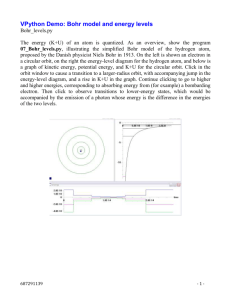
Charlotte Fraza (3829693) - Utrecht University Repository
advertisement

The correspondence principle of Niels Bohr Charlotte Fraza (3829693) Department of History and Philosophy of Science Utrecht University “There was rarely in the history of physics a comprehensive theory which owed so much to one principle as quantum mechanics owed to Bohr's correspondence principle”1 Introduction In modern literature the Correspondence principle is often defined as the requirement for the quantum theory to go over to the classical theories in the limit of large quantum numbers, or in other words as ℎ → 02. Already in the time of Bohr it was the trend to perceive the principle in this matter, and to use it accordingly. Nobelprize winner Max Born also cites Bohr’s correspondence principle as such. In his lectures of 1933 Born states that; “die neue Mechanik für den Grenzfall großer Massen oder großer Bahndimensionen in die klassische Mechanik übergeht”3. In 1925, in the paper The Fundamental Equations of Quantum Mechanics, P. A. M. Dirac named the correspondence principle as the requirement that: “the classical theory gives the right results in the limiting case when the action per cycle of the system is large compared to Planck’s constant ℎ, and in certain other special cases.”4 According to these formulations, the correspondence principle functions as a constraint on theorizing; this means that it only acts in a limit. However this correspondence limit sometimes called ‘Planck’s correspondence principle’ does not capture the full body of the correspondence principle as defined by Niels Bohr. Léon Rosenfeld recalled that he once suggested to Bohr that Planck’s found correspondence between classical theory and quantum theory was the first germ of the correspondence principle, but that Bohr disagreed fervently: ‘He said emphatically that; “it is not the correspondence argument. The requirement that the quantum theory should go over to the classical description for low modes of frequency, is not at all a principle. It is an obvious requirement of the theory”.5 But if it is not as Bohr himself states a limiting principle, what is the full definition of the correspondence principle? 1 Jammer, Max. The conceptual development of quantum mechanics. Tomash, 1989., p. 118. As was the case with Planck’s radiation law in relation to the Rayleigh-jeans law. 3 Born, Max. "Moderne Physik." The Journal of Physical Chemistry 37.6 (1933): 830-831. 4 Dirac, Paul AM. "The fundamental equations of quantum mechanics."Proceedings of the Royal Society of London. Series A, Containing Papers of a Mathematical and Physical Character (1925): 642-653. 5 Rosenfeld, L., 1979 [1973], “The Wave-Particle Dilemma”, in R. Cohen and J. Stachel (eds.) Selected Papers of Léon Rosenfeld (Boston Studies in the Philosophy of Science, Volume 21), Dordrecht: D. Reidel Publishing Co., 688–703. Originally published in J. Mehra (ed.)The Physicist's Conception of Nature, Dordrecht: D. Reidel Publishing Co., pp. 251–263 2 1 While doing a survey on the work of Bohr we will find that there is quite some ambiguity surrounding the correspondence principle and that it may not be fully clear what this principle exactly covers. We will see that for this reason the correspondence principle is often ill understood by fellow scientists. It is therefore important to differentiate between Bohr’s own understanding of the correspondence principle and how his peers eventually used it. In the first chapter we shall mainly focus on Bohr’s own writing on the correspondence principle and in the second chapter elaborate on what status it held in the larger physics community. Also I want to give some special attention to the philosophical considerations of Bohr in chapter 3, as I feel that this philosophical side shaped him as a physicist. I shall refer to earlier papers on the correspondence principle and elaborate on what these papers conclude. It is worth mentioning that Bohr’s statements concerning the correspondence principle changed considerably in his later works and therefore it does not reflect well the way he thought about the correspondence principle in the beginning of his career. For this reason we will leave out his later statements concerning the correspondence principle in the first two chapters and focus primarily on the time period between 19131925. A. Bokulich divided the formulations made by Bohr on the correspondence principle into three categories6; ‘first we have the frequency theorem interpretation, according to which the correspondence principle is an agreement between the Fourier decomposition of the classically defined frequency and the quantum frequency. This interpretation holds in the limit of large quantum numbers and is therefore only an asymptotic agreement. Secondly, there is the intensity interpretation, which signifies the thought that there is a statistical relation between the classical intensity and the quantum intensity. The quantum intensity is then depending on the number of quantum transitions and therefore the probability for this transition to occur. The classical intensity is equal to the square of one component of the classical described motion (Fourier decomposition). Finally, there is also the selection rule interpretation, which is to be understood as the principle that each allowed quantum transition corresponds to one harmonic component in the classical motion. We will see that in the works of Bohr and his peers we will encounter all three interpretations of which we will try to define ‘the’ correspondence principle. I make use of these three interpretations when analyzing Bohr’s work, as they are generally used in literature surrounding Bohr’s correspondence principle. Although not all Bohr’s remarks on the correspondence principle can be subdivided into these categories, it is good as a basic to think about whilst trying to define the correspondence principle. However the use of these three interpretations will actually also underline the point I will make; that the correspondence principle is very difficult to place under one coherent interpretation. I will also show in the paper that the correspondence principle was in its first formulations more a mathematical principle, although not clearly stated in one version by Bohr, and that it slowly changed. At the end of 1925 we will see that it had become more of a guiding principle, which helped Bohr and his peers to look in the right direction for answers surrounding 6 Bokulich, Alisa. "Three Puzzles about Bohr's Correspondence Principle." (2009). 2 atomic questions. So the role and the formulation of the correspondence principle underwent changes that can be brought back to the changes in the atomic model on which it was based. 1 The changing correspondence principle in the work of Bohr (1913-1925) 1.1 The foundation of the correspondence principle Niels Bohr was a Danish physicist who lived from 1885 until 1962. He was born and died in Copenhagen. In 1922 he won the Nobel Prize in physics and is best known for his work on quantum physics. Besides his scientific work, Bohr spent a great deal of his time on the philosophical interpretation of it; in his later years he even became more known for these philosophical contributions than for his contributions to physics. He was in the beginning of his career first and foremost a physicist. His lasting contributions to the quantum theory included the Copenhagen interpretation, the Bohr atom, the thesis of complementarity, and the correspondence principle. The correspondence principle can be called one of the most fascinating and interesting parts of Bohr’s philosophy. It reflects perfectly his personal way of thinking and it was a prime example of what Einstein called Bohr’s ‘musicality’ and ‘unique instinct and tact’.7 To understand the basics correspondence principle one first needs to understand the context in which it was formed: the old quantum theory. The old quantum theory was an “interim” theory, developed on the basis that classical theories could not fully describe atomic systems. Max Jammer, an Israeli physicist and philosopher of physics, aptly describes the pre-1925 'theory' as follows: “Every single quantum-theoretic problem had to be solved first in terms of classical physics; its classical solution had then to pass through the mysterious sieve of quantum conditions or, as it happened in the majority of cases, the classical solution had to be translated into the language of quanta in conformance with the correspondence principle. Usually, the process of finding "the correct translation" was a matter of skilful guessing and intuition rather than of deductive and systematic reasoning.”8 Especially this ‘mysterious sieve of quantum conditions’ was a part of the old quantum theory that was more based on the intuition of a scientist, than on hard mathematical rules. This was also the reason that many scientists first rejected the quantum theory. 7 Kragh, Helge. Niels Bohr and the Quantum Atom: the Bohr model of atomic structure 1913-1925. Oxford University Press, 2012, p. 271. 8 Max, Jammer. "The conceptual development of quantum mechanics.", (1966). 3 The first theory that seemed to solve the problem that classical theories could not describe atomic systems was proposed by Niels Bohr in the Philosophical Magazine of July 1913 in a three-part paper titled ‘On the Constitution of Atoms and Molecules’. It is called an ‘epoch-defining paper’9 and caused the first definite split with the classical theories. In the paper Bohr articulated the postulates of the old quantum theory. In a long letter to Ernest Rutherford, a British physicist known for his great experimental contributions to the atomic theory, on March 6, 1913 he sent his paper and wrote: "Enclosed I send the first chapter of my paper on the constitution of atoms. [...] I hope that you will find that I have taken a reasonable point of view as to the delicate question of the simultaneous use of the old mechanics and of the new assumptions introduced by Planck's theory of radiation. I am very anxious to know what you may think of it all...".10 We see that Bohr knew that his blending of quantum and classical ideas was highly ‘delicate’, but he defined the rules and combined the two theories in an ingenious manner. Bohr used in his paper Ernest Rutherford's model of the atom, according to which a relatively high central charge is concentrated into a very small volume in comparison to the rest of the atom (the nucleus) and with this central volume also containing the bulk of the atomic mass of the atom. The electrons orbit the nucleus in planetary trajectories. However, in an attempt to explain some of the properties of matter with this atom-model Bohr encountered a problem; it was unstable according to classical electrodynamics. According to the model, the electron, an accelerated charged body, radiates energy in such a manner that it would eventually collapse into the nucleus. Bohr's solution to this problem was to incorporate Max Planck's theory: "The result of the discussion of these questions seems to be a general acknowledgment of the inadequacy of the classical electrodynamics in describing the behavior of systems of atomic size. Whatever the alteration in the laws of motion of the electrons may be, it seems necessary to introduce in the laws in question a quantity foreign to the classical electrodynamics, i. e. Planck's constant".11 Bohr summarized his quantum theory by means of two assumptions or postulates: 9 Kragh, Helge. Niels Bohr and the Quantum Atom: the Bohr model of atomic structure 1913-1925. Oxford University Press, 2012, p.1. 10 Applied Physics. Physics 50 years later: Papers as presented to the XIV General Assembly of the International Union of Pure and Applied Physics on the occasion of the union's fiftieth anniversary, September 1972. Ed. Sanborn Conner Brown. Vol. 14. National Academies, 1973. 11 Bohr, Niels. "I. On the constitution of atoms and molecules." The London, Edinburgh, and Dublin Philosophical Magazine and Journal of Science 26.151 (1913): 1-25. 