states of matter – thermal energy
advertisement

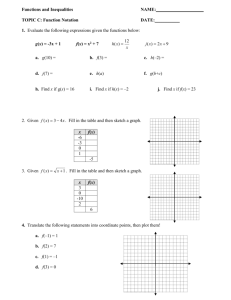
STATES OF MATTER – THERMAL ENERGY PURPOSE Matter consists of atoms and molecules and all substances consist of matter. Changing the state of a substance requires energy. Adding or removing thermal energy from a substance causes a change of state. Energy affects the attraction between the atom or molecules and their rate of movement. A substance’s temperature determines whether it occurs in a solid, liquid or gas state. A substance changes from a solid to a liquid at its melting point, from a liquid to a gas at its boiling point, and from a liquid to a solid at its freezing point. A different amount of energy is needed to change the state of different substances. For example, it takes more energy to melt a solid metal into a liquid than to melt an ice cube into water. In this virtual lab you will examine how energy affects the state, the atomic or molecular activity, and the temperature of a substance. QUESTION How does thermal energy affect the state of a substance? PROCEDURE 1. Go to the website below. http://www.glencoe.com/sites/common_assets/science/virtual_labs/E17/E17.html 2. Click the Video button. Then click Play on the video controller. Watch the video about melting and boiling points of substances. In the space below, write a summary of the video: 3. When you select the Water, Mercury, or Iron buttons, the selected substance appear in its solid form inside the Energy box. The Energy box is able to add thermal energy to the substance or remove it from the substance at a constant rate. Inside the Energy box, a sensor senses the temperature of the substance. Water Predict what will happen to the state of the substance, the atom activity and the shape of the graph when thermal energy is ADDED Click the Heat button on the Energy box. Observe what happens to the state of the substance, the atom activity, and the temperature of the substance. Sketch or describe your results Click the Graph button to see a printout of a temperature versus time graph that shows what happened when heat was added to the substance. Sketch the graph Click the Heat button on the Energy box. Observe what happens to the state of the substance, the atom activity, and the temperature of the substance. Sketch or describe your results Mercury Iron Click the Graph button to see a printout of a temperature versus time graph that shows what happened when heat was added to the substance. Sketch the graph Predict what will happen to the state of the substance, the atom activity and the shape of the graph when thermal energy is REMOVED. Click the Cool button on the Energy box. Observe what happens to the state of the substance, the atom activity, and the temperature of the substance. Sketch or describe your results Click the Graph button to see a printout of a temperature versus time graph that shows what happened when heat was added to the substance. Sketch the graph Click the Cool button on the Energy box. Observe what happens to the state of the substance, the atom activity, and the temperature of the substance. Sketch or describe your results Click the Graph button to see a printout of a temperature versus time graph that shows what happened when heat was added to the substance. Sketch the graph Other Observations/Questions/Comments Questions 1. After observing heat being added to each substance, were your predictions correct or incorrect? Explain. 2. After observing heat being removed from each substance, were your predictions correct or incorrect? Explain 3. What do the horizontal portions of the graph represent? When heat is added to or removed from the substance and the temperature remains the same, when is the thermal energy going? Why? 4. Determine the approximate melting, boiling and freezing points of the substance by reading the graph. Record this data in the table. Substance Melting Point (C) Boiling Point (C) Freezing Point (C) Iron Mercury Water 5. Why do you think it would be impossible to contain gaseous iron inside of a container?