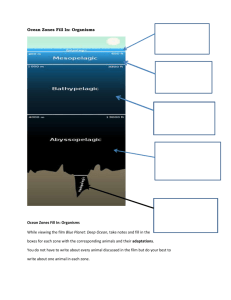
Water properties
advertisement

Water Studies We will be examining these important factors: turbidity, temperature, salinity, pH and compounds, all of which are influential in determining when and where life will be found in the ocean. Turbidity: the measure of water clarity and transparency. Water is considered turbid when its suspended matter is visible. All natural waters have some degree of suspended matter. Turbidity is affected by size, shape and amount of suspended particles in the water. Examples of suspended particles which contribute to turbidity include fish scales, animal oils, oxygen bubbles and fecal waste. Factors that can affect turbidity are high flowing waters, runoff, erosion, waste water and septic pollutants and algal blooms. Turbid waters can have a negative effect on the environment. Light is unable to penetrate to deeper waters and can limit photosynthesis in plants. In addition, waters will become warmer when the suspended particles in the water column absorb sunlight. Higher temperatures create less dissolved oxygen in the water. A turbidimeter is used to measure turbidity. As light passes through the salt water, the light is scattered by the particles hitting it. The intensity of scattered light is measured in Nephelometric Turbidity Units (NTU). The higher the reading, the more turbidity. Temperature: the temperature range in the ocean is about -35.6⁰F to 86⁰F with the average temperature being about 38.3⁰F. The sun can only warm the surface waters, hence a colder average temps due to lack of sunlight in most of the ocean. Most marine organisms are coldblooded which means their internal body temperature is essentially the same as the surrounding waters. Marine mammals are warm-blooded, which means they have a constant internal body temperature independent of their surroundings. Extreme heating or cooling may cause death to marine organisms. Therefore, many animals seek out waters with optimal ranges of temperature. Water temperature can be measured with a thermometer. Temperatures at great depths are measured with a bathythermograph. Water temperatures are affected by available sunlight, atmospheric conditions (wind currents), turbidity, confluence and deforestation. Photosynthesis rates will change with an increase of temperatures. Enzymes involved in the chemical reactions of photosynthesis are temperature sensitive and are destroyed (denatured) at higher temperatures. Thermal pollution is a sudden increase or decrease in temperature of a natural body of water which may be ocean, lake, river or pond by human influence. This normally occurs when a plant or facility (an industry or other human-made organization) takes in water from a natural source and either cools it down or heats it up. They then eject that water back into the natural resource, which changes the dissolved oxygen levels and can have disastrous effects on local ecosystems and communities. Salinity: the characteristic that distinguished the ocean from all other bodies of water. Salinity is a measure of the quantity of dissolved salts in the water. The average salinity for the ocean as a whole is 34.7 parts salt per 1000 parts water(‰), The refractometer allows for quick measurement of salinity using only a few drops of seawater. pH: the measure of acids (H⁺) and bases (OH⁻) in a solution. The pH scale runs from 0-14. A pH of 7 is neutral, equal number of H⁺ and OH⁻ ions. pH less than 7 is acidic (more H⁺ ions) and more than 7 is basic(more OH – ions). The pH of ocean water is slightly basic, usually between 8 and 8.4 at the surface and 7.4 to 7.9 in deeper water. Most lakes and streams have a pH between 6 and 8, although some lakes are naturally acidic even without the effects of acid rain. Acid rain primarily affects sensitive bodies of water, which are located in watersheds whose soils have a limited ability to neutralize acidic compounds (called “buffering capacity”). Lakes and streams become acidic (i.e., the pH value goes down) when the water itself and its surrounding soil cannot buffer the acid rain enough to neutralize it. In areas where buffering capacity is low, acid rain releases aluminum from soils into lakes and streams; aluminum is highly toxic to many species of aquatic organisms. The pH of natural, unpolluted rainwater actually has a pH of about 5.6 (acidic). The acidity of rainwater comes from the natural presence of three substances (CO2, NO, and SO2) found in the troposphere (the lowest layer of the atmosphere). Marine life cannot survive if the pH is very far outside of these ranges. At pH 5, most fish eggs cannot hatch. At lower pH levels, some adult fish die. Some acid lakes have no fish. The ocean has its own buffer system that stabilizes pH fluctuation. Man can alter the pH by introducing untreated, toxic chemical waste into the sea, industrial wastes and detergents which can create pockets of deadly water. Compounds: there are many compounds in seawater which are important to the ecology of the ocean. In the ocean, compounds form a very dilute soup of non-living “building blocks”. Compounds are measured in parts per million (ppm). One part per million means 1 g of a substance in 1,000,000 g seawater. Nitrates (NO3) are fertilizers of the sea and are vital for plant growth. They are taken up from the water by plant plankton(phytoplankton) in the process of photosynthesis and are used to create plant tissues. Phytoplankton is then eaten by animals. When plants and animals die, their tissues are broken down by bacteria and the compound is released back into the water as ammonia (NH3) and nitrates (NO3). Certain levels of these compounds are necessary for life in the sea to exist.