4 “(1) That the dynamical equilibrium of the systems in the stationary states can be discussed by help of the ordinary mechanics, while the passing of the systems between different stationary states cannot be treated on that basis. (2) That the latter process is followed by the emission of a homogeneous radiation, for which the relation between the frequency and the amount of energy emitted is the one given by Planck's theory."12 So the atomic systems can only exist in a series of individual states, which can be thought of as stable periodic orbits around the nucleus. Bohr also often compared these orbits to the orbits of our planetary system, from which he got his inspiration. These periodic orbits are labeled by the principal quantum number 𝑛, with the lowest orbit labeled 𝑛 = 1, the next 𝑛 = 2 etc. When the electron is in one of these states, its motion can be correctly described by classical mechanics. However, when an electron makes a jump from one state to the next, the theory no longer applies. So we see a mix of quantum and classical theories. Whereas the first postulate still relies on the classical mechanics as defined by Isaac Newton, the second postulate is the official break with the classical theories. We will see that, in later papers, this second postulate will develop in to the frequency interpretation. Caution is needed when we speak of the quantum jumps made from the stationary states, because it is now generally accepted that a photon is released, however Bohr himself did not embrace this theory up until the mid-1920s. Bohr preferred to think of the emitted radiation as a wave, rather than a particle. He stated his distrust of light quanta clearly in his lecture in 1922 where he said that: “in spite of its heuristic value, however, the hypothesis of light-quanta, which is quite irreconcilable with so-called interference phenomena, is not able to throw light on the nature of radiation.”13 Bohr’s resistance for accepting the particle theory was also tied to the importance of being able to analyze radiation into its harmonic components, which was in essence tied to the basis of the correspondence principle. As John Stachel notes: “It was indeed, his reliance on the correspondence principle that seems to have been a principal motive for Bohr's distrust of the photon concept”14. In the end Bohr would be led to embrace the concept of the photon and recognize its heuristic value, although not before the collapse of his atomic model. 1.2 “On the quantum theory of line spectra” (1918) Bohr continued to elaborate his work from 1913 and finally in 1917 sent his first part of the paper called “On the quantum theory of line spectra” to the proceedings of the Danish Academy. Bohr had recently been selected a member of the prestigious society and worked 12 Idem Bohr, Niels. "The structure of the atom." Nobel lecture 11 (1922): 14. 14 Stachel, John. "Bohr and the Photon." Quantum Reality, Relativistic Causality, and Closing the Epistemic Circle. Springer Netherlands, 2009. 69-83. 13 5 hard to prove the value of his atomic theory. He chose first to only publish the paper in the academy proceedings rather than submitting it to a regular physics journal. This decision meant a more limited circulation of his paper, but Bohr had his reason, as he wrote to Rutherford in 1917: “This last term I have used all my spare time to complete a long paper on the general principles of the quantum theory. On account, however, of the great difficulties of postal communication I have thought it advisable first to publish it here in the transactions of the Royal Danish Academy, where I could publish it in English, and where I had the special advantage to be able to start the printing before the end of the long paper was quite finished… As soon as I have got a corrected proof of it all I look forward to send it to you.”15 We can see his hesitation to make his paper public, as Bohr felt himself that it was not yet finished. He felt that certain assumptions made in the paper would not be received well, especially concerning the mixing of quantum and classical concepts, and therefore chose to wait before sending it to the larger scientific journals. In 1918 Bohr wanted to publish a series of three papers, which would give a clear overview of the quantum theory thus far. The trilogy of 1918 was “somewhat unmanageable”16 as Bohr himself stated. The sentences are long and often repeated. Bohr did not break up his chapters and often made no clear conclusions. Altogether it was quite hard for the reader to get a grip of Bohr’s thoughts. Pauli later called it the ‘subtlety of Bohr’s style’: “Bohr knew well what he wished not to say when he strove in long sentences to express himself in his scientific papers”17, but it was probably this ‘subtlety’ that got him misunderstood a lot of times. In the introduction of the paper we will find the leading motive in the coming work of Bohr; the search for connections between the classical theory and the quantum theory, that isn’t to say that Bohr believed that there would be such a connection, but he found it a good starting point for further scientific reasoning: “On this state of the theory it might therefore be of interest to make an attempt to discuss the different applications [of the quantum theory] from a uniform point of view, and especially to consider the underlying assumptions in their relations to ordinary mechanics and electrodynamics. Such an attempt has been made in the present paper, and it will be shown that it seems possible to throw some light on the outstanding difficulties by trying to trace the analogy between the quantum theory and the ordinary theory of radiation as closely as possible.”18 15 Nielsen, J. Rud, ed. The correspondence principle (1918-1923). Elsevier, 2013. Kragh, 2012 17 Idem 18 Bohr, Niels. On the quantum theory of line-spectra. Courier Corporation, 2005. 16 6 By “the relation to ordinary mechanics” Bohr meant the adiabatic principle, as described by Paul Ehrenfest, a Dutch and Australian theoretical physicist. This principle plays a role not only in fixing or determining the allowed stationary states, but also in justifying the use of the classical electro-dynamical laws to characterize motion in those states. The Adiabatic Principle “demands that the conditions for the stationary states must be of such a kind that they define certain properties of the motion of the system, which will not change during an adiabatic transformation, if the motion is described ... by help of the usual electrodynamic laws”19. In modern quantum theory, this is to say that a system remains in its eigenvalue if perturbation acting on it is changing slow enough20. With “the relation to electrodynamics” Bohr meant what he would later call the correspondence principle and what we defined as the frequency correspondence principle. One of the interesting changes in the paper compared to his paper in 1913 is that he now called his postulates “fundamental assumptions” in which we can see a growing confidence in the vital nature of his postulates. Moreover, he altered his second assumption to the form of the now better-known frequency condition: “II. That the radiation absorbed or emitted during a transition between two stationary states is ‘unifrequentic’ and possesses a frequency 𝜈, given by the relation 𝐸’ − 𝐸’’ = ℎ𝜈, Where ℎ is Planck’s constant and where 𝐸’ and 𝐸’’ are the values of the energy in the two states under consideration.”21 The second assumption is obviously in contrast with the classical ideas of electrodynamics, as the radiation given off in a transition between stationary states could not be described by classical mechanics. Bohr stated however that it was a necessary assumption to account for experimental facts. The renewed belief in his own ideas can be traced back to Debye and Sommerfeld who had been able to deduce the Zeeman effect in the hydrogen atom from the from the general quantization of multi periodic systems. And Einstein’s theory of radiation had given independent support to the relation 𝐸’ − 𝐸’’ = ℎ𝜈. In the outline of the old quantum theory, Bohr’s two postulates did not yet suffice in giving the correspondence between classically allowed orbits and orbits corresponding to stationary states. First the stationary states and their orbits needed to be determined. In order to determine these stationary states a “quantum condition” needed to be used: ∮ 𝑝𝑑𝜃 = 𝑛h Here the integral is taken over one period of the orbit of the electron, and 𝑝 is the angular momentum, 𝜃 is the angle in the plane of the electron orbit, and n is the quantum number. Max Jammer summarizes: “applying the old quantum theory consists of essentially three steps: first, the application of classical mechanics for the determination of the possible motions of the system; second, the imposition of certain quantum conditions for the 19 Bohr, Niels, Hendrik Anton Kramers, and John Clarke Slater. "LXXVI. The quantum theory of radiation." The London, Edinburgh, and Dublin Philosophical Magazine and Journal of Science 47.281 (1924): 785-802. 20 M. Born and V. A. Fock (1928). "Beweis des Adiabatensatzes". Zeitschrift für Physik A 51 (3–4): 165-180. 21 Bohr 1918a 7 selection of the actual or allowed motions; and third, the treatment of the radiative processes as transitions between allowed motions subject to the Bohr frequency formula”22. To understand how exactly the correspondence principle was used in this context, it is useful to first look at how a one-dimensional atomic-system undergoing a simple periodic motion is classically described. The trajectory is than given by 𝑥(𝑡), which would be the solution of the equations of motion from Newton and will be periodic with a frequency 𝜔, also known as the fundamental frequency. A Fourier series can then describe the position of the motion as follows: 𝑥(𝑡) = 𝐶1 cos(𝜔𝑡) + 𝐶2 cos(2𝜔𝑡) + 𝐶3 cos(3𝜔𝑡) + ⋯ According to the classical theory the frequency of the atom as described by this series should be given by the frequencies in the harmonics23 of the motion: 𝜔, 2𝜔, 3𝜔, etc.; and therefore the spectrum of such a motion is by classical interpretation described by a series of discrete evenly spaced lines. If we follow the reasoning of Bohr in 1918 the radiation of the electron in its orbit is not a result of an accelerated motion (classically thought), but rather an electron jumping from one of the stationary states to the other and by this jump a single frequency, 𝜈, is emitted. The value of this frequency 𝜈 is then given by the Bohr frequency condition24. The spectral lines as seen from experiments are built up out of atoms undergoing transitions between different stationary states, and these lines will be discreet and evenly spaced in the limit of large quantum numbers. 1918 was the first time Bohr mentioned the full description of what could be interpreted as the frequency interpretation of the correspondence principle: “Although, of course, we cannot without a detailed theory of the mechanism of transition obtain an exact calculation of the latter probabilities, unless 𝑛 is large, we may expect that also for small values of 𝑛 the amplitude of the harmonic vibrations corresponding to a given value of τ will in some way give a measure for the probability of a transition between two states for which 𝑛’ – 𝑛’’ is equal to 𝜏.”25 He also speaks of an expectation for the small values of 𝑛 to have an agreement with the classical harmonic system, but he does not further specify how this relation should be interpreted and why it would also hold for small quantum numbers. We can see that although Bohr considered extending his principle to small quantum numbers, he did not formulate his reasoning for this extension, making it quite obscure. Bohr tested this correspondence hypothesis by using it as a premiss in a theoretical calculation of the Rydberg constant, which is a physical constant relating to the atomic spectra. The calculated value of the constant appeared to agree well with the experimentally determined value. Before Bohr’s atomic model this constant could not be 22 Jammer 1966, p. 90. A integer multiply of the fundamental frequency (usually defined for a wave). 24 Bokulich, Alisa. "Bohr's correspondence principle." (2010). 25 Bohr 1918a 23 8 calculated by any of the then existing theories, so this was a strong indicator that Bohr’s atomic model was indeed correct. We should notice that Bohr always spoke of 'agreement of calculations’ concerning this type of correspondence by which he meant that there was a statistical asymptotic agreement between the quantum frequency, 𝑣𝑛’→𝑛’’ of radiation emitted in a quantum jump of difference 𝜏 from state 𝑛’ to 𝑛’’ and the classical frequency 𝜔𝜏 in the 𝜏 𝑡ℎ harmonic of the classical motion in the 𝑛’ stationary state: 𝑣𝑛’→𝑛’’ = 𝜔𝜏 = 𝜏𝜔, for large 𝑛, where 𝑛′ − 𝑛″ = 𝜏. We note here once more that this equation only holds for large quantum numbers, and not for low quantum number transitions, that is to say the equation is only true in the appropriate limit. Bohr described this also aptly during the Solvay conference: “In the limit where the action involved is sufficiently large to permit the neglect of individual quanta, the quantum phenomena can be described as a rational generalization of the classical physical description.”26 So we see here that Bohr now states that the relation only holds when the action is ‘sufficiently’ large, but he does not state when this sufficiency is to be expected and also his earlier considerations to extend the principle to small quantum numbers does not align with this statement. While classically all frequencies will be sent out at the same time, in the quantum mechanical interpretation only a single wave will be emitted with a single frequency between the quantum jumps, therefore one must consider an ensemble of these atoms to compare with the classical spectrum. Einstein introduced this statistical agreement in his theory of heat radiation, on which Bohr commented: “this raises the serious question of whether we must rest content with statements of probabilities for individual processes. As matters stand at present, we are so far from being able to give a real description of these processes that we may well assume that Einstein’s mode of treatment may actually be the most appropriate”27. There seems to be a hint of skepticism in Bohr’s writing on this use of ensembles of atoms to describe quantum processes. Although he embraces the results this method brings he does not quite accept its origins, which rested mostly on the belief that light can be thought of as particles (light quanta). He also declared explicitly that in this context the ties with the classical physics should be mostly understood as a mere heuristic device: “You understand, of course, that I am by no means trying to give what might ordinarily be described as an explanation; nothing has been said here about how and why the radiation is emitted. On one point, however, we may expect a connection with the ordinary conceptions, namely, that it will be possible to calculate the emission of slow electromagnetic oscillations on the basis of electrodynamics”.28 Bohr refrained from giving his thoughts on the origin of radiation and 26 Bohr, N. "The Solvay meetings and the development of quantum mechanics." (1958). Bohr 1922 unpublished lecture; BCW 4, p. 348. 