the adenovirus and its application in virotherapy
advertisement

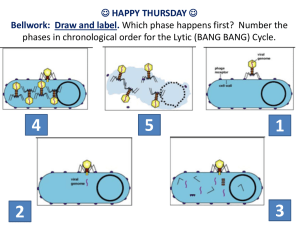
Conference Session (A1) Paper 6010 THE ADENOVIRUS AND ITS APPLICATION IN VIROTHERAPY Pegah Zarandi (ppz2@pitt.edu, 0012; Lora 6:00), Dominic Dawes (ddd28@pitt.edu, 0012; Bursic 2:00) Abstract- As it currently stands, cancers such as leukemia and pancreatic cancer have very low survival rates 50% and 1.1% respectively [1][2]. Conventional treatments such as radiotherapy and chemotherapy, indiscriminately destroy both normal and cancerous cells and can take a severe toll on the human body. Oncolytic virotherapy is a more progressive alternative that utilizes modified viruses that can discern between the tumors and normally functional tissues, while selectively terminating the mutated cells. [3] The notion that viruses could be repurposed for therapeutic uses is not a new concept. The effect of viral infection on tumor cells was first observed during the 1950’s and 1960’s when several cases of cancer remission were reported after the patient was exposed to a virus [4]. Since its early stages of research, the field of virotherapy has made significant quantitative progress. In some countries, viral therapies such as Onyx-15 and Rigvir have become approved as viable medical treatment options for cancer patients [3] [4]. However, there are still several factors, such as delivery and transduction efficiency of target cells, that need to be improved in order for virotherapy to become a mainstream cancer treatment [5]. This paper will focus on the genetic modification of the adenovirus H101 for use in cancer virotherapy. The paper’s first objective will be to provide an overview of the process of virotherapy and the science behind the modification the H101 virus, and how it takes advantage of its specific cellular tropism, in order to infect specific types of cancer cells. Subsequently, we will give an overview of the biotransport and cellular biological phenomena that are involved in the delivery of the virus, and then assess the current issues that affect the therapy's efficacy such as, virus delivery and transduction efficiency of target cells. Furthermore, we will analyze various perspectives from sources such as, scientific journals and trusted articles on the viability and implementation of the H101 adenovirus in oncolytic virotherapy in order to demonstrate that oncolytic virotherapy is an applicable biotechnology. Since the ethical and regulatory guidelines of oncolytic virotherapy can be ambiguous, biomedical ethical handbooks will be examined and evaluated in order to concretely delineate how oncolytic virotherapy should be administered as a medical treatment. University of Pittsburgh Swanson School of Engineering 1 1-26-16 ANNOTATED BIBLIOGRAPHY [6] BMES. (2004). “Biomedical Engineering Society Code of Ethics.” BMES. (Code of Ethics). http://bmes.org/files/2004%20Approved%20%20Code%20o f%20Ethics(2).pdf The Biomedical Engineering Society created a strict list of obligations and ethical conducts that must be upheld by all biomedical engineers. It is made up of two biomedical engineering professional obligations, two biomedical engineering health care obligations, two biomedical engineering research obligations, and two biomedical engineering training obligations. We will include this information within our ethics of virotherapy section in the paper. [1] Chen, C. Y., J. S. Senac, E. A. Weaver, S. M. May, D. F. Jelinek, P. Greipp, T. Witzig, and M. A. Barry. "Species D Adenoviruses as Oncolytics against B-cell Cancers." Clinical Cancer Research 17.21 (2011): 6712-722. Web. