FraG might provide a dock for 144 initiating channel assembly, to be discussed below. We also studied the septum structure in strain CSVM34 (∆fraG) (27), by electron 145 146 FraG is localized around the cyanophycin mass in the heterocyst 147 Previous studies, using GFP-tagged FraG, localized FraG to the intercellular septa, but 148 optical microscopy lacks the resolution needed to define this location precisely. To locate FraG 149 better, we immunolabeled samples of the WT strain (grown with or without combined nitrogen) 150 using antibody raised against the FraG N-terminal coiled-coil domain (anti-FraG_CC) and a 10- 151 nm gold-labeled secondary antibody (27). Very few gold particles were observed in cells grown 152 under N+ conditions (supplement Fig. S2), even in septa where FraG-GFP had been localized by 153 confocal microscopy (26). The specificity of the gold particles in the septum could not be 154 confirmed in comparison with gold particles detected in the background, probably due to the low 155 expression level of FraG. This point will be elaborated in the next paragraph. However, with 156 samples grown under nitrogen-fixing conditions, labeling was observed in the heterocyst neck, 6 157 around the cyanophycin mass, that is, close to the heterocyst-vegetative cell junction. Only a few 158 gold particles were seen on the larger part of the cyanophycin that is close to the polar thylakoid 159 mass. As was the case with cells grown under N+ conditions, very few gold particles were 160 identified in the septum between vegetative cells and between heterocysts and vegetative cells. 161 Figure 4B shows a control experiment in which WT samples were incubated with the secondary 162 gold-labeled antibody alone, without the FraG primary antibody. No gold particles were detected 163 in this control experiment, which indicates that the signal we detect is due to the FraG antibody. 164 The cells shown in Figures 4A and 4B correspond to samples in which the cyanophycin has 165 dropped out during the preparation of the sample. The absence of cyanophycin must have made 166 the FraG-antigenic domain accessible to the antibody in the heterocyst neck. This hypothesis was 167 confirmed by immunogold labeling of cells with intact cyanophycin, which showed very few 168 bound gold particles (supplement Fig. S3). 169 In light of these results, we reinvestigated the C-terminal GFP-tagged FraG localization in 170 heterocysts, using 3D deconvolution fluorescence microscopy in the strain CSAM137 (26). The 171 fluorescence is spread through the outermost part of the heterocyst neck. These results agree with 172 the immunolocalization of FraG in the heterocyst neck around the cyanophycin mass (Fig. 4A). 173 Figure 4F shows two distinguishable spots at each pole of the cell, suggesting the presence of 174 FraG-GFP towards the vegetative cell, as well as around the cyanophycin and more specifically 175 in the part that is present in the heterocyst neck. To further investigate FraG localization around 176 the cyanophycin mass, we collected tomograms for the immunogold-labeled samples (Fig. 5 A- 177 C). The immunotomogram with anti-FraG_CC shows the gold particles around the cyanophycin. 178 Figures 5A to 5C show gold particles at different depths around the cyanophycin mass, between 179 the heterocyst membrane and the cyanophycin. Figure 5D shows the distribution of gold particles 180 around the cyanophycin mass, confirming the localization of FraG in the heterocyst neck. Note 181 that, in the EM images, the cyanophycin always appears split into two parts. The gap is probably 182 due to the cyanophycin breakage that was observed in all heterocysts in this study and in previous 183 studies (20). 