28 N. Bohr ‘Om Brintspektret’ Fysisk Tidsskrift 1914 27 9 instead wanted to give attention to the relation with classical theories that the atomic system shows. He did not want to give some vague explanation without experimental groundings. He merely noticed the problems surrounding the origin of radiation and defined the use of classical concepts in these problems as heuristically interesting, but nothing more than that. One of the main reasons for this disinclination to give an interpretation of the relations between quantum mechanics and classical mechanics was that they were easily misunderstood as trying to gloss over the differences between both theories, which would not be well received by other physicists. The intensity interpretation of the correspondence principle is also found in his paper of 1918, according to which in the limit of large quantum numbers there is a correspondence between the probability of the quantum jumps and the amplitudes of the classical motion: “We must further claim that a relation, as that just proved for the frequencies, will, in the limit of large 𝑛, hold also for the intensities of the different lines in the spectrum. Since now on ordinary electrodynamics the intensities of the radiations corresponding to different values of 𝜏 are directly determined from the coefficients 𝐶𝜏 , we must therefore expect that for large values of 𝑛 these coefficients will on the quantum theory determine the probability of spontaneous transition from a given stationary state for which 𝑛 = 𝑛’ to a neighboring state for which 𝑛 = 𝑛’’ = 𝑛’ − 𝜏””.29 𝑃𝑛’→𝑛” ~𝐶𝜏 (𝑛)2 for large 𝑛 Thus in the limit when 𝑛 gets large the amplitudes of the harmonic components of the electron’s classical orbit can be used to calculate the intensities of the spectral lines. In the classical case the intensity is directly related to the amplitude of the electron, however in the quantum mechanical case the intensity of the spectral line will be determined by how many photons will be admitted and therefore the probability of a particular quantum jump to occur. This result was partially supported by observations on the absorption spectra, where only certain lines with corresponding intensities where viewed. He also elaborates in his paper on conditionally period systems, to prove that the found correspondences hold even outside of periodic systems. He comments: “As far as the frequencies are concerned, we thus see that for conditionally periodic systems there exists a connection between the quantum theory and the ordinary theory of radiation of exactly the same character as that shown to exist in the simple case of periodic systems of one degree of freedom.”30 This was a big step forward in the quantum theory, as before no one could fully explain systems that where not fully periodic. So the second postulate together with the quantum condition gave predictions about the frequencies of the radiation emitted or absorbed in the transitions between the stationary states and gave a connection between the classical amplitudes of the motion and quantum intensities. Despite the empirical successes of the correspondence principle, some physicists remained skeptical about the renowned predictions it was supposed to give. 29 30 Bohr 1918a Idem P.34 10 Rynasiewicz, professor in history of science at the Johns Hopkins University, also noticed that the formulizations of 1918 do not yet yield any predictions about the state of polarization of emitted radiation and also they do not make any predictions about the emission and absorption probabilities. Although later the correspondence principle will get the reputation to do just that: “The above formulation (second postulate of 1918) of the correspondence principle as a constraint on quantum theorizing provides for empirical content at best constraining the determination of stationary states in the limit (of slow frequencies or large quantum numbers). It cannot milk out of the two fundamental postulates any information about transition probabilities or polarization.”31 So the correspondence principle must hold something more than these two relationships or at least it is accounted to hold something more. In 1918 the third use of the correspondence principle can also be found, the selection rule interpretation, which says that; a transition from a stationary state 𝑛’ to another stationary state 𝑛’’ whose separation is 𝜏 is allowed if and only if the exists a 𝜏 𝑡ℎ harmonic in the classical motion of the electron in the stationary state. A visual description of this interpretation is depicted in figure 1. Figure 1: The selection rule interpretation of the correspondence principle where a transition between stationary states corresponds to one harmonic component of the classical motion. If this harmonic component is not found in the classical motion the transition will not occur.32 Bohr concluded that if there was no corresponding harmonic in the Fourier series for large 𝑛 that quantum jump would not occur: “Thus in general there will be a certain probability of an atomic system in a stationary state to pass spontaneously to any other state of smaller energy, but if for all motions of a given system the coefficients 𝐶 [in the Fourier series] are zero for certain values of 𝜏 , we are led to expect that no transition will be possible, for which 𝑛’ – 𝑛’’ is equal to one of these values.”33 We can illustrate this with an simple example34; suppose the solution to Newton’s second equation 𝐹 = 𝑚𝑑2 𝑥 𝑑𝑡 2 and the quantum condition ∮ 𝑝𝑑𝜃 = 𝑛h is; 31 Rynasiewicz, Robert. "The Correspondence Principle." (2013). Based on Fig. 3 of Fedak and Prentis 2002, [http://plato.stanford.edu/entries/bohr-correspondence] 33 Bohr 1918a P.16 34 Bokulich, Alisa. "Bohr's correspondence principle." (2010). 32 11 1 1 𝑥(𝑡, 𝑛) = 𝑛 cos (𝑛2 𝑡) + 𝑛1/2 cos (3𝑛2 𝑡) Which is the Fourier series of a classical periodic motion of an electron in an allowed 1 stationary state 𝑛. We see here that the fundamental frequency is given by 𝜔 = 𝑛2 . There are only first and third harmonics present in the classical motions. Now according to Bohr’s rule of selection, this means that there can only exist quantum jumps between the stationary states that are one or three stationary states apart from each other. We see here that unlike the other interpretations of the correspondence principle this one holds for all quantum numbers and is not only applicable for the limit of large ones. He writes: “This peculiar relation suggests a general law for the occurrence of transitions between stationary states. Thus we shall assume that even when the quantum numbers are small the possibility of transition between two stationary states is connected with the presence of a certain harmonic component in the motion of the system.”35 As this interpretation of the correspondence principle holds for all quantum numbers it has let some of the contemporary history of science researchers to point this relationship as ‘the’ correspondence principle. One of the firmest among them is A. Bokulich who points out that: “Bohr emphasizes in his work that the correspondence principle holds even for low quantum number transitions. For those who see the correspondence principle as only a asymptotic agreement in the region of large quantum numbers between the classical theory and the quantum theory, this will prove a difficult point.”36 Bohr used all three corresponding relationships in his work of 1918, but he did not yet name any one of the ‘the’ correspondence principle, he merely noticed the existence of these correspondences, and instead named them ‘formal analogies’. Therefore, we must look at some of his later work concerning the correspondence principle to get a better background on it. 1.3 “Uber die Serienspektra der Elemente” (eleboration of the CP between 1920-1923) In 1920 Bohr gave in Berlin a lecture called “Uber die Serienspektra der Elemente” to the Deutsche Physikalische Gesellschaft. It was during this occasion that for the first time the term “Korrespondenzprinzip” was used, instead of the term ‘formal analogy’. With this formal naming of his analogies Bohr made clear that he then considered the correspondence principle a fully-grown concept. He reiterated the frequency interpretation of the correspondence principle and also mentioned the intensity interpretation. He explains his expectations as to how the frequency theorem will lead to an expectation for the intensities. He states: “Through this agreement, we obviously obtain a connection between the spectrum and the atomic models of ~ hydrogen that is as intimate as one could hope, if one considers the fundamental difference between the ideas of quantum theory and the 35 1920, “On the series spectra of the elements”, Lecture before the German Physical Society in Berlin (27 April 1920), translated by A. D. Udden, in Bohr (1976), 241–282. 36 Bokulich, Alisa. "Three Puzzles about Bohr's Correspondence Principle." (2009). 12 conventional radiation theory in mind”. 3738 Bohr keeps on emphasizing this ‘fundamental difference’ between quantum theories and classical ones, where the correspondence principle would serve as a bridge to connect the two. He restates this consideration in 1920 in Berlin: “If we now inquire into a deeper meaning of the correspondence established, we are naturally led to expect first that the correspondence arises not only in an agreement of the frequencies of spectral lines determined by the two methods, but will remain valid also for their intensities; an expectation that is equivalent to the proposition that the relative probability of a given transition between two stationary states is connected in an easily stated way with the amplitude of the corresponding harmonic component of the motion.”39 He doesn’t explicitly explain how the connection to the intensities was discovered, but he says that he was ‘naturally led’ to this conclusion, where we see the famous ‘physics intuition’ Bohr was renowned for having. In the years after 1920 Bohr continued to elaborate and rethink his atomic theory and the correspondence principle. He admitted that “the correspondence principle, like all other notions of quantum theory, was of a somewhat formal character”, by which he meant that the validity is independent of any empirical procedure. He also emphasized the successes experimentally obtained with the principle, to soften this notion of a formal character. He said “a reality of the assumptions of spectral theory” of a kind that allowed other physical and chemical properties of atoms to be explained on the same basis. 40 In other words the electron orbits were maybe not as real as our solar system, but to Bohr they provided a universal explanation, which included chemical as well as electoral properties of different atoms. There were, however, a few withstanding problems with Bohr’s atomic theory, as Sommerfeld expressed in a few letters to Bohr: “It seems to me that the goal of determining the number of electrons in each ring still lies far in the future”; “there is too much that is hypothetical about the positions and populations of the rings”41. Sommerfeld noted here one of the biggest difficulties with the model, although it could explain quite some experimental facts, it was not able to determine the position and number of electrons in the rings. This was crucial to know in order for a lot of the chemical properties of the atoms to be explained. Bohr also noted this problem with the atomic theory himself, but he could not seem to find a solution to it. 37 Own translation off: “Durch diese Übereinstimmung haben wir offenbar eine Verbindung zwischen dem Spektrum und dem Atommodelle des ~Wasserstoffs erhalten, die so innig ist, wie man es nur hoffen konnte, wenn man sich den grundsätzlichen Unterschied zwischen den Vorstellungen der Quantentheorie und der üblichen Strahlentheorie vor Augen hält.“ 38 Bohr, Niels. "Über die Serienspektra der Elemente." Zeitschrift für Physik A Hadrons and Nuclei 2.5 (1920): 423-469. 39 Niels Bohr. Uber die Serienspektra der Elemente. Zeitschrift für Physik, 2:423–469, 1920. Translated into English by A. D. Udden as, 431. 40 Niels Bohr. Atomic structure. Nature, 107(2682):104–107, March 1921. 