Handout for six graders
advertisement

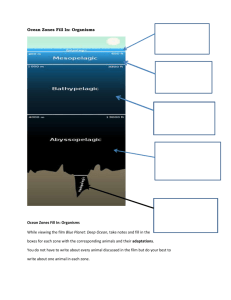
Handout for six graders Name: Date: Period: Ocean Acidification and Acid Rain Activity 1: Acidity and pH 1) List the following liquids from more acidic to more basic. a) Pure water, b) laundry detergent, c) milk, d) orange juice, e) vinegar f) baking soda solution, g) bleach Neutral More Basic Pure water More Acidic Liquid Name Pure water 2) Using the universal pH indicator determine the pH. For each liquid do the following: Add 5 drops of liquid and 1 drop of universal pH indicator to a well in the microplate. Compare the colors with the chart and determine the pH. color pH 7 3) List again the liquids. How close were you the first time? 0 Neutral 7 Pure water More Acidic More Basic 14 Activity 2: Simulating ocean acidification, acid rain and infrastructure damage Background: Ocean Acidification and Acid Rain: Humans are emitting carbon dioxide, the most important green house gases, at an alarming rate via the burning of fossil fuels, such as coal, oil and gasoline. Extra carbon dioxide in the atmosphere acts like a blanket that traps heat and warms the planet, causing global warming. Additionally, extra carbon dioxide in the atmosphere makes rain more acidic which deteriorates buildings and is harmful to plants. Extra carbon dioxide in the ocean makes the ocean more acidic, which causes calcium carbonate to dissolve, an essential substance for various phytoplankton, coral and other animals in the ocean to survive. Question: What happens to the pH of water when we mix in carbon dioxide? Hypothesis: ______________________________________________________________________________ ______________________________________________________________________________ ______________________________________________________________________________ Question What happens to calcium carbonate when put in acid? Hypothesis: ______________________________________________________________________________ ______________________________________________________________________________ ______________________________________________________________________________ Materials: - 5 250ml beakers -Dry Ice -Vinegar -Universal pH Indicator -Solutions: water; water plus buffer (alkalinity) -Plastic pippet with built in dropper. -Chalk -Stir rod Procedure: 1. Label the beakers as follow: A, B, C, Acid, No Acid. 2. There will be two containers at the front labeled Solution 1 and Solution 2. Solution 1 has a low alkalinity (no buffer) and solution 2 has a high alkalinity (buffer). Add 150 mL of solution 1 to beakers A, and B and 150 mL of solution 2 to beaker C. Only one student at the time per group should be collecting the solutions. Make sure to carry only two beakers at the time. 3. Put beakers Acid and No Acid aside we will use them later. 4. Add 3 drops of universal pH indicator to beaker A, B, C and stir with an stirring rod 5. Record the color of the solution in Table 1 and estimated the pH form the chart. Record pH in Table 1 as well. Table 1. Initial pH values of the three beakers Beaker A(control) B C Color pH 6. When ready rise your hand and one of the teachers will bring dry ice (frozen CO2) for the rest of the experiment. 7. The teacher will add the same amount of dry ice to beakers B and C. Make observations of what you see. Fill out Table 2. Table 2. Qualitative Observations of beakers when dry ice is inserted and final pH Beaker A Beaker B (no buffer) Beaker C (buffer) (control, no dry ice) Color after dry ice is gone pH after dry ice is gone 8. Put aside beaker A, B and C you will work now with beakers Acid and No Acid. 9. Add 80 mL of tap water and 20 mL of vinegar to the beaker labeled Acid and stir for 5 seconds. 10. Add 100 mL of tap water to beaker labeled No Acid. 11. Add a piece of chalk to beakers Acid and No Acid 12. Record your observations in Table 3. Acid Table 3: Qualitative Observations of beakers after calcium carbonate and dry ice are inserted. No Acid Discussion: Explain your observations from Table 2: ______________________________________________________________________________ ______________________________________________________________________________ _____________________________________________________________________________ Explain your observations from Table 3: ______________________________________________________________________________ ______________________________________________________________________________ ______________________________________________________________________________ Questions: 1) Why is carbon dioxide increasing in the atmosphere and ocean? ______________________________________________________________________________ ______________________________________________________________________________ ______________________________________________________________________________ ______________________________________________________________________________ 2) What happened to the pH of the water when we added CO2 (dry ice)? ______________________________________________________________________________ ______________________________________________________________________________ ______________________________________________________________________________ ______________________________________________________________________________ 3) How does extra carbon dioxide in the ocean affect the ocean? ______________________________________________________________________________ _____________________________________________________________________________ ______________________________________________________________________________ ______________________________________________________________________________ 4) What are the possible effects of acid rain on buildings or statues? ______________________________________________________________________________ ______________________________________________________________________________ ______________________________________________________________________________ ______________________________________________________________________________ 5) How can we reverse the process of ocean acidification? ______________________________________________________________________________ ______________________________________________________________________________ ______________________________________________________________________________ ______________________________________________________________________________