184 185 186 7 187 188 189 FraG immunogold localization in the septum between vegetative cells 190 expression of FraG. In order better to localize FraG between vegetative cells, we constructed a 191 mutant, W30, overexpressing FraG. The mutant did not show any phenotypic difference 192 compared to WT (same growth rate in N+ and same pattern and morphology for differentiating 193 heterocysts). Western blots of W30 extracts show a 7-fold increase in the amount of FraG 194 compared to WT (Supplement Fig. S4). Immunogold labeling of W30 using anti-FraG_CC shows 195 the N-terminal domain mainly on the edge of the septa between vegetative cells (Fig. 6A and 6C) 196 distributed close to the cytoplasmic membrane. More than 100 cells were analyzed and all of 197 them show similar gold distribution. (See supplement Fig. S5 for more cells). The septal 198 localization of the native FraG protein corroborates the data obtained with the FraG-GFP fusion 199 protein (26). The specificity of the signal in W30 was confirmed by immunolabeling the deletion 200 mutant CSVM34, in which no signal was detected (Fig. 6B). The weak gold signals in the WT vegetative cell septa could be explained by the low 201 202 203 204 FraG topology 205 different cyanobacteria predict 9, 10 or 11 trans-membrane segments (28). Our analyses for FraG 206 topology in Anabaena using interPro, an integrated database of predictive protein signatures (29) 207 and Protter (30) supported a 10-transmembrane span model (supplement Fig. S6). This fact 208 affects localization of the coiled-coil domain, which has been described as essential for the 209 function of the protein (26). Topology prediction of FraG is not clear, since analysis of homologous sequences from 210 To investigate FraG topology experimentally, we fused GFP to the first 391 amino acids 211 of FraG. This sequence includes the N-terminal domain and most of the linker domain of FraG, 212 but excludes all potential trans-membrane domains (Fig. 7A). The construct, called CCL-GFP 213 (coiled-coil-Linker-GFP), was expressed in WT strain yielding strain WGF. Additionally, to 214 exclude any localization due to interaction with the WT FraG, the reporter construct was 215 expressed in a ∆fraG background producing strain ∆GF. Vegetative cells of both WGF and ∆GF 216 show a signal in the division plane, forming a ring similar to the FtsZ division ring (Fig. 7B, 7D 217 and 7E). No signal was detected in the mature septa. Surprisingly, in heterocysts of WGF, the 8 218 CCL-GFP construct was also localized in the poles even though it lacks the predicted trans- 219 membrane domains of FraG (Fig. 7C). 9 220 221 Discussion 222 Septa of several fragmentation mutants that are impaired in intercellular communication 223 were examined by electron tomography. None of the fraC, fraD and fraG mutants showed a total 224 loss of channels in their septa, but rather a decrease in the number of channels and different sizes 225 for these channels were observed, especially for ∆fraC and ∆fraG, indicating that FraC and FraG 226 either affect the assembly of the channels or are involved in the assembly of different channels. 227 The difference in channel distribution between CSVT1 and CSVT2 suggest different 228 functions for FraC and FraD, respectively. fraC deletion results in the reduction of channel 229 number in the septum, which suggests that FraC is either a part of the channel structure or a 230 regulator of channel assembly. On the other hand, fraD deletion does not seem to affect channel 231 formation between vegetative cells. FraD is probably involved in maintaining a stable cell-cell 232 contact in the septum. We cannot exclude a role for FraD in recruiting components of other types 233 of channels that are not detected using our methods. The ∆fraC/D double mutant (CSVT22) 234 showed a 7-fold increase of the width of the septa between vegetative cells and heterocysts 235 compared to the 2-fold increase observed in corresponding WT septa. These results suggest that 236 FraC or FraD or both play a role in expansion of the peptidoglycan and the channels that connect 237 heterocysts with vegetative cells. Aberrant heterocyst neck structures were previously shown in 238 fraC and fraD mutants (25), suggesting that FraC and FraD are important for maintaining a tight 239 junction at the septum during the restructuring or remodeling of the peptidoglycan layer 240 throughout the heterocyst differentiation process. The heterocyst neck in these mutants lacks the 241 typical cup-like structure found in the WT. The increase in the septum width and the decrease in 242 the contact area between the heterocyst and the vegetative cell could explain the fragmentation 243 phenotype of the fraC, fraD and fraC/D mutants (15, 23, 25). 