41 Sommerfeld to Bohr, 18 May 1918, quoted from J. L. Heilbron, "The Kossel Sommerfeld Theory and the Ring Atom" 13 Bohr’s thoughts on the atomic theory after 1920 appeared in different formulations and versions of paper that were not necessarily mutually consistent. Although not consistent in some manner, his thoughts where fruitful as he came to insight that there was a problem with the logical point of view behind the correspondence principle. He once again noticed that the use of classical conception in the formulation of a quantum law may not seem consistent. He therefore altered his original expression for the correspondence principle “a formal analogy between the quantum theory and the classical theory”42 and remarked on this insight: “Such an expression might cause misunderstanding, since, in fact … The correspondence Principle must be regarded purely as a law of quantum theory. Which can in no way diminish the contrast between the postulates and electrodynamic theory.”43 The thought; ‘that the essence of the quantum theory and the classical theory was not overbridged, or trying to be overbridged, by the correspondence principle’, was so fundamental to Bohr that he repeated it later in his paper: “In the limiting region of large quantum numbers there is in no wise a question of a gradual diminution of the difference between the description by the quantum theory of the phenomena of radiation and the ideas of classical electrodynamics, but only of an asymptotic agreement of the statistical results.”44 He noted here that there was indeed only a asymptotic agreement of the existing correspondences, which is primarily the essence of the frequency and intensity interpretation of the correspondence principle, but as we see Bohr also states that the correspondence principle is a ‘law of quantum theory’, which implies that it should hold for all quantum numbers. This is only accounted for by the selection rule correspondence. By these statements it not uniformly clear which is the correspondence principle. Although we do see that Bohr used the correspondence principle every time a connection was made between the classical theory and the quantum theory. So Bohr became to call the correspondence principle a law of quantum mechanics and dropped his original inclination to perceive it only as a heuristic tool. He started to consider it as a principle that holds universally true, irrespective of the value of the quantum number (so not only in the region of large quantum numbers). Which would therefore imply, as earlier stated, that the correspondence embodies more than just the frequency or intensity interpretation. He concluded his Nobel lecture in 1922 with the statement: “In spite of the fundamental differences between the quantum points of view and the ordinary conceptions of the phenomena of radiation, it still appears possible on the basis of the general correspondence between the spectrum and the motion in the atom to employ these conceptions in a certain sense as guides in the investigation of spectra”.45 We get here the feeling that instead of a well-defined principle, the correspondence principle would become 42 1918, “The quantum theory of line-spectra”, Det kongelige Danske videnskabernes Selskab, Matematiskefysike Meddelser, 4(1): 1–36; reprinted in Bohr (1976), pp. 67–102. 43 [1923] 1924, “On the application of the quantum theory to atomic structure,” Proceedings of the Cambridge Philosophical Society (supplement), Cambridge: Cambridge University Press, pp. 1–42. First published in Zeitschrift für Physik, 13 (1923): 117. Reprinted in Bohr (1976), pp. 457–499 44 Idem 45 Nielsen, J. Rud, ed. The correspondence principle (1918-1923). Elsevier, 2013. 14 more of a guiding tool, in the end, that would eventually lead to modern quantum mechanics. This was also noted by Jan Faye who said: “The guiding principle behind Bohr's and later Heisenberg's work in the development of a consistent theory of atoms was the correspondence rule.”46 Although the correspondence principle started as a set of semiformal rules, after the fall of Bohr’s atom model it lost most of its grounds on which it was founded on. We will see that especially after this decline of the old atomic models, the correspondence principle developed towards a guiding principle. 1.4 The second atomic theory The optimism of the previous year’s surrounding Bohr’s atomic model was slightly reduced as Bohr’s theory encountered more and more empirical and conceptual problems. One of these problems was that the energies and spectra of many elements and the interactions between radiation and matter such as dispersion could not be explained fully according to Bohr’s atomic theory47. Eventually these faults in Bohr’s theory would bring it to be replaced by Heisenberg’s matrix mechanics and Schrodinger’s wave mechanics and Bohr’s theory would from henceforth be called the old quantum theory. Of course, the role of the correspondence theory was altered in the face of the loss of Bohr’s atomic theory on which it was originally based. In June 1922 Bohr gave a lecture in Göttingen about the so-called ‘second atomic theory’. The need for a renewal of his old atomic theory was no doubt stimulated by the vexing case of the helium atom. It became apparent after several scientists48 tried to solve the problem with help of Bohr’s atomic model of 1918 that it was inadequate to explain it. None of them could find an electron configuration that agreed with the experimental data on the spectrum of helium. As early as 1921 Bohr already stated that the earlier physical models contradicted the correspondence principle and where in fact wrong. He said certain symmetries couldn’t be upheld, from which “we are led... directly to the conclusion that we cannot expect in actual atoms configurations of the type in which the electrons within each group are arranged in rings or configurations of polyhedral symmetry, because the formation of such configurations would claim that all the electrons within each group should be originally bound by the atom at the same time.”49 Why the correspondence principle was the key to understanding the atomic structure was not explained explicitly, as Bohr spoke only in general terms about the principle50. He probably noted that the atomic models did not explain the found experimental data correctly, but the correspondence principle did still 46 Faye, Jan, "Copenhagen Interpretation of Quantum Mechanics", The Stanford Encyclopedia of Philosophy (Fall 2014 Edition), Edward N. Zalta (ed.), 47 Max, Jammer. "The conceptual development of quantum mechanics.", chapter 3 and 4, (1966). 48 Among the physicists who tried to solve the helium problem were J. Franck, F. Reiche, Land, E. C. Kemble, J. H. Van Vleck, Langmuir, Born, Werner Heisenberg, Pauli, Bohr, and H. A. Kramers. 49 Bohr, "Atomic Structure" (February 1921), p. 106. 50 Niels Bohr's Second Atomic Theory, Helge Kragh, Historical Studies in the Physical Sciences, Vol. 10 (1979), pp. 123-186 15 have some heuristic value, as the ties between quantum and classical theory still remained in a certain sense. From the manuscript of the lecture in Göttingen, we can construct the general concept of the revised atomic theory that was primarily built on the correspondence principle. The model that Bohr proposed was, in short, the following; electrons make up various groups and subgroups that correspond to certain quantum numbers and orbits, which are in line with the classic Keplerian ellipses. During the orbiting the electrons penetrate the region of internal electrons, causing a coupling of the revolving electrons, whose motions and numbers where specified by the correspondence principle. Figure 1; Bohr's illustrations for his atomic theory of 1921 extracted from the paper "On the Ground States of the Atoms of the Elements", (Bohr Archive, Copenhagen.) The figures and formulas in the lower left square refer to the 1913 model, whereas those in the right square refer to the 1921 model, which introduced elliptical orbits and the penetration effect; the formulas refer to the mechanical treatment of the new model as Bohr explained in Göttingen in 1922. Although the results of the second atomic theory where quite astonishing; as for the first time the constitution of several noble gases could be explained. The origins of his theory remained quite obscure. No one except for maybe Bohr and his wonder student Kramers could quite follow how the correspondence principle was to be applied and how it restricted the motions that the electrons where allowed to make. The reactions on the theory were mainly positive though. For example Ernest Rutherford expressed his high value of Bohr’s new theoretical ideas, but also announces his bafflement over its origins, he stated: “it is very difficult for me to form an idea of how you arrive at your conclusions. Everybody is eager to know whether you can fix the 'rings of electrons' by the correspondence principle or whether you have recourse to the chemical facts to do so.”51 This was the general response of most of the physicists; that Bohr had with his second atomic theory found a way to formally deduce the atomic movements, but that it was just very ‘difficult’ to understand. 51 Rutherford to Bohr, 26 September 1921 (BSC). 16 Bohr seemed to have made simultaneous and intricate use of two types of considerations while formulating his second atomic theory52; the first one was mostly inductive and relied on the empirical stream of information provided by series spectra, X-ray spectra, chemical properties etc. So although the evidence provided by the spectra was valid, the conclusion drawn on the basis of this evidence was probable. The second type pointed to the deductive use of the general assumptions of the quantum theory, for which Bohr had the hope that they would be self-sufficient. The correspondence principle played a major part in the two types of considerations. Firstly, as a means of connecting empirical spectra and electronic orbits and as an a priori procedure for deriving “exclusion rules” that forbid certain electronic configurations from occurring. The second atomic theory is mostly based on an analogy to the Bohr-Kramers theory of the helium atom, which assumed that the element was stable, and would therefore prove eventually to fail when it appeared that the helium atom is not stable. In 1922 Bohr was awarded the Nobel Prize “for his services in the investigation of the structure of atoms and of the radiation emanating from them”53. In Nobel lectures on the atomic theory Bohr did not overestimate the ground on which his reasoning’s where based and knew them to be shaky. He therefore emphasized the empirical power of the correspondence principle where he paid particular mind to the calculations done by Kramer as proof that: “It is seen that the theory reproduces completely the main feature of the experimental results, and in the light of the correspondence principle we can say that the Stark effect reflects down to the smallest details the action of the electric field on the orbit of the electron in the hydrogen atom, even though in this case the reflection is so distorted that, in contrast with the case of the Zeeman effect, it would scarcely be possible directly to recognize the motion on the basis of the classical ideas of the origin of electromagnetic radiation.”54 Bohr always considered empirical evidence the most important factor of a strong theory and therefore placed strong faith in the correspondence principle, as it was align with the experiments. From here on Bohr continues to emphasize that the allowed transitions between stationary states are connected to the harmonic components in the classical motion, and if these do not occur that the transition will not happen. In the 7th Guthrie lecture, given on March 24, 1922, he says: “This law, which has been called the correspondence principle, states that the occurrence of each transition between two stationary states accompanied by emission of radiation is correlated to one of the constituent harmonic oscillations into which the electric moment of the atom considered as a function of time can be resolved, to the extent that the probability of the occurrence of a transition shall depend on the amplitude of the corresponding harmonic oscillation of the atom, in 52 Darrigol, Olivier. From c-numbers to q-numbers: the classical analogy in the history of quantum theory. Univ of California Press, 1992. 53 The Nobel Prize in Physics 1922". Nobelprize.org. Nobel Media AB 2014. Web. 2 Apr 2015. <http://www.nobelprize.org/nobel_prizes/physics/laureates/1922/> 54 Bohr, 1922, Nobel speech, reprinted in; Bohr, Niels. The Periodic System (1920-1923). Vol. 4. Elsevier, 1977. 