Structure of Proteins Handout_no questions2014
advertisement

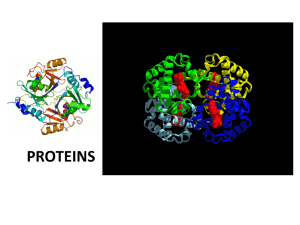
PROTEINS AND PROTEIN STRUCTURE OVERVIEW The functions of proteins are the essence of life itself. They make up more than 50% of the dry mass of animals. There are thousands of different proteins within the cells of living things. Examples and functions are given below. Many of an organism’s proteins are enzymes, special proteins that speed up the rate of chemical reactions in the cell. Enzymes are like tiny molecular tools that temporarily combine with the reactants for a specific reaction and hold them at the correct angle for a reaction to occur. The enzyme may also pull on bonds and loosen them. This lowers the amount of energy needed for the reaction to proceed so it can occur at a much lower temperature than would be necessary without the enzyme (meaning that your cells don’t have to heat up every time a reaction occurs:) Each of the approximately 2000 known enzymes is specific to one particular reaction. Some Functions of Proteins Type of protein Enzymes Structural proteins Hormones Contractile proteins Storage proteins Transport proteins Immunological proteins Toxins Example amylase keratin, collagen insulin, glucagon actin, myosin Function Promotes the break down of starch to the simple sugar glucose. Hair, wool, nails, horns, hoofs, tendons, cartilage Regulates use of blood sugar Contracting fibers in muscle ferritin Stores iron in spleen hemoglobin serum albumin antibodies Carries oxygen in blood Carries fatty acids in blood Rid the body of foreign proteins neurotoxin Cobra venom blocker of nerve functions 1 STRUCTURE OF PROTEINS ELEMENTS found in proteins: Nitrogen in addition to C, H, O Monomers of proteins : amino acids. .Proteins are formed from chains of amino acids. Amino acids are SIMILAR in that each has an amino group NH2, a central carbon, and a carboxyl group (COOH). What makes amino acids different from each other are their side chains – usually labeled as R (see next page) The R group of each amino acid is circled. The R group contributes the unique properties of each of the different amino acids. Proteins are long, unbranched chains of amino acids that fold up into complex shapes . It is the order of the amino acids in a protein that determines its shape and it is the shape that determines the function. Proteins have distinctive shapes because of interactions between the R groups of amino acids in different parts of the chain. This folding forms a 3 dimensional structure which actually allows the protein to be functional. This complex 3-D structure is made up of four “stages” or structures. The primary structure of a protein is the sequence of amino acids. The secondary protein structure occurs when the sequence of amino acids are linked by hydrogen bonds. This level of structure takes the form of either a pleated sheet or a helix. The tertiary structure describes the folding and other contortions of a polypeptide chain that result from the molecular interactions among the R groups of the different amino acids. The arrangement of two or more polypeptide chains in a protein make up its quaternary structure. An example of this type of protein is hemoglobin. 2 Hemoglobin- A protein with quaternary structure 3 PROBLEMS IN PROTEINS Clearly, it is critical for every cell to have a process that guarantees accurate ordering of amino acids in every protein that it needs to carry out its life activities. Having certain amino acids in certain positions is crucial to the protein’s overall shape and consequently to its function. For example, the change of just one amino acid alters the shape of hemoglobin enough to create the condition of sickle cell anemia. Though proteins themselves do not mutate, a mutation in the genetic material of an organism is expressed as a change in the order of amino acids of a protein. Hemoglobin is comprised of four polypeptide subunits (each has tertiary structure). All four components carry a heme group that can bind to oxygen, and all four components must be present to form hemoglobin. The shape of the hemoglobin affects its ability to carry oxygen, and travel freely throughout the circulatory system. A condition that is a result of a malformed hemoglobin unit is sickle-cell anemia. In this condition, a specific glutamic acid is replaced by a valine, and an ionic cross-link is not formed. The result is a severe change of shape of the tertiary structure of the hemoglobin. The new shape of the red blood cell is a crescent or sickle which reduces the oxygen carrying capacity of the red blood cell. The body recognizes sickled cells as “defective” and so they are removed from circulation faster than normal cells. The result is that most sickle cell patients suffer from anemia. The sickled cells can also clump together causing blockages, pain and organ damage3. References Davies, J., Shaffer Littlewood, B., Elementary Biochemistry – An Introduction to the Chemistry of Living Cells, Prentice-Hall Inc, New Jersey, 1979. Koolman, J., Rohm, K-H, Colour Atlas of Biochemistry, Thieme, Stuttgart, 1996. Timberlake, K.C., Chemistry – 5th Edition, Harper-Collins Publishers Inc, NY, 1992. Devlin, T.M., The Textbook of Biochemistry – 3rd Edition, Wiley-Liss Inc, NY, 1992. 4