244 245 Tomograms for the septa between vegetative cells of CSVM34 (∆fraG), using cells fixed 246 with potassium permanganate, were previously analyzed (12). Structures called “septosomes”, 247 were measured to be 18 nm long in CSVM34 compared to 27 nm in WT. The septosome 248 frequency was difficult to measure due to lack of resolution. Using cryo-fixation and staining 249 with osmium tetroxide, which highlights peptidoglycan, and two-axis tomograms, we observed 10 250 channels with similar length in WT and CSVM34 although there were fewer channels in 251 CSVM34. This difference in observations suggests that different structures connecting cells 252 might be revealed when using cryo-fixation and tomography compared to the structures observed 253 in cells fixed with potassium permanganate (12). However, in agreement with our observations, 254 strain CSVM34 shows a reduced number of septal peptidoglycan nanopores (15% of the wild 255 type) (31). 256 FraG-GFP has been seen near the septum between vegetative cells (Flores et al., 2007). In 257 this work we have improved the resolution of the localization of FraG by means of immunogold 258 labeling. The N-terminal coiled-coil domain of FraG was detected close to the septum between 259 vegetative cells and in the heterocyst, in the neck around the cyanophycin mass. The positions of 260 the gold particles around the cyanophycin mass in heterocysts assign the location of the N- 261 terminal domain of FraG to a position facing the cyanophycin cavity. We cannot exclude the 262 possibility that FraG is anchored to a putative hydrophobic layer surrounding the cyanophycin 263 plug. However, due to the fact that the gold particles could be ~30 nm from their antigenic target 264 (32), the trans-membrane domains of FraG could be located in the cytoplasmic membrane with 265 its N-terminal coiled-coil domain facing toward the cyanophycin plug, as modeled in Figure 8. 266 In heterocysts, the (artefactual) lost cyanophycin apparently made FraG antigens 267 accessible to gold labeling; FraG antibodies reacted with their respective antigen within the 200- 268 nm section. In the case of vegetative cells and that of heterocysts with intact cyanophycin, 269 immunogold-labeled antibodies react only with their respective antigens that are located on the 270 surface of each section. Some gold particles can be located on the surface near the septum 271 between vegetative cells (Supplement Fig. S2) and between heterocysts and vegetative cells (Fig. 272 4A and supplement Fig. S3). In these areas, the gold-labeled secondary antibody is probably 273 bound to exposed FraG coiled coils. The distribution of the gold particles around the cytoplasmic 274 membrane between vegetative cells (Fig. 6A and 6C) suggests a localization of the N-terminal 275 closer to the cytoplasmic membrane of the cells rather than the peptidoglycan. 276 It has been established in bacteria that GFP fused to the C-terminal domain of cytoplasmic 277 membrane proteins fluoresce when the GFP is located in the cytoplasm (33), or in the periplasm 278 when it is exported by the TAT system (9). The C-terminal GFP-tagged FraG shows its 279 fluorescence signal around the heterocyst neck (Fig. 4), which indicates that the FraG C-terminal 280 domain is located in the cytoplasm. Moreover, the C-terminal GFP-tagged FraG (CCL-GFP) 11 281 containing the coiled-coil and part of the linker domain of FraG and lacking the trans-membrane 282 domain (Fig. 7A), shows its fluorescence signal in a ring in dividing vegetative cells and in the 283 heterocyst poles (Fig. 7). Since a TAT signal peptide is not detected in the N-terminal sequence 284 of FraG, GFP fluorescence indicates that the FraG N-terminal domain might be located in the 285 cytoplasm. The absence of GFP signals in all septa between vegetative cells in WGF and GF is 286 probably due to the absence of the trans-membrane domain that anchors the protein to the plasma 287 membrane. The GFP fluorescence signal in the division planes of vegetative cells in the CCL- 288 GFP construct are in agreement with recent results showing that FraG interacts with FtsQ, a 289 protein involved in cell division (34). On the other hand, the presence of the GFP signal in WGF 290 within the cyanophycin plug could be due to interaction of the CCL-GFP construct with either 291 WT FraG or the cyanophycin. 292 The Anabaena open reading frame alr2338 was first denoted fraG (22). Later, sepJ was 293 preferred because it better reflected the subcellular localization found in (26). In this work, we 294 show that the alr2338 product is not located exclusively in the septum. Therefore, we prefer fraG 295 for that gene, reflecting its phenotype. 