17 such a way that in the limit when the quantum-number is large, the intensity of the emitted radiation in unit time in the mean shall be the same as that which would follow from the classical laws of electrodynamics. A similar connection with the classical theory will be exhibited by the polarization of the emitted radiation. If, for instance, the corresponding harmonic oscillation in all states of the atom is a linear vibration or a circular rotation, the radiation will have the same constitution as that which on the classical theory would be emitted by an electron executing harmonic motion of that type.”55 Bohr thus presents all three correspondence relations: the selection rule correspondence, the intensity correspondence and the frequency correspondence. Would the correspondence principle then be a combination of all of these? He does not explicitly states what the correspondence principle entails, but it seems clear that almost every found experimental connection between the classical theories and quantum theory could theoretically be a form of the correspondence principle. As the atomic model begins to crumble, however, we will see that the correspondence principle will undergo a new metamorphosis to be included in the new quantum mechanics. 1.5 The collapse of the old quantum theory At the beginning of 1924 problems began to accumulate surrounding Bohr’s atomic theory. Some of them where old, others new, but in the end they all caused the same effect: the replacement of the old atomic theory. One of the more severe problems belonged to the anomalous Zeeman effect, which showed op for atoms with an odd atomic number (mainly hydrogen). This could not be explained by the rules following Bohr’s model. According to Kragh ‘the atomic models after 1922 became increasingly abstract, symbolic, and formal, which soon would lead to the proposal of a total abandonment of the model concept’56. We can notice with this abstraction of the atomic theory a firm resistance of Bohr, and other physicists, to fully let go of the atomic models, which until then worked quite well. One of the first that would wholly refuse to use the atomic models any further would be Pauli, followed by Heisenberg. In 1924 Bohr, Kramers and Slater proposed a new atomic theory: The Bohr-KramersSlater (BKS) theory. This was perhaps the last attempt to understand the interaction of matter and electromagnetic radiation on the basis of the so-called old quantum theory. It was an attempt to understand radiation from atoms without making use of the concept of light quanta. One aspect, the idea of modelling atomic behaviour under incident electromagnetic radiation using "virtual oscillators" at the absorption and emission 55 Niels Bohr. The effect of electric and magnetic fields on spectral lines. Proceedings of the Physical Society (London), 35:275–302, 1922-3. The Seventh Guthrie Lecture 56 Niels Bohr's Second Atomic Theory, Helge Kragh, Historical Studies in the Physical Sciences, Vol. 10 (1979), p. 313. 18 frequencies, rather than the (different) apparent frequencies of the Bohr orbits. Following Slater, the basic description of the BKS theory was to assign to each atom with an unspecified number of virtual oscillators that produced a virtual radiation field through which the atoms entered in a mutual communication. It remained unclear, however, what these virtual oscillators were, besides that they were to be taken to be abstract and unphysical quantities. This abstract use of the virtual oscillators made the theory vague and unmanageable for many other theorists in the same field. This was also one of the main reasons for the short lifespan of the theory. As mentioned earlier one of the disadvantages of light quanta was that the picture of correspondence was not aligned with it. This could have been one of the reasons why Bohr tried to deny its existence until the very end. In the BKS-theory the correspondence principle was included in the following manner: “It must be remembered that the analogy between the classical theory and the quantum theory as formulated through the correspondence principle is of an essentially formal character, which is especially illustrated by the fact that on the quantum theory the absorption and emission of radiation are coupled to different processes of transition, and thereby to different virtual oscillators” 57. We can see here that the correspondence principle has fully developed in its form as a guiding principle. The principle changed from a set of rules for the connections between classic and quantum theory to a general relation between quantum and classical theory. The new BKS theory was the most appropriate way to include the correspondence idea in terms of space-time pictures. There was a price to be paid though and Bohr made it in form of a relaxation of the energy principle and the “virtualization” of the electromagnetic field. He said: “we abandon on the other hand any attempt at a casual connection between the transitions in distant atoms, and especially a direct application of the principles of conservation of energy and momentum so characteristic for the classical theories.”58 The BKS theory maintained only statistical energy conservation. Which was one of the biggest reasons that the majority of physicists rejected the theory. For example, Pauli wrote as a response to the draft version of the BKS theory: “I laughed a little… about your warm recommendations of the words “communicate” and “virtual” …On the basis of my knowledge of these two words… I have tried to guess what your paper is all about. But I have not succeeded”.59 It was nonetheless influential and would eventually lead to the development of what we now call modern quantum mechanics. Bohr himself was not actively involved in the creation of the new quantum mechanics, but he did talk about the recent developments in the quantum theory. After having received a copy of the work of Heisenberg he commented on it as “a brilliant realization of the efforts which hitherto have served as a guideline in the development of 57 Kragh, Helge. "Bohr—Kramers—Slater Theory." Compendium of Quantum Physics. Springer Berlin Heidelberg, 2009. 62-64. 58 Bohr, Niels, Hendrik Anton Kramers, and John Clarke Slater. "LXXVI. The quantum theory of radiation." The London, Edinburgh, and Dublin Philosophical Magazine and Journal of Science 47.281 (1924): 785-802. 59 Pauli to Bohr, 21 February 1924, reprinted in Hermann et al. 1979, p.147. 19 the atomic theory”.60 He further noted that: “In the nature of matter, there is no longer a question of an independent correspondence principle in the above-mentioned scheme, but rather the whole formulation of quantum mechanics may well be regarded as a sharpening of the content of this principle”61. We see that in Bohr’s opinion the new quantum theory should be regarded as a revolution that found its origin in the correspondence principle. Which is quite interesting, because in the new quantum theory the correspondence principle would not be specifically used. The only form in which it was used was as a guiding principle in translating the classical concepts to the quantum concepts. Although Heisenberg used the correspondence principle in the formulation of his new quantum theory, he denied the fundamental nature of the correspondence principle and did not call it, unlike Bohr, a law of quantum mechanics: “it must be emphasized that this [correspondence] is a purely formal result; it does not follow from any of the physical principles of quantum theory”62. So it was only a mathematical result that did not have any further physical implications. This also signifies a general difference in the attitude of Bohr and Heisenberg in physics. While Heisenberg remained decidedly mathematical and formal in his theories, Bohr took a more philosophical approach and always searched for higher serving relationships between theory and reality. In the end Bohr restricted the principle to the case of the transition from classical theory to quantum theory. He stated: “The correspondence principle expresses the tendency to utilise in the systematic development of the quantum theory every feature of the classical theories in a rational transcription appropriate to the fundamental contrast between the postulates and the classical theories”.63 So the original formulations of the correspondence principle as the frequency theorem interpretation, intensity interpretation and selection rule interpretation were mainly based on the old atomic model, as defined by Bohr, and after this model turned out to be insufficient the correspondence principle developed into a possibility to transcribe the classical components to the quantum ones, which therefore gave a bridge between classical and quantum theoretical theories. 1.6 The Correspondence Principle Defined While reading the works of Bohr and trying to define “the” correspondence principle one comes across a few hardships. First of all, there are two main analogies between the classical and quantum mechanics that Bohr uses throughout his work; the frequency analogy and the intensity analogy. Secondly, there is the use of the selection rule principle that is also named as a correspondence rule; Thirdly, the scope in which the correspondence principle applies is unclear: is it a law of quantum mechanics that is valid for all quantum numbers or is it only valid in an approximation to large quantum numbers? Finally, if the 60 Stolzenburg, Klaus, ed. The Emergence of Quantum Mechanics (Mainly 1924-1926). Elsevier, 2013. Bohr 1925a. Manuscript on ‘Atomic Theory and Mechanics, in Kragh 2012 p. 355. 62 Heisenberg, W. "The Physical Principles of the Quantum Theory (1930)."Scientific Review Papers, Talks, and Books Wissenschaftliche Übersichtsartikel, Vorträge und Bücher (2013): 118., p. 83 63 Bohr, Niels. "Atomic theory and mechanics." Nature 116.2927 (1925). 61 20 correspondence principle is one of these or a combination of these analogies, why does it, according to Bohr, still hold in modern matrix quantum mechanics, as defined by Heisenberg, while the new quantum mechanics is in general seen as the break with all classical relations? One of the important things to remember while looking at the correspondence principle is that it was primarily based on Bohr’s atomic model, which underwent quite a few changes and with it also the correspondence principle. ‘He first considered strictly periodic systems obeying ordinary mechanics (1913-1916), motions of multi periodic systems subjected to ordinary mechanics (1917), multi periodic motions of not necessarily multi periodic systems still subjected to ordinary mechanics (1918-1922), multi periodic motions eluding ordinary mechanics (1922-1925); and finally after 1925 he gave up on the notion of definite electronic orbits.’64 We saw that in the beginning the correspondence principle was primarily the frequency interpretation. Later also the intensities of the system were considered and the selection rules came about, but as soon as Bohr gave up the notion of the definite electronic orbits, these rules also lost their grounds. After that the correspondence principle, became a more ‘obscure’ guiding rule that gave the last connections to the classical theories. One of the most influential scientific elaborations on the correspondence principle appeared in the work of Max Jammer ‘The conceptual Development of Quantum Mechanics’. Jammer writes: “In the limit, therefore, the quantum-theoretic frequency coincides with the classical mechanical frequency. By demanding that this correspondence remain approximately valid also for moderate and small quantum numbers, Bohr generalized and modified into a principle what in the limit may be regarded formally as a theorem.”65 Jammer focuses in his interpretation of the Correspondence principle mainly on the frequency relation. He thereby accounts for Bohr’s writing; that the correspondence principle should hold also for small quantum numbers, by stating that this it approximately valid for small quantum numbers. Jammer is thereby rather dismissive of Bohr's claim that the correspondence principle should be thought of as a law of quantum theory. He writes: “For taking resort to classical physics in order to establish quantum-theoretic predictions, or in other words, constructing a theory whose corroboration depends on premises which conflict with the substance of the theory, is of course a serious inconsistency from the logical point of view. Being fully aware of this difficulty, Bohr attempted repeatedly to show that the correspondence principle must be regarded purely as a law of the quantum theory.”66 He states Bohr wanted to cover up the inconsistencies of his theory by declaring the fundamental nature of the correspondence principle. Oliver Darrigol gives a slightly different interpretation in his overview of the quantum history in his book ‘From c-numbers to q-numbers’. He sees the correspondence principle as primarily the intensity interpretation: “Bohr assumed that, even for moderately excited states, the probability of a given quantum jump was approximately given by the intensity of the corresponding harmonic component of the motion in the initial stationary state. This is what Bohr called the correspondence principle.”67 Strictly speaking this intensity correspondence is exact only in the limit of large quantum numbers, and cannot be 64 Darrigol, Olivier. From c-numbers to q-numbers: the classical analogy in the history of quantum theory. Univ of California Press, 1992. 