File
advertisement

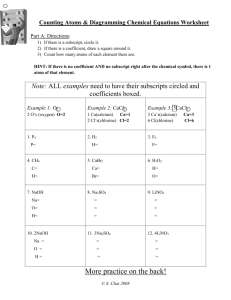
Science - SNC 2P Unit 2: Chemistry Activity 2: Types of Chemical Reactions Overview You will investigate chemical reactions encountered in everyday life and their practical applications. You will describe, using your observations, the reactants and products of a variety of chemical reactions, including synthesis, decomposition, and displacement reactions. You will apply WHMIS in the safe handling and storage of chemicals. You will continue to develop laboratory skills and will be required to report on a lab, including the chemical equations expressing what occurred. Expectations Overall Expectations: CHV.02P investigate chemical reactions encountered in everyday life and their practical applications. Specific Expectations: CH1.03P describe, using their observations, the reactants and products of a variety of chemical reactions, including synthesis, decomposition, and displacement reactions (e.g., the burning of magnesium, the production of oxygen from hydrogen peroxide, the reaction of iron in copper sulphate); CH2.01P through investigations and applications of basic concepts select and use appropriate apparatus, and apply WHMIS safety procedures for the handling, storage, disposal, and recycling of laboratory materials (e.g., wear safety goggles and aprons; use proper techniques to handle, dispose of, and recycle acids, bases, and heavy metal ions; describe procedures to be followed in an emergency); CH2.08P conduct experiments to determine the factors that affect the rate of a chemical reaction (e.g., temperature, surface area of a solid, concentration of a solution); CH2.09P represent simple chemical reactions using word equations, balanced chemical equations, and, where appropriate, molecular models. Content We encounter thousands of chemical reactions every day. When we get up in the morning and have breakfast, we are triggering the process of digestion. As we get into cars or onto buses, we need still more chemical reactions to move us around. Our clothes are the result of chemical reactions, our water bottles, pens, paper, binders, perfume and just about every thing else. Some reactions are beneficial, some are harmful, and some are determined by the way in which we use them. People who work with chemicals need to have a good idea of what is going to happen when chemicals are mixed. If they didn’t, there would be many more accidents and explosions in labs. After centuries of experience, scientists generally have a rough idea of what to expect, based on how a substance reacts with other substances. There are several standard types of reactions: Synthesis Reactions occur when elements are joined to make compounds. These compounds can be simple or complex. The starting substances are called reactants (because they react) and the end results are called products (because they are produced). Raw materials are used to synthesize a product. Example Hydrogen + oxygen = water (word equation) 2H + O (balanced chemical equation) H2O Decomposition Reactions are the opposite of Synthesis Reactions. They occur when a substance is broken down, or decomposed. Example Lithium oxide = lithium and oxygen Li2O 2Li + O (word equation) (balanced chemical equation) Combustion Reactions occur when a substance is burned in oxygen. This causes the substance to combine with oxygen. When metals are burned, they produce a metal oxide. When non-metals are burned, they produce a dioxide. Example Sodium + oxygen = sodium oxide Na + O NaO (word equation) (balanced chemical equation) Example Carbon + oxygen = carbon dioxide (word equation) C + 2O (balanced chemical equation) CO2 Other types of reactions involve the displacement of one molecule or element with another. Single Displacement reactions occur when one substance is displaced by another. Example fluorine + calcium bromide = calcium fluoride and bromine 2Fl + CaBr2 CaFl2 + 2Br (word equation) (balanced chemical equation) The fluorine has displaced the bromine in the above reaction. A Double Displacement Reaction occurs when the molecules of two substances switch places. Example Barium sulphide + potassium iodide = barium iodide + potassium sulphide (word equation) BaS + 2KI BaI2 + K2S (balanced chemical equation) Did you know? Have you ever wondered why substances like carbon dioxide and carbon monoxide are so dangerous? They bond more easily with the iron in our red blood cells, and so will “displace” oxygen in that reaction. So rather than our blood carrying oxygen to the cells, they are carrying carbon monoxide. Our cells cannot use carbon monoxide, and need oxygen to survive. This can result in brain damage and even death. Check Your Understanding 1. What would balance the following equation? Na + Cl2 NaCl a. adding another Na b. adding another NaCl c. adding another Na and Cl2 d. adding another Na and NaCl Answer 2. The following is a _______________ equation. H2 + Cl2 2HCl Answer 3. Is the following statement true or false? In a double displacement reaction occurs when two compounds recombine to make two new compounds. Answer Assignment See Mrs. Viirre for handouts