296 12 297 Experimental procedures 298 299 Anabaena strains and growth conditions 300 Anabaena sp. strain PCC 7120 and derivative strains were grown photoautotrophically at 301 30 °C under constant white light (35 μE m-2 s-1), in a CO2 enriched atmosphere. The medium 302 composition is similar to BG11 with some modification (35). For heterocyst induction, cells were 303 harvested by centrifugation and resuspended in NO3- deficient medium, for which NaNO3 was 304 replaced by NaCl. Mutant strains CSVT1 (∆fraC), CSVT2 (∆fraD), CSVT22 (∆fraC/D), and 305 CSVM34 (∆fraG) have been described previously (23, 25, 27). 306 supplemented when appropriate with Neomycin (50 g mL-1). Strain W30 was constructed by 307 transferring pAN130 (22), a replicative plasmid containing the promoter and coding region of 308 fraG to WT Anabaena by conjugation as described (36). Growth media were 309 In order to express the CC-linker GFP-tagged FraG, a 1941-bp fragment covering the 5’ 310 non-coding region and part of the fraG orf was amplified using primers FFB 311 (GGATCCTGAAATATGAGTTATGGCTGGGGAC) 312 GGCGGAGGAGTTG), which also creates BamHI and NheI restriction sites, respectively. DNA 313 from Anabaena sp. PCC7120 was used as template. The PCR product, digested with BamHI and 314 NheI, was cloned into pRL25N (37) digested with the same enzymes, yielding pRCG. The 315 plasmid was sequenced to verify the fidelity of the PCR. pRCG was transferred to WT and to 316 CSVM34 by conjugation as described above, yielding WGF and ∆GF, respectively. 317 Embedding Anabaena for electron microscopy and FRN (GCTaGcTGGTGCA 318 Anabaena cells were harvested by centrifugation and transferred to an aluminum sample 319 holder and cryoprotected with 0.15 M sucrose. Samples were frozen in a Baltec HPM 010 high- 320 pressure freezer, then freeze-substituted in 2% osmium tetroxide (EMS), using an Automated 321 Freezing Substitution machine (ASF2, Leica), in anhydrous acetone at -80 C for 72 h. The 322 temperature was then increased from -80 C to -20 C over 12 h. Samples were then washed with 323 acetone three times at -20 C, then transferred to 4 C, held overnight, and then warmed to room 324 temperature. Samples were then infiltrated with increasing concentrations of EPON resin (5%, 13 325 10%, 25%, 50%, 75% and 100%) finally polymerized at 60 C for 24 h (see (38) for more 326 details). 327 For immunogold labeling, the high-pressure frozen samples were substituted in 0.1% 328 uranyl acetate in acetone at -80°C for 3 days and then warmed to -50°C for 12 h. After three 329 acetone rinses, samples were slowly infiltrated under controlled time and temperature conditions 330 in a Leica AFS system at -50°C with Lowicryl HM20 resin according to the following schedule: 331 5, 10, 25, 50, 75, and 100% (24 h incubation for each concentration). After the last incubation 332 with 100% HM20, samples were rinsed with fresh 100% HM20 three times, with 1h for each 333 wash. Samples were finally polymerized at the same temperature under UV light for 32 h. 334 Immunocytochemistry 335 336 Rabbit antibodies raised against the 188-amino acid coiled-coil domain of FraG were used 337 to detect FraG in Anabaena heterocysts and vegetative cells. Samples embedded in Lowicryl 338 HM20 were cut into 150-nm-thick sections and placed on Formvar-coated gold slot grids. 339 Immunocytochemistry was performed essentially as described by Otegui et al. (39). Sections 340 were blocked for 20 min with a 5% (w/v) solution of nonfat milk in TBS plus 0.1%Tween 20 341 (TBST). Primary antibodies were diluted 1:20 in a solution of 2.5% nonfat milk in TBST at room 342 temperature for 1 h. The sections were rinsed in a stream of TBS plus 0.5% Tween 20 and then 343 transferred to the secondary antibody (goat anti-rabbit IgG 1:20 in TBST) conjugated to 10-nm 344 gold particles, for 1 h. Control procedures omitted the primary antibody. For gold quantification, 345 over 1000 gold particles were counted in more than 30 pairs of cells. See supplement Fig. S7, for 346 the calculation of gold distribution in the septum vs the rest of the cell and the background. 