test review - Rancho High School
advertisement

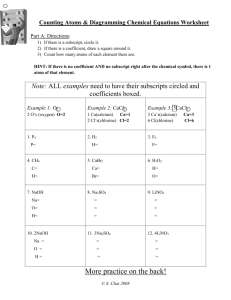
Chapter 11 Review Balance: _Fe + _Cl2 → _FeCl3 In a combustion reaction, one of the reactants is always ____. And the products of combustion reaction are always? Chapter 11 Review Mg(s) + 2HCl(aq) → MgCl2(aq) + H2(g) is an example of what type of reaction? Which set of symbols and explanations is correct: a) (g), grams, or b) (aq), dissolved in water, or c) (s), solid product? Chapter 11 Review Write the following as a balanced equation: nitrogen trifluoride → nitrogen + fluorine Write the equation for the reaction that takes place when solid phosphorus combines with oxygen gas to form solid diphosphorus pentoxide. Chapter 11 Review What must be true in order for this reaction to occur?(think back to activity series) 2Al + 6HCl → 2AlCl3 + 3H2 does the symbol Δ in a chemical equation mean? What Chapter 11 Review Which of the following is a balanced equation representing the decomposition of lead (IV) oxide: a) PbO2 → Pb + 2O, or b) PbO2 → Pb + O2? Chapter 11 Review What type of reaction is: H3PO4 + 3KOH → K3PO4 + 3H2O? Write the balanced equation for the reaction: potassium hydroxide + barium chloride → potassium chloride + barium hydroxide. Chapter 11 Review Chemical equations must be balanced to satisfy ____. Chapter 11 Review Which is true about single replacement reactions: a) any metal can replace any other metal, or b) two reactants produce two products? Chapter 11 Review What are the expected product’s formulas when Na2CO3(aq) reacts with Sn(NO3)2(aq)? Chapter 11 Review In a balanced chemical equation, each side of the equation has the same number of ____. Chapter 11 Review The products of a combustion reaction do NOT include: a) water, or b) hydrogen? Chapter 11 Review Balance the following reaction: KClO3(s) → KCl(s) + O2(g) What type of reaction is: 2C3H7OH + 9O2 → 6CO2 + 8H2O Chapter 11 Review In order to predict whether or not a single-replacement reaction takes place, you need to consult a chart that shows the ____. Which of the following is NOT true about what happens in all chemical reactions: a) the starting substances are called reactants, or b) new atoms are formed as products. How many significant figures are there in 5300054000 What is the correct name for Sn3(PO4)2 When group I A elements form ions they (lose/gain) how many electrons? What are binary covalent compounds? What do binary compounds decompose into? Calculate using correct sig figs 56.3 x 1.23021 What is the product of Mg + N2 What is a different name for a solid produced when two aqueous solutions react? What are the signs that chemical reaction happened?