Calculations Sheet
advertisement

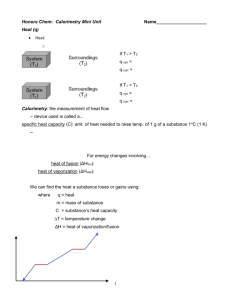
CHEM 1030L — Temperature Measurements Name: ______________ Date: Laboratory Report ______________ Section: ______________ Calculations Print this page and demonstrate how to perform the calculations in each of the sections below. Copy each resulting answer into the shaded spaces in the Data Collection section for grading. Show all work for full credit. For each of the following calculations, circle the final answer. Hint: Remember to include units of measure and demonstrate how they cancel out during the calculation. A. Heating Curve of Water Calculate Mass of Water Calculate Change in Temperature Equation needed to calculate the amount of heat absorbed Calculate Calories of Heat Absorbed Equation needed to convert from calories to kilojoules Convert the above calories to kilojoules Calculations continued on next page CHEM 1030L — Temperature Measurements B. Temperature Measurements Using a Glass Thermometer Convert Temperatures in °C to °F Equation needed to convert from °C to °F Convert Room temperature from °C to °F Convert Tap Water temperature from °C to °F Convert Ice/Water Mixture temperature from °C to °F Convert Ice/Water/Salt Mixture temperature from °C to °F Convert Temperatures in °C to K Equation needed to convert from °C to K Convert Room temperature from °C to K Convert Tap Water temperature from °C to K Convert Ice/Water Mixture temperature from °C to K Convert Ice/Water/Salt Mixture temperature from °C to K Laboratory Report