347 Sectioning 348 349 EPON sections (100 to 300 nm) were cut using a Leica EM AFS2 Automatic Freeze- 350 Substitution Processor and collected on 1% Formvar (EMS) copper slot grids. Sections were 351 stained with 2% uranyl acetate and 0.5% lead citrate for 8 and 5 min, respectively. For tomogram 352 collection, 300-nm sections were used and 10 L of 15-nm colloidal gold (BBI solutions) were 353 applied for 10 min on each side of the grid as fiduciary markers. 14 354 Electron Tomography 355 356 Tomograms were collected using a Tecnai G2 F30 (FEI) electron microscope operating at 357 300 kV. Images were taken at 15,000x from -60 to +60 with 1 interval. Each tomogram was 358 collected in two perpendicular axes. Etomo was used to build the tomograms and to merge the 359 two single-axis tomograms into one dual-axis tomogram. Tomograms were then displaced, 360 analyzed and modeled using the 3DMOD software (40). 361 Fluorescence microscopy 362 363 Anabaena cells were visualized with a Leica DM6000B fluorescence microscope and an 364 ORCA-ER camera (Hamamatsu) using an FITC L5 filter (excitation, band-pass [BP] 480/40 365 filter; emission, BP 527/30 filter) and the Leica SP5 2-photon confocal microscope. The images, 366 including BlindDeblur deconvolution of 3D images, were produced using the LAS AF Leica 367 software. 368 369 Preparation of Cell Extracts and Western blots. 370 Total cell extracts were prepared as described in (41). Proteins were separated using 371 Novex 14% Tris-Glycine gels (Novex, Life Technology). Chlorophyll concentration was used to 372 ensure equivalent loading of cell extracts. 1.2 g chl was loaded per 1-mm well for blotting and 373 Coomassie staining. 374 For immunoblots, proteins were transferred to PVDF membranes (immobilon, Millipore) 375 using a Bio-Rad gel transfer system (Bio-Rad). Blots were blocked with Tris-buffered saline 376 supplemented with 0.1% Tween and 5% dry skimmed milk and incubated with the primary 377 antibody (1:500 dilution for FraG and 1:10,000 dilution for FNR) over-night at 4 °C. After 378 washing, blots were incubated 1 h at room temperature with a 1:15,000 dilution of peroxidase- 379 conjugated anti-rabbit IgG (Promega, Madison, WI). The signal was visualized using ECL 380 chemiluminescent substrate (SuperSignal West Pico Chemiluminescent, Thermo Scientific). 381 Images were generated with a CCD camera and analyzed using ImageJ software. 382 383 15 384 Acknowledgments. This work was supported by the Ellison Medical Foundation and The 385 University of Chicago. We thank Prof. Enrique Flores for support in part from grant no. 386 BFU2011-22762 from Plan Nacional de Investigación, Spain, co-financed by the European 387 Regional Development Fund. We also thank Sean Callahan for plasmid pAN130 and Amel Latifi 388 for plasmid pRL25N. William Buikema and Ghada Ajlani provided critical reading of the paper. 389 390 16 391 392 393 394 395 396 397 398 399 400 401 402 403 404 405 406 407 408 409 410 411 412 413 414 415 416 417 418 419 420 421 422 423 424 425 426 427 428 429 430 431 432 433 434 435 References 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. 11. 12. 13. 14. 15. 16. 17. Haselkorn R (2008) Cell–cell communication in filamentous cyanobacteria. Mol Microbiol 70(4):783–785. Jüttner F (1983) 14C-labeled metabolites in heterocysts and vegetative cells of Anabaena cylindrica filaments and their presumptive function as transport vehicles of organic carbon and nitrogen. J Bacteriol 155(2):628–33. Cumino AC, Marcozzi C, Barreiro R, Salerno GL (2007) Carbon Cycling in Anabaena sp. PCC 7120. Sucrose Synthesis in the Heterocysts and Possible Role in Nitrogen Fixation. Plant Physiol 143(3):1385–1397. López-Igual R, Flores E, Herrero A (2010) Inactivation of a Heterocyst-Specific Invertase Indicates a Principal Role of Sucrose Catabolism in Heterocysts of Anabaena sp. J Bacteriol 192(20):5526–5533. Vargas WA, Nishi CN, Giarrocco LE, Salerno GL (2010) Differential roles of alkaline/neutral invertases in Nostoc sp. PCC 7120: Inv-B isoform is essential for diazotrophic growth. Planta 233(1):153–162. Hejazi M, et al. (2002) Isoaspartyl dipeptidase activity of plant-type asparaginases. Biochem J 364(Pt 1):129–136. Burnat M, Herrero A, Flores E (2014) Compartmentalized cyanophycin metabolism in the diazotrophic filaments of a heterocyst-forming cyanobacterium. Proc Natl Acad Sci:201318564. Flores E, Herrero A (2010) Compartmentalized function through