65 Jammer 1966, p. 111 66 Jammer 1966, p. 116 67 Darrigol 1992, 126 21 extended to small quantum numbers. Darrigol noticed this inconsistency in Bohr’s writing, but he states about the correspondence principle that: “the correspondence principle even in the hands of its creator, failed to provide unambiguous guidance in the building of atoms.”68 Because of the many, not always in alignment, statements of the correspondence principle both Jammer and Darrigol agree that there could also be a possibility that there is no such thing as ‘the’ correspondence principle. Jammer, for example, writes, “Bohr’s numerous and often somewhat conflicting statements, on the essence of the correspondence principle make it difficult, if not impossible, to ascribe to Bohr a clear-cut unvarying conception of the principle”.69 Similarly Darrigol writes: “Confronted with this paradoxical appearance of Bohr’s work, many physicists and historians have renounced the project of giving a rational account of it. In their opinion, Bohr’s success owed much to an unusual tolerance for contradiction …”70. One would except in face of these many contradictions, that the correspondence principle was not well received, however, it is noticeable, that the reactions on the work of Bohr, where mainly positive. There was a general belief in the physics community that there was a concrete basis for the use of the correspondence principle. This belief can be traced back to his original successes with his first atomic theory and therefore could have allowed the later theories of Bohr to hold ‘an unusual tolerance for contradiction’; we will see this also in the second chapter, which elaborates more on the reactions of the physics community on the correspondence principle. Alisa Bokulich states in her paper Three Puzzles about Bohr’s Correspondence principle that the correspondence principle is defined as the selection rule interpretation. Stating that it is the formulation that each allowed quantum transition has a harmonic component in the electron’s classically described motion and if this harmonic is missing then the quantum transition is not allowed. She recognizes in her paper the other two correspondences as formulated by Bohr, but dismisses them as evidence for the selection rule correspondence principle, she writes: “It is important to recognize, however, that none of these particular correspondences, which, in the limit of large 𝑛, allow for a direct calculation of various quantum quantities from the classical harmonic components, are themselves the correspondence principle. Rather they can be used alternatively as inductive evidence for the correspondence principle, or, once the correspondence principle is established, as applications or consequences of the correspondence principle. The correspondence principle is a more general relation underlying these various particular correspondences.”71 So Bokulich found that Bohr intended the correspondence principle to be primarily the selection principle, however this leads to a counterpoint that the selection principle is just a special case of zero probability, but Bohr connects his principle explicitly with both probabilities and polarizations as noted by Rynasiewicz. In general all three correspondence relationships are found in the works of Bohr and all three of them can be accounted as the correspondence principle. We can find parts in the work of Bohr where he names only one of them: “The possibility of the occurrence of a transition, accompanied by radiation, between two states of a multiply periodic system, of quantum numbers for example 𝑛1 , … , 𝑛𝑢 ′ and 𝑛1 ′′, … , 𝑛𝑢 ′′, is conditioned by the presence of certain harmonic components in the expression given by…[the Fourier series expansion of 68 Idem Jammer 1966, 117 70 Darrigol 1997, p. 546 71 Bokulich, Alisa. "Three Puzzles about Bohr's Correspondence Principle." (2009). 69 22 the classical electron motion]. The frequencies 𝜏1 𝜔1 + ⋯ + 𝜏𝑢 𝜔𝑢 of these harmonic components are given by the following equation 𝜏1 = 𝑛1′ − 𝑛1′′ , … , 𝑛𝑢′ − 𝑛𝑢′′ . We, therefore, call these the “corresponding” harmonic components in the motion, and the substance of the above statement we designate as the “Correspondence Principle.”72 This is a prime example of the correspondence principle as defined by the selection rule interpretation. We can however also find writings where two or all three of the interpretations are simultaneously used together: “The demonstration of the asymptotic agreement between spectrum and motion gave rise to the formulation of the “correspondence principle,” according to which the possibility of every transition process connected with emission of radiation is conditioned by the presence of a corresponding harmonic component in the motion of the atom. Not only do the frequencies of the corresponding harmonic components agree asymptotically with the values obtained from the frequency condition in the limit where the energies of the stationary states converge, but also the amplitudes of the mechanical oscillatory components give in this limit an asymptotic measure for the probabilities of the transition processes on which the intensities of the observable spectral lines depend.”73 Here we see that Bohr uses all three of the interpretations, but he again doesn’t explicitly define the correspondence principle. As mentioned before, the atomic theory of Bohr underwent quite some changes in the years between 1913 and 1925 and therefore the correspondence principle also changed. For this reason there is a last opinion in explaining what the correspondence principle holds, which I also account for, that there is no such thing as the correspondence principle. It got altered, changed in basis and formulation and later even used on a more philosophical ground, and did not remain unambiguous in definition. Rynasiewicz also takes this somewhat more extreme vision on the correspondence principle. He says “Bohr was not always clear nor necessarily consistent”74 and states that the correspondence principle evolved as the quantum mechanics evolved; “One comes to suspect that, to the extent that the content of “the” correspondence principle can be articulated, that content must be seen to evolve hand in hand with the evolution of the quantum theory.”75 Thus the correspondence principle changed, got altered and evolved together with its basis: the atomic theory. One can therefore not define ‘the’ correspondence principle. One can only state what the main correspondence relationship was during a certain period in the atomic theory. A general feature of the correspondence principle does remain in all Bohr’s work: a bridge between classical and quantum theory. Also Darrigol writes; “The precise expression and scope of the CP depended on the assumptions made about the electric motion. Whenever this motion was a priori determined the ‘correspondence’ aided in deducing properties of emitted radiation. In the opposite case, characteristics of the electronic motion could be induced from the observed atomic spectra. This ambiguity made the CP a very flexible tool that was able to draw the most from the permanent inflow of empirical data”76. So the correspondence principle was not set in its formulation and changed according to the context it was used in and as this context, the atomic theory, changed so did the correspondence principle. 72 Bohr 1924, p. 22 Bohr 1925a. Manuscript on ‘Atomic Theory and Mechanics 74 Rynasiewicz, Robert. "The Correspondence Principle." (2013). 75 Idem 76 Darrigol 1992, p.83 73 23 2 The reception of the correspondence principle among Bohr’s peers In chapter one we already saw that there was quite some flexibility in the definition of the Correspondence principle and that there was not one coherent interpretation of it. If the principle was however not even definably clear in the work of its founder Bohr, how did his peers perceive it? It is important to understand the distinction between the correspondence principle in Bohr’s own understanding and what it became to mean for the physics community of his time. We can divide the responses to Bohr’s correspondence principle into roughly three categories77; ‘those who misunderstood the principle (e.g., Born and Rosenfeld), those who embraced and developed it (e.g., Kramers and van Vleck), and those who seemed to understand it, but nevertheless mistrusted it (e.g., Sommerfeld and Pauli)’. We cannot say who understood the correspondence principle completely and if it was even possible to do so, as it was so flexible in its definition. Even when we look at the supporters of the correspondence principle we can see that there were quite a few whom, according to Bohr, didn’t fully grasp the correspondence principle. For example Léon Rosenfeld, a Belgian physicist and collaborator of Bohr, wrote about Bohr’s annoyance ‘over his failure to have correctly understood the substance of this principle’.78 Bohr himself was quite aware of the misinterpretations that his often-difficult written essays on the topic could induce. He realized a lot of physicists regarded his work as being ‘philosophical’ and ambiguous. For example he wrote to Sommerfeld: “I should not like you to get the impression that my inclination to pursue obscure, and undoubtedly often false analogies makes me blind to that part of the formation of our conceptions that lies in unveiling of the systematic of the facts. Even if I were blind, I would only need to glance at your book to be healed”79. The book Bohr talks about was Sommerfeld’s famous Atombau that at that time gave one of the most complete overviews of the quantum theory. There is indeed quite an obscurity surrounding the correspondence principle and its use, which persists even in today’s literature. A possible explanation could be that Bohr’s general writing style was complex and sometimes difficult to read, seeing how Bohr’s process of writing was quite different than most scientists. A common writing process is first thinking a thought through before writing it down, for Bohr writing was an important part of his cognitive process. He tried to explain his thoughts in his papers with as much clarity as possible, which sometimes led to a painful process vividly described by Heisenberg: “Bohr would always change the sentences again and again. He could have filled half a page with a few sentences and then everything was crossed out and changed again. And even when the whole paper was almost finished- say, ten pages or so – the next day everything would be 77 Bokulich, Alisa, "Bohr's Correspondence Principle", The Stanford Encyclopedia of Philosophy (Spring 2014 Edition), Edward N. Zalta (ed.). 78 Rosenfeld, L., 1979 [1973], “The Wave-Particle Dilemma”, in R. Cohen and J. Stachel (eds.) Selected Papers of Léon Rosenfeld (Boston Studies in the Philosophy of Science, Volume 21), Dordrecht: D. Reidel Publishing Co., 688–703. Originally published in J. Mehra (ed.) The Physicist's Conception of Nature, Dordrecht: D. Reidel Publishing Co., pp. 251–263. 79 Bohr to Sommerfeld, 21 November 1924, in Stolzenburg 1984 p.38. 24 changed over again. So it was a continuous process of improvement, change and discussions with others… The final text of Bohr’s paper was so subtle and he would think about half an hour whether in a certain case he would use indikativ or konjunktiv and so” 80. We can see that Bohr considered it important to clarify his thoughts as much as possible, however, this sometimes had the opposite effect, making it even more ambiguous. Could it have happened that, because of this ambiguity and the certain flexibility in the interpretation of Bohr’s texts on the correspondence principle, Bohr’s original principle underwent supplementation, modification and metamorphosis? Did it come to have a different function in the physics community than originally intended by Bohr? Multiple physicists, for example Hendrik Kramers and John van Vleck, used and extended the term “correspondence principle” to cover a wide range of correspondence-type arguments that were important in the development of quantum mechanics, but which were not necessarily implied in the formulations of Bohr. Among the physicists whom ‘correctly’ understood Bohr’s principle there were also a lot of critics. Their main argument against Bohr’s atomic theory, not entirely unjustified, was that the basis of it was a messy, patchy combination of classical and quantum elements. Or as Henry Mergenau later phrased it: “Bohr’s atom sat like a baroque tower upon the Gothic base of classical electrodynamics”.81 Among skeptics the most noticeable were Arnold Sommerfeld and Wolfgang Pauli. In this chapter I will try to give some insight into the question “how was the correspondence principle, as formulated in the old quantum theory, received by the physics community?” I will pay attention specifically to the thoughts of the physicists who were closely involved in the development of the quantum theory such as; Sommerfeld, Heisenberg, Pauli, Kramers and others. I believe by looking at the general responses on the correspondence principle of Bohr, we can obtain more insight in the correspondence principle and how it was used and altered. 