Dissolved Gases Article
advertisement

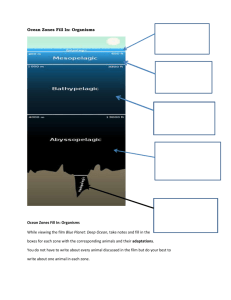
Ocean Dissolved Gases Seawater has many different gases dissolved in it, especially nitrogen, oxygen and carbon dioxide. It exchanges these gases with the atmosphere to keep a balance between the ocean and the atmosphere. This exchange is helped by the mixing of the surface by wind and waves. Temperature and Pressure also play a role in the amount of gases found in different parts of the ocean. Gases and life Dissolved oxygen and carbon dioxide are vital for marine life. Marine plants use dissolved carbon dioxide, sunlight and water to make carbohydrates through the process of photosynthesis. This process releases oxygen into the water. All marine organisms use oxygen for respiration, which releases energy from carbohydrates and has carbon dioxide and water as byproducts. Marine animals with gills, such as fish, use these organs to extract oxygen from the seawater. Variation in dissolved gases Some of the properties of seawater affect how much gas can be dissolved in it: Cold water holds more gas than warm water. You will have seen this with bottles of sodas, which are basically carbon dioxide in water. Warm sodas cannot hold its gas, so as soon as you open a bottle of it, the carbon dioxide leaves the water in a big spray of bubbles. It is less messy to open a cold bottle of soda. Seawater with low salinity holds more gas than high salinity water. Deep water, which has a high pressure, holds more gas than shallow water. The use and creation of dissolved gases by living things can over-ride the effect of these properties. For example, warm water with lots of plankton in it can hold more carbon dioxide than cold water with few living things in it. Carbon dioxide Carbon dioxide is one of the most important gases that dissolve in the ocean. Some of it stays as dissolved gas, but most reacts with the water to form carbonic acid or reacts with carbonates already in the water to form bicarbonates. This removes dissolved carbon dioxide from the water. Many plants and animals use the bicarbonate to form calcium carbonate shells. When these organisms die, some of the bicarbonate is returned to the water, but a lot of it settles down to the sea bed. This process locks up, for long periods of time, carbon that originated in carbon dioxide in the atmosphere. If the ocean and atmosphere stayed the same, there would be a balance between the concentrations of carbon dioxide in each, but carbon dioxide levels in the atmosphere are rising, so more of the gas is dissolving in the ocean. Published 22 June 2010 Photosynthesis and Chemosysnthesis Photosynthesis and chemosynthesis are both processes by which organisms produce food; photosynthesis is powered by sunlight while chemosynthesis runs on chemical energy. Ecosystems depend upon the ability of some organisms to convert inorganic compounds into food that other organisms can then exploit (or eat!). In most cases, primary food production occurs in a process called photosynthesis, which is powered by sunlight. In a few environments, primary production happens though a process called chemosynthesis, which runs on chemical energy. Together, photosynthesis and chemosynthesis fuel all life on Earth. Close up of a tubeworm “bush,” which mines for sulfide in the carbonate substrate with their roots. The sulfide is metabolized by bacteria living in the tubeworms and the chemosynthetic energy produced sustains both organisms. It is a classic symbiotic relationship. Lophelia II 2010 Expedition, NOAA-OER/BOEMRE Photosynthesis occurs in plants and some bacteria, wherever there is sufficient sunlight – on land, in shallow water, even inside and below clear ice. All photosynthetic organisms use solar energy to turn carbon dioxide and water into sugar and oxygen. There is only one photosynthetic formula: CO2 + 6H2O -> C6H12O6 + 6O2. Chemosynthesis is the use of energy released by inorganic chemical reactions to produce food. Chemosynthesis is at the heart of deep-sea communities, sustaining life in absolute darkness, where sunlight does not penetrate. All chemosynthetic organisms use the energy released by chemical reactions to make a sugar, but different species use different pathways. For example, the most extensive ecosystem based on chemosynthesis lives around undersea hot springs. At these hydrothermal vents, vent bacteria oxidize hydrogen sulfide, add carbon dioxide and oxygen, and produce sugar, sulfur, and water: CO2 + 4H2S + O2 -> CH20 + 4S + 3H2O. Other bacteria make organic matter by reducing sulfide or oxidizing methane. Chemosynthetic bacterial communities have been found in hot springs on land and on the seafloor around hydrothermal vents, cold seeps, whale carcasses, and sunken ships. Did You Know? Our knowledge of chemosynthetic communities is relatively new, brought to light by ocean exploration. The thriving communities associated with hydrothermal vents shocked the scientific world when humans first observed a vent on the deep ocean floor in 1977. The discovery of hydrothermal vents and cold-water methane seeps gave us a new vision of primary production in the deep sea. The irony is that once scientists knew what to look for, they went to other wellknown ecosystems that were rich in hydrogen sulfides, such as salt marshes, and found the same mutualistic association of chemosynthetic bacteria and animals that had stunned them in the deep vents. No one had ever thought to look for them, but these communities were there all along. http://oceanexplorer.noaa.gov/facts/photochemo.html 1. What are three gases that become dissolved in the ocean waters? 2. What factors help mix the gases into the ocean waters – name at least two: 3. What life form relies on carbon dioxide and what do these life forms make using the carbon dioxide? 4. What gas must be present for all life forms to do respiration, thus getting energy for life activities? 5. What three properties impact the quantity of a dissolved gas mixing into the water? 6. Once carbon dioxide combines with water to make bicarbonate – what do the ocean life forms use the bicarbonate for? 7. What is the difference between Photosynthesis and Chemosynthesis?