cell differentiation in filamentous cyanobacteria. Nat Rev Microbiol 8(1):39–50. Mariscal V, Herrero A, Flores E (2007) Continuous periplasm in a filamentous, heterocystforming cyanobacterium. Mol Microbiol 65(4):1139–1145. Mariscal V, Flores E (2010) Multicellularity in a heterocyst-forming cyanobacterium: pathways for intercellular communication. Adv Exp Med Biol 675:123–135. Flores E, Herrero A, Wolk CP, Maldener I (2006) Is the periplasm continuous in filamentous multicellular cyanobacteria? Trends Microbiol 14(10):439–443. Wilk L, et al. (2011) Outer membrane continuity and septosome formation between vegetative cells in the filaments of Anabaena sp. PCC 7120. Cell Microbiol 13(11):1744– 1754. Giddings TH, Staehelin LA (1978) Plasma membrane architecture of Anabaena cylindrica: occurrence of microplasmodesmata and changes associated with heterocyst development and the cell cycle. Eur J Cell Biol (16):235–249. Giddings TH, Staehelin LA (1981) Observation of microplasmodesmata in both heterocystforming and non-heterocyst forming filamentous cyanobacteria by freeze-fracture electron microscopy. Arch Microbiol 129(4):295–298. Bauer CC, Buikema WJ, Black K, Haselkorn R (1995) A short-filament mutant of Anabaena sp. strain PCC 7120 that fragments in nitrogen-deficient medium. J Bacteriol 177(6):1520–1526. Lang NJ, Fay P (1971) The Heterocysts of Blue-Green Algae. II. Details of Ultrastructure. Proc R Soc Lond B Biol Sci 178(1051):193–203. Wildon D, Mercer F (1963) The Ultrastructure of the Vegetative Cell of Blue-Green Algae. Aust J Biol Sci 16(3):585–596. 17 436 437 438 439 440 441 442 443 444 445 446 447 448 449 450 451 452 453 454 455 456 457 458 459 460 461 462 463 464 465 466 467 468 469 470 471 472 473 474 475 476 477 478 479 480 18. Lehner J, et al. (2013) Prokaryotic multicellularity: a nanopore array for bacterial cell communication. FASEB J Off Publ Fed Am Soc Exp Biol 27(6):2293–2300. 19. Mariscal V (2014) Cell-Cell joining proteins in heterocyst-forming cyanobacteria. In: Flores, E. and Herrero, A. (eds) The Cell Biology of Cyanobacteria. Caister Academic Press. pp. 293-304. 20. Omairi-Nasser A, Haselkorn R, Austin J (2014) Visualization of channels connecting cells in filamentous nitrogen-fixing cyanobacteria. FASEB J:fj.14–252007. 21. Buikema WJ, Haselkorn R (1991) Isolation and complementation of nitrogen fixation mutants of the cyanobacterium Anabaena sp. strain PCC 7120. J Bacteriol 173(6):1879– 1885. 22. Nayar AS, Yamaura H, Rajagopalan R, Risser DD, Callahan SM (2007) FraG is necessary for filament integrity and heterocyst maturation in the cyanobacterium Anabaena sp. strain PCC 7120. Microbiology 153(2):601–607. 23. Merino-Puerto V, Mariscal V, Mullineaux CW, Herrero A, Flores E (2010) Fra proteins influencing filament integrity, diazotrophy and localization of septal protein SepJ in the heterocyst-forming cyanobacterium Anabaena sp. Mol Microbiol 75(5):1159–1170. 24. Mullineaux CW, et al. (2008) Mechanism of intercellular molecular exchange in heterocystforming cyanobacteria. EMBO J 27(9):1299–1308. 25. Merino-Puerto V, et al. (2011) FraC/FraD-dependent intercellular molecular exchange in the filaments of a heterocyst-forming cyanobacterium, Anabaena sp. Mol Microbiol 82(1):87– 98. 26. Flores E, et al. (2007) Septum-Localized Protein Required for Filament Integrity and Diazotrophy in the Heterocyst-Forming Cyanobacterium Anabaena sp. Strain PCC 7120. J Bacteriol 189(10):3884–3890. 27. Mariscal V, Herrero A, Nenninger A, Mullineaux CW, Flores E (2011) Functional dissection of the three-domain SepJ protein joining the cells in cyanobacterial trichomes. Mol Microbiol 79(4):1077–1088. 28. Nürnberg DJ, et al. (2014) Branching and intercellular communication in the Section V cyanobacterium Mastigocladus laminosus, a complex multicellular prokaryote. Mol Microbiol 91(5):935–949. 29. Mitchell A, et al. (2015) The InterPro protein families database: the classification resource after 15 years. Nucleic Acids Res 43(Database issue):D213–221. 30. Omasits U, Ahrens CH, Müller S, Wollscheid B (2013) Protter: interactive protein feature visualization and integration with experimental proteomic data. Bioinformatics:btt607. 31. Nürnberg DJ, et al. (2015) Intercellular Diffusion of a Fluorescent Sucrose Analog via the Septal Junctions in a Filamentous Cyanobacterium. mBio 6(2):e02109–14. 