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3207036/. This article from the National Center for Biotechnology Information is of an experiment where 15 adenoviruses selected to represent Ad species B, C, D, E, and F were tested in vitro against cell lines and primary patient B cell cancers for their ability to infect, replicate in, and kill these cells. The results showed that species D adenoviruses have a unique ability to infect and replicate in B cell cancers as compared to other adenovirus species. We will use this article for statistics of survival rates of cancer and how species D adenovirus successfully infiltrated the cancer cells. [5] Eager, R. M., and J. Nemunaitis. "Clinical Development Directions in Oncolytic Viral Therapy." Cancer Gene Therapy Cancer Gene Therapy 18.5 (2011): 305-17. Web. http://www.nature.com/cgt/journal/v18/n5/full/cgt20117a.ht ml. This article from the Cancer Gene Therapy provides a brief description of novel anticancer biologics. To date, oncolytic virotherapy has shown to be safe, and has generated clinical responses in tumors that are resistant to chemotherapy or radiotherapy. In addition, the researchers Pegah Zarandi Dominic Dawes wanted to maximize the efficacy of the viral therapeutics, and to establish stable systemic delivery mechanisms. We will use this information as evidence as to why virotherapy is a safer and more practical option for cancer treatment. [3] Garber, Ken. "China Approves World's First Oncolytic Virus Therapy for Cancer Treatment." Http://jnci.oxfordjournals.org/. Oxford University Press, 2006. Web. 19 Jan. 2016. http://jnci.oxfordjournals.org/content/98/5/298.full. This article from the Journal of the National Cancer Institute discusses how Chinese regulators approved the world's first oncolytic viral therapy for cancer, Shanghai Sunway Biotech's genetically modified adenovirus H101. They expect to begin marketing the H101 virus for treating head and neck cancer. This information will be used in our paper to further explain how the ONYX-H101 virus is currently being used in other countries as the main treatment for cancer and should also be implemented in the United States. [4] Kelly, Elizabeth, and Stephen J. Russell. "History of Oncolytic Viruses: Genesis to Genetic Engineering." Nature.com. Nature Publishing Group, n.d. Web. 13 Jan. 2016. http://www.nature.com/mt/journal/v15/n4/abs/6300108a.htm l. This article from the Nature Publishing Group and it offers specific, qualitative history about oncolytic viruses. The authors aim to provide an in-depth description of the methods for virus propagation in the 1950-60s, and how the advancement in technology has allowed for a significant progression in the science. We will use this journal in a section of our paper that explains how technology has allowed for virotherapy to grow and become safe. [7] Latvian State Agency of Medicines Registry http://www.zva.gov.lv/zaluregistrs/?iss=1&lang=en&q=Rigvir&ON=&SN=&NAC=on &RN=&ESC=on&AK=&SAT=on&RA=&DEC=on&LB=& PIM=on| accessed 21 January 2015 This is the Medicinal Product Register of the Republic of Latvia, where the ONYX-015 H101 virus has become approved as a viable medical treatment option for patients’ with cancer. The ONYX-015 H101 is an experimental oncolytic virus created by genetically engineering the adenovirus that is implemented in Latvia, and other countries, to use in replacement of chemotherapy and University of Pittsburgh Swanson School of Engineering 2 1-26-16 radiotherapy to treat cancer. This information will be used in our paper to discuss how other countries are using alternative methods to curing cancer. [2] "Pancreatic Cancer Survival Statistics." Pancreatic Cancer Survival Statistics. Cancer Research UK, 15 May 2015. Web. 11 Jan. 2016. http://www.cancerresearchuk.org/healthprofessional/cancer-statistics/statistics-by-cancertype/pancreatic-cancer/survival#heading-Zero. Scientists from the Cancer Research UK in England recently discovered the statistical survival rate for pancreatic cancer, which arises when cells in the pancreas begin to multiply and form a mass. These statistics show how the current cancer treatments currently used are not sufficient enough to kill the cancer cells. We will include this information within our abstract as evidence that virotherapy is a more practical and effective treatment. [8] Russell, Stephen J., Kah Whye Peng, and John C. Bell. "ONCOLYTIC VIROTHERAPY." N.p., n.d. Web. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3888062/ This article from the National Institutes of Health discusses the challenges scientists face to select the best oncolytic virus platforms and engineered derivatives, to transiently suppress the immune system to maximize both virus spread and anticancer immunity. This information will be incorporated in our background section of oncolytic virotherapy.