2.1 Sommerfeld; a magic wand; Die Zauberkraft des, Korrespondenzprinzips. Even though the correspondence principle successfully explained various phenomena, most notable the derivation of selection rules for quantum transitions, it didn’t convince one of Bohr’s biggest competitors at the time, Arnold Sommerfeld. As Darrigol has recounted in detail: “Sommerfeld was never comfortable with Bohr's correspondence principle, and only begrudgingly admitted its fertility”.82 As early as 1919 Sommerfeld wrote to Bohr as a reaction on his paper On the quantum theory of line spectra: “Your formal theory is very interesting and fruitful. However, the hypothesis of Rubinowicz, although not nearly as far-reaching, seems for the present more satisfactory to me.”83 At that time Rubinowicz, a polish physicist, had indeed succeeded in deriving some of the quantum 80 Heisenberg, Werner. "Interview conducted by TS Kuhn (Munich, February 19, 1963)." Archive for the History of Quantum Physics (1963): 16-17. 81 Margenau, Henry. "The nature of physical reality: A philosophy of modern physics." (1950). 82 Darrigol 1992, pp. 138–145. 83 Sommerfeld to Bohr, 5 feb 1919, reprinted in Darrigol 1992. 25 selection rules without the help of the Correspondence principle. He had used an extension of the conservation of angular momentum to radiation processes. Although Bohr found Rubinowicz’s deduction interesting and added his findings in a footnote in the second part of his 1918 memoir, he paid little attention to Sommerfeld’s comment; that it was ‘physically more instructive’ than his correspondence principle. In 1920 in his Berlin lecture Bohr made the following comment: “The conclusions which we can draw by means of the correspondence principle are of a more detailed character than can be obtained solely from a consideration of the conservation of angular momentum. For example that 𝑘 cannot change by more than unity, while the correspondence principle requires that 𝑘 shall vary by unity for every possible transition and that its value cannot remain unchanged. Further, this principle… also enables us to draw conclusions about the relative probabilities of the various possible types of transitions from the values of the amplitudes of the harmonic components”84. From the comment we can see that Bohr had a strong faith that the correspondence principle would prove to be the better theory. One of the strongpoints of the correspondence principle was that it was highly supported by the experimental data. This experimental data accounted for the fact that azimuthal quantum number should be 𝑘 = ±1, which was in alignment with Bohr’s principle, but not that of Rubinowicz’s, which was not able to exclude 𝑘 = 0. This was where the correspondence principle thrived and the theory of Rubinowicz would end up lacking. One of the most important contributions to the quantum physics of Sommerfeld was his famous book Atombau und Spectrallienen, first published in 1919. It was perceived at the time as a good indicator of the current knowledge on quantum physics. Sommerfeld also mentioned the correspondence principle in his book. Although not in the most positive manner “In this way, by remarkably rigorous manner of deduction, reminiscent of the incontrovertible logic of numerical calculations, we have arrived from the principle of conservation of angular momentum at a principle of selection and a rule of polarization […] On the other hand, Bohr has discovered in his principle of correspondence a magic wand, which allows us immediately to make use of the results of the classical wave theory in the quantum theory”.85 The term “magic wand” was clearly ironically intended, as no doubt Sommerfeld preferred a rational claim to the use of magic. Sommerfeld could not get clear insight in the exact use of the correspondence principle, only that it provided the same solutions as the experimental data. The basis of the principle was vague, according to Sommerfeld, and therefore not the right approach to the solution of the quantum problems. In a letter to Bohr on 11 November in 1920 Sommerfeld expressed again his uneasiness with the Correspondence principle, but he also softens his criticism by noting its heuristic value; “In the appendixes to my book you can see that I have gone to some pains to treat your correspondence principle better than in the first edition… Nonetheless I must 84 Bohr, Niels. "Über die Serienspektra der Elemente." Zeitschrift für Physik A Hadrons and Nuclei 2.5 (1920): 423-469. 85 Sommerfeld, A. "Atombau und Spektrallinien. Friedrich Vieweg und Sohn, Braunschweig (1919)." English see Atomic Structure and Spectral Lines, translated from the third German edition by HL Brose, Methuen (1923). 26 confess that your principle, the origin of which is foreign to the quantum theory, is still a source of distress to me, however much I recognize that through it a most important connection between quantum theory and classical electrodynamics is revealed”.86 This distress was caused by Bohr’s use of classical concepts in quantum theoretical context. According to Sommerfeld, Bohr used these concepts without proper explanation and, although they gave the right answers, Sommerfeld found that this was not an appropriate way to conceive physics. In the second (1921) and third (1922) editions of Sommerfeld’s Atombau there was however an honorable place for the correspondence principle: “Bohr’s method is not only of greater consequence [than Rubinowicz’s] in the question of intensity, but also leads to sharper and more definite results as regards the question of polarization… In the matter of method the principle of correspondence has the great advantage that it postulates that Maxwell’s theory be generally valid for long waves, and that it does not throw overboard the many useful results, which the classical theory gives for optical waves and Rontgen rays. In the matter of atomic models we must now recognize the complete superiority of the correspondence principle. For here it seems that Bohr, by applying classical mechanics and electrodynamics to obtain definite statements about the periodic system and the atomic shells, has succeeded to advance where other roads have been inaccessible”.87 This change in attitude came out of the fact that the correspondence principle was able to successfully explain many new empirical results. Sommerfeld had adopted Kramers’ correspondencebased analysis, concluding that now the fine structure of one-electron atoms was perfectly understood: “The exemplary agreement with experiment, which Kramers has obtained in particular in the case of Paschen’s He+ observations and also in the case of the stark effect… confirms convincingly the fertility of Bohr’s correspondence principle.”88 Kramers’ calculation explained the lines observed by Paschen in the hydrogen spectrum. The hydrogen spectrum is an emission spectrum that is divided into a number of spectral lines, where the lines represent the electron making a transition between two energy levels. The Paschen series are then the series of transitions of electrons going from a layer n to the layer n = 3. Kramers’ calculations explained perfectly the transition from the (4,1) → (3,1), however, according to the same reasoning as used in the theory there should have been a fairly noticeable component from the (4,3) → (3,1) transition which was missing in the experiments, but this was strangely not noted for a long time. You could state that Sommerfeld never fully came to terms with the correspondence principle. Although he learned to appreciate its heuristic value he said: “Nonetheless I cannot view it as ultimately satisfying on account of its mixing of quantum-theoretical and classical viewpoints.”89 We once again see here Sommerfeld’s critical attitude towards the mixing of classical and quantum concepts by Bohr. Sommerfeld was not alone in his aversion 86 Sommerfeld to Bohr, 11 November 1920 in Rud Nielsen 1976, p. 690. Sommerfeld, A. "Atombau und Spektrallinien. 88 Idem. 89 Idem. 87 27 towards the ambiguous nature of the correspondence principle. In France, Louis de Broglie commented on the correspondence principle with a hint of criticism. He wrote in 1924 in his thesis: “This idea of correspondence, which seems still imprecise and rather elastic, will have to guide the bold researchers who wish to set up a new electromagnetic theory that agrees better with quantum phenomena than the present theory does”.90 This elasticity was the point that most physicists found offensive and could not accept. In contrast to this ‘elastic’ principle, Sommerfeld and his followers tended to strive toward a more axiomatic deductive approach, where you start with a few self-evident axioms and from there, through the rules of logic, derive new theories. According to him, Bohr’s approach didn’t have the desirable approach in this direction. We can see from Sommerfeld’s opposing attitude that there was definitely some reluctance towards the correspondence principle. This could have been, because of its ambiguous nature or because Bohr never fully explained what it entailed. As we saw the main point of the opposition was the mixing of the quantum and classical rules, as the two theories were fundamentally different. Sommerfeld's critical attitude towards the correspondence principle would prove influential on Wolfgang Pauli and Werner Heisenberg, both of whom were his doctoral students. Especially Pauli would later follow his teacher and fully oppose the correspondence principle. 2.2 Heisenberg; ‘The correspondence principle, as important as the entire quantum theory’ In the beginning of his career Heisenberg, then a young student in München, did not quite agree with his esteemed professor Sommerfeld on the ambiguous nature of the correspondence principle. In his earlier development of the core model of the atom, he placed strong faith in the correspondence principle. In 1921 he wrote to Landé: “The fact that the entire model interpretation of a process can be calculated from purely empirical material is another brilliant achievement of the Bohr correspondence principle, which I am beginning to want to consider as important as the entire quantum theory”91. We see here an example of this faith in the theory of Bohr, which can be traced back to the glamour surrounding Bohr’s name at the time. In June 1922 Bohr gave a series of lectures sponsored by the Wolfkehl Foundation in Gottingen, which became known as the Bohr Festspiele (Bohr festival). He explained his ideas to a large group of physicist, among them also young practitioners Heisenberg, Pauli and Jordan. Heisenberg, then 21 years old, was very impressed with Bohr’s core model and his philosophical views on the atomic theory. He stated: “Bohr’s long stream of thoughts, of which only the beginning was clear, and whose end disappeared in the darkness of a philosophical attitude that was very stimulating to me”.92 Hund, a German physicist whom 90 De Broglie, Louis. Recherches sur la théorie des quanta. Diss. Migration-université en cours d'affectation, 1924. 91 Heisenberg to landé, 29 October 921, quoted in Cassidy 1979 p. 211. 92 Heisenberg 1969, p 45, quoted in Kragh 2012 p. 213. 28 also attended the festival, said about it: “The glamour that surrounded this even cannot be communicated in words today; for us it was as brilliant as the Händel-Festival93 of those days in Göttingen”.94 We can see that Bohr had made quite a name for himself among young physicists, which also gave him great authority in the matters of atomic theory. For example we see this authority in a letter from Heisenberg to Pauli: “Bohr can only explain the diamagnetism by assuming that in a magnetic field He is always oriented in such a way that its moment about the axis of the field is 0. This doesn’t please me very much, but of course one cannot doubt that Bohr is right”95. Heisenberg mentions here one of the problems with the Bohr model for par helium; that the total angular momentum was in apparent contradiction with the experimentally established fact that helium is diamagnetic, but of course no one could doubt that Bohr was right. This reputation provided a large audience to which Bohr could display his new ideas and it also attracted a lot of bright students to his lab, including Heisenberg, who would arrive in 1924 in Copenhagen to work together with Bohr. Here he would come to appreciate the correspondence principle even more. He wrote to Pauli about the role of the correspondence principle in the multiplet problem: “Together with Bohr I have again examined the problem carefully, and we arrived at the conclusion that it is not as Sommerfeld says that the sum-rules cannot be understood with the help of the correspondence principle; on the contrary they are a necessary consequence of the correspondence principle. We are very happy about this interpretation for now the attacks against the correspondence principle are completely refuted. Since recently the correspondence principle has been blamed so much, it would be good to publish it [your results confirming the correspondence principle] [for the greater glory of the correspondence principle].”96 The sentences ‘we are very happy with this interpretation’ referred to the findings that he had made with Bohr to explain the intensities of the multiples lines. It was however not completely clear if the sum-rules could be derived from the correspondence principle or if they were only in consensus with it, but as we will see, Bohr must have been quite convincing in his belief in the correspondence principle, for almost every young student whom visited his lab became convinced of the superiority of the correspondence principle. Someone who was not easy to convince was Pauli, who tended to side with Sommerfeld, believing that the correspondence principle did not reach as far as people wished to believe. Sommerfeld had replied to all the uproar about the correspondence principle by admitting ‘that the correspondence principle is a (highly valuable) limiting theorem of quantum theory, but not its foundation (fundamental principle)’97. Pauli felt in particular that the claim that the correspondence principle could derive the Ornstein sum 93 International music festival that started in 1922 and concentrated on the music of George Friedrich Handel. Hund 1922, quoted in Kragh 2012, p. 224. 95 Heisenberg to Pauli, 6 march 1922, in, Hermann et al. 1979, p.57. 96 Heisenberg to Pauli, September 30th, 1924; quoted in Mehra and Rechenberg pp. 156-157 97 Sommerfeld to Kramers, 5 July 1924, Mehra, Jagdish, and Helmut Rechenberg. The discovery of quantum mechanics, 1925. Vol. 2. Springer Science & Business Media, 2001. 94 29 rules was false. These rules stated that the sum of the intensities of all the lines of a multiplet, which belong to the same initial or final state, is proportional to the statistical weight of the initial or final state98. Concerning this Heisenberg commented: “If by correspondence principle one means, as you do, the false statement that one can come from averaging the classical intensities to those of quantum theory, then you are right that one cannot come to Ornstein’s rule by means of the correspondence principle; however, if one understands a meaningful logical connection to the classical theory, then I am right.”99 This comment illustrates that there was indeed some dispute on what the correspondence principle meant and that it could probably be understood in different ways. Heisenberg said that the correspondence principle is ‘a meaningful logical connexion’, but how this connection is translated into mathematical terms and formulas, is not specified. We can consider this ambiguity surrounding the correspondence principle also one of its strengths. As almost no one was quite sure how the correspondence principle came exactly to play in the quantum theory, it could be bendable to the situation and user. In 1923 Heisenberg would for the first time start with the symbolic translation of the correspondence principle to the new theory of quantum mechanics. He wrote: “The model representations have only a symbolic meaning; they are the classical analogue of the ‘discrete’ quantum theory”.100 Hereby Heisenberg denied any direct physical meaning to the previous made atomic models (which came to a failure in the helium model), but he did integrate a symbolic meaning, which came to play a role in the new discrete theory. In other words the correspondence principle gave a formal analogy between classical mechanics and new discontinuous mechanics. Bohr wrote a letter, which was published in Nature in December 1925 about the importance of Heisenberg’s attempt to use the correspondence between quantum theoretical spectrum and the harmonics of classical motion to define a new kind of quantum theory. He wrote: “In contrast to ordinary mechanics, the new quantum mechanics does not deal with a space-time description of the motion of atomic particles. It operates with manifolds of quantities, which replace the harmonic oscillating components of the motion and symbolize the possibilities of transitions between stationary states in conformity with the correspondence principle. The quantities satisfy certain relations which take the place of the mechanical equations of motion and the quantization rules … In brief, the whole apparatus of the quantum mechanics can be regarded as a precise formulation of the tendencies embodied in the correspondence principle”101. So Bohr stated that the new quantum theory was in full conformity with the correspondence principle and was actually fully based on the correspondence principle. It is true that, as earlier said, Heisenberg made use of a symbolic translation, but to state that this was based on the correspondence is 98 Herzberg, Gerhard, and John William Tranter Spinks. Atomic spectra and atomic structure. Courier Corporation, 1944 99 Heisenberg to Pauli, 8 October 1924, in Hermann et al. 1979, p. 167. 100 Darrigol, Olivier. From c-numbers to q-numbers: the classical analogy in the history of quantum. theory. Univ of California Press, 1992, p.197. 101 Bohr, Niels. "Atomic theory and mechanics." Nature 116.2927 (1925). 30 quite far fetched. It is better formulated that Bohr and the correspondence principle inspired Heisenberg’s translation. Heisenberg, however, also stated that his work was “an attempt to establish a theoretical quantum mechanics analogous to classical mechanics (with use of the correspondence principle)”, but Darrigol has refuted this. He stated this acknowledgment of Heisenberg ‘that the correspondence principle was the basis of his work’ to be a matter of endless debate; was he really trying to describe his own strategy? Or was he trying to win over Bohr, Pauli, and Born, who, at the time, all advocated a radical elimination of unobservable quantities? We can conclude that Heisenberg did a bit of both. He did inscribe in his program a translation of the classical quantities to the quantum ones, but in the end we cannot know how much he was thereby inspired by the correspondence principle and if this use of the CP is in accordance with the original meaning as defined by Bohr. The roots are found in the principle; namely the idea that a formal analogy exists between the laws of quantum theory and those of classical theory. It is interesting to note the change in Heisenberg’s attitude towards the correspondence principle after it became apparent that it failed in describing the helium atom and its properties. He increasingly began to distance his work from the correspondence principle. Mara Beller noted “Heisenberg does not cite Bohr´s work at all (in the 1925 matrix paper), despite the fact that the paper was built on Bohr's correspondence principle in a fundamental way… Instead Heisenberg presented his work as flowing from the positivist principle of elimination of unobservables”.102 Heisenberg expresses his increasing distrust towards the correspondence principle in his book The Physical Principles of the Quantum Theory: “It is true that an ingenious combination of arguments based on the correspondence principle can make the quantum theory of matter together with a classical theory of radiation furnish quantitative values for the transition probabilities… Such a formulation of the radiation problem is far from satisfactory, however, and easily leads to false conclusions.”103 We see that although Heisenberg did use the correspondence principle to guide his work, but he did not give it the original role as a law of quantum mechanics. Heisenberg would continue his belief that the quantum theory does not depend on laws from outside, such as the original classic theories. So the correspondence with the classical theory is a purely mathematical result, not revealing any deeper physical connection. He calls the quantum mechanics a ´Abgeschlossene Theorie´ by which he meant that it was a closed theory, that is to say an axiomatic system complete in and on itself. 2.3 Pauli; The model concepts are in a severe, fundamental crisis, which, I believe, will finally end with a further radical sharpening of the contrast between classical and quantum theory…104 In 1945 Pauli received the Nobel Prize for his exclusion principle and him general contribution to Quantum mechanics. In his speech he described the two main players, Niels 102 Beller, Mara. "Jocular Commemorations: The Copenhagen Spirit." Osiris(1999), p.140. Heisenberg, Werner. "The physical principles of quantum mechanics." U. Chicago Press, Chicago (1930): 21. 104 Pauli to Sommerfeld, 6 december 1924, in Eckert and Märker 2004, p.177. 103 31 Bohr and Arnold Sommerfeld, in the quantum area and their course to dealing with the quantum theory: “At that time there were two approaches to the difficult problems connected with the quantum of action. One was an effort to bring abstract order to the new ideas by looking for a key to translate classical mechanics and electrodynamics into quantum language, which would form a logical generalization of there. This was the direction, which was taken by Bohr’s correspondence principle. Sommerfeld, however, preferred, in view of the difficulties which blocked the use of the concepts of kinematical models, a direct interpretation, as independent of models as possible, of the laws of spectra in terms of integral numbers…”105. This overview of Pauli actually gave the perfect description of the two main ‘sides’ that were picked in quantum theory. One side preferred to maintain the successes of the classical theories and incorporate them in the new quantum theory. The other side, however, wanted an independent theory that did not really on concepts from outside of the theory. Pauli stated to have been influenced by both the sommerfeldian approach and the borisch correspondence approach, but found himself more and more inclined to the second one. He had two major issues with Bohr’s atom theory. First, Pauli objected to the use of a ‘formal’ notion of an electron orbit, on which the correspondence principle is primarily based. Second, Pauli opposed to the thought that the correspondence principle would be able to explain the closing of the electron shells as Bohr had hoped. Pauli believed that the orbital meaning of the azimuthal quantum number (through the correspondence principle) would not hold in the context of multi periodic models. He wrote in his letter to Bohr of 1924: “Against the point of view which you were still holding last fall, I now believe that even for the quantum number k (not only j) essential features of the true laws cannot be reproduced by the theory of multi periodic systems.”106 In Pauli’s opinion, Bohr was wrong to retain classical concepts while he gave up classical laws. In a letter of 1923 to Eddington he alleged that this was the root of all the paradoxes surrounding quantum theory and for that reason should be disposed of. If quantum theory was ever to establish itself as a proper theory it should be formed on an entirely new set of concepts. Pauli understood the correspondence principle mostly as a kind of selection rule. In other words that transitions between stationary states are connected with harmonics in the classical motions. He did not object to this primarily, but he disregarded the extension of the principle; to be able to explain the closing of electron groups in atoms. He wrote to Bohr in 1924: “I have already often said to you that I am of the opinion that the Correspondence Principle has in reality nothing to do with the problem of the closing of the groups in the atom. The exclusion of certain stationary states (not transitions), which is what is in question here, has more similarity in principle with the exclusion of the state 𝑚 = 0 or 𝑘 = 0 in the H-atom than, for example, with the selection rule 𝑘 = ±1. Do you still cling to your application of the Correspondence Principle in this case? There is moreover no need whatever to talk of harmonic interplay.”107 The closing of the electron shells would in the end be explained by the exclusion rule of Pauli, which said that ‘the numbers of the electrons in a closed shell of an atom can be properly reduced to only one electron per state, by which the state is defined as the limited number of quantum states available’. The only problem with the exclusion principle of Pauli was that it couldn’t be properly justified. When Bohr suggested that the correspondence principle was the 105 Pauli, Wolfgang. "Remarks on the History of the Exclusion Principle." Science103.2669 (1946), p.27. Darrigol, Olivier. From c-numbers to q-numbers. 107 Pauli to Bohr, December 12th, 1924; quoted in Serwer 1977, p. 235. 106 32 foundation needed for this justification, Pauli replied: “I personally do not believe, however, that the correspondence principle will lead to a foundation of the rule For weak men, who need the crutch of the idea of unambiguously defined electron orbits and mechanical models, the rule can be grounded as follows: If more than one electron have the same quantum numbers in strong fields, they would have the same orbits and would therefore collide. The justification of the exclusion of the above-mentioned cases in the H-atom by pointing to the collision with the nucleus has never pleased me much. It would be much more satisfying if we could understand directly on the grounds of a more general quantum mechanics (one that deviates from classical mechanics).”108 Pauli was actually quite offended by the idea that the classically based correspondence principle would be the basis of his exclusion principle. We see in his writing the growing influence of Sommerfeld’s ‘direct’ interpretation and his distrust of the model-based reasoning. Pauli would become one of the firmest anti-model voters and would together with Sommerfeld continue for a more direct approach (without the need