32. Hermann R, Walther P, Müller M (1996) Immunogold labeling in scanning electron microscopy. Histochem Cell Biol 106(1):31–39. 33. Drew D, et al. (2002) Rapid topology mapping of Escherichia coli inner-membrane proteins by prediction and PhoA/GFP fusion analysis. Proc Natl Acad Sci 99(5):2690–2695. 34. Ramos-León F, Mariscal V, Frías JE, Flores E, Herrero A (2015) Divisome-dependent subcellular localization of cell-cell joining protein SepJ in the filamentous cyanobacterium Anabaena. Mol Microbiol:n/a–n/a. 35. Ughy B, Ajlani G (2004) Phycobilisome rod mutants in Synechocystis sp. strain PCC6803. Microbiology 150(Pt 12):4147–56. 18 481 482 483 484 485 486 487 488 489 490 491 492 493 494 495 496 497 498 36. Elhai J, Vepritskiy A, Muro-Pastor AM, Flores E, Wolk CP (1997) Reduction of conjugal transfer efficiency by three restriction activities of Anabaena sp. strain PCC 7120. J Bacteriol 179(6):1998–2005. 37. Zhang L-C, Risoul V, Latifi A, Christie JM, Zhang C-C (2013) Exploring the size limit of protein diffusion through the periplasm in cyanobacterium Anabaena sp. PCC 7120 using the 13 kDa iLOV fluorescent protein. Res Microbiol 164(7):710–717. 38. Austin JR 2nd (2014) High-Pressure Freezing and Freeze Substitution of Arabidopsis for Electron Microscopy. Arabidopsis Protocols, Methods in Molecular Biology., eds SanchezSerrano JJ, Salinas J (Humana Press), pp 473–486. 39. Otegui MS, Mastronarde DN, Kang B-H, Bednarek SY, Staehelin LA (2001) ThreeDimensional Analysis of Syncytial-Type Cell Plates during Endosperm Cellularization Visualized by High Resolution Electron Tomography. Plant Cell 13(9):2033–2052. 40. Kremer JR, Mastronarde DN, McIntosh JR (1996) Computer visualization of threedimensional image data using IMOD. J Struct Biol 116(1):71–76. 41. Omairi-Nasser A, de Gracia AG, Ajlani G (2011) A larger transcript is required for the synthesis of the smaller isoform of ferredoxin:NADP oxidoreductase. Mol Microbiol 81(5):1178–1189. 19 499 Figures 500 501 Figure 1. The septum between two vegetative cells of WT Anabaena and three 502 fragmentation mutants. A) Electron tomographic image of the WT septum. B) Septum 503 shown in “A” is rotated 90° around the y axis showing the channel distribution within the 504 septum. Several channels are observed in the middle of the septum (white holes). C) 505 Electron tomographic image of the septum of CSVT1 mutant (∆fraC). The septum contains 506 fewer channels compared to WT. D) Septum shown in C is rotated 90° around the y axis. E) 507 Electron tomographic image of the septum of CSVT2 mutant (∆fraD). The septum contains 508 similar number of channels compared to WT. F) Septum shown in E is rotated 90° around 509 the y axis. G) Electron tomographic image of the septum of CSVT22 mutant (∆fraC/D). H) 510 Septum shown in G is rotated 90° around the y axis showing the channel distribution. 511 Arrowheads indicate the channels observed on their corresponding panel before rotation. 512 “t” indicates thylakoids. 513 tomographic slices. Scale bar 50 nm All images are composed of 10 superimposed 2.2 nm serial 514 515 Figure 2. Heterocyst-vegetative cell septa in WT and fragmentation mutants. A) Electron 516 tomographic image of a WT heterocyst junction. White arrowheads point to the edges of the 517 septum. Yellow arrowheads show the channels that connect the heterocyst and the 518 vegetative cell. B) Electron tomographic image of the CSVT22 (fraCD) heterocyst junction. 519 The septum in this mutant is thicker and only 1-2 channels are present compared to WT. 520 The yellow arrow points to the only channel observed in this tomogram. Black arrowheads 521 point to the plasma membrane in vegetative cells in each panel. 522 All tomographic images are composed of 10 superimposed 2.2-nm tomographic slices. Het: 523 heterocyst. Veg: vegetative cell. Scale bar: 200 nm. 524 525 Figure 3. The septum between two vegetative cells of WT Anabaena and of fragmentation 526 mutants. A) Electron tomographic image of the WT septum. B) Septum shown in “A” is 527 rotated 90° around the y axis showing the channel distribution within the septum. Several 528 channels are observed in the middle of the septum (white holes). C) Electron tomographic 20 529 image for the septum of mutant CSVM34 (∆fraG). D) Septum shown in C is rotated 90° 530 around the y axis showing the channel distribution within the septum; only 2 channels are 531 observed compared to 15 to 20 in WT. Arrowheads in B and D indicate channels observed 532 in the septum in panels A and C, respectively. “t” indicates thylakoids. All images are 533 composed of 10 superimposed 2.2 nm serial tomographic slices. Scale bar 50 nm 534 535 Figure 4. Subcellular localization of FraG in Anabaena heterocysts. A) Immunogold 536 labelling of WT Anabaena using antibodies (black dots) raised against the N-terminal 537 coiled-coil domain of FraG. B) Control immunogold labeling of WT using only secondary 538 antibody; no dots. C) Light transmission micrograph of WT Anabaena grown under N- 539 conditions. D) Autofluorescence of the same cells shown in C. Heterocysts do not show 540 autofluorescence due to loss of PS II chlorophyll. E) Light transmission micrograph of the 541 CSAM137 mutant (FraG-GFP) grown under N- conditions. F) Autofluorescence (red) and 542 GFP fluorescence (green) of the same cells shown in E. GFP fluorescence locates FraG at the 543 poles of the heterocysts. C: Cyanophycin. Het: heterocyst. Veg: vegetative cell. Scale bar: 200 544 nm. 545 546 Figure 5. Serial immunoelectron tomography of WT Anabaena grown under N- conditions. 547 (A) to (C) Serial 2.2-nm tomographic slice images (every 50th slice) through a heterocyst 548 neck labeled with anti-FraG antibody. Note that as one proceeds from the top of the section 549 (A) to the bottom (C), different groups of 10-nm gold labels, indicated by arrows and 550 numbers, are seen at different depths within the cyanophycin interior; 1 and 2 at the top 551 (A), 3 in the middle (B) and 4 at the bottom (C) of the cyanophycin plug. D) Tomographic 552 model of the immunoelectron tomogram showing the location of FraG around the 553 cyanophycin (blue). Green arrows indicate gold particles in the cyanophycin at different 554 depth with n° 1 and 2 seen in section 10 (A), n° 3 in section 60 (B) and n° 4 in section 110 555 (C). Note that there is no contact between the gold particle and the channels (or 556 peptidoglycan). Peptidoglycan layer is red. Het: heterocyst. Veg: vegetative cell. Scale bar: 557 100 nm. 558 21 559 Figure 6. FraG localization between vegetative cells. (A) Immunogold labeling of W30, 560 overexpressing FraG, using anti-FraG antibody. (B) Immunogold labeling of CSVM34 561 (fraG) shows no gold particles. (C) Zoom in to the septum in A; eleven gold particles seen 562 on both sides of the septum. Scale bar: 200 nm. 22 563 Figure 7. FraG N-terminal localization. (A) Cartoon showing the different domains of FraG 564 and the location of the GFP insertion in the linker domain of FraG in the pRGF plasmid. The 565 plasmid was introduced into both WT and ∆fraG yielding WGF and ∆GF. (B) and (C) 566 Autofluorescence (red) and GFP fluorescence (green) in WGF grown in N+ and N- 567 respectively. (D) Autofluorescence (red) and GFP fluorescence (green) in W30 grown in N+ 568 (Note that the mutant cannot grow in N- media). (E) Same micrograph shown in D rotated 569 45 around the y axis and showing only GFP fluorescence. Notched arrowheads indicate the 570 location of the CCL-GFP construct in the divisome plane. Straight arrowheads indicate cells 571 at the end of division, hence the presence of a GFP signal. GFP fluorescence shows the FraG 572 N-terminal-linker domain as rings in the divisome plane of vegetative cells. CC; predicted 573 Coiled Coil. L; predicted Linker domain. TM; Trans-membrane domain. N-ter; N-terminal 574 domain. C ter; C terminal domain. M1; FraG 1st Methionine. P391; Proline- the 391st amino 575 acid in FraG linker domain where GFP was fused. Scale bar: 2 m. 576 577 Figure 8. Model for the heterocyst-vegetative cell junction showing putative localization 578 and interactions of FraC, FraD and FraG. These proteins appear to be located in the plasma 579 membrane and/or septum and involved in channel formation, either directly or by 580 recruiting other factors. In a fully-developed heterocyst, FraG is found in the heterocyst 581 neck around the cyanophycin, implicating FraG in an additional role to channel formation, 582 possibly assembly or maintenance of heterocyst neck formation. CC; predicted Coiled Coil. 583 L; predicted Linker domain. MD; Trans-membrane domain. PM; Plasma membrane. PG; 584 Peptidoglycan. OM; Outer membrane. C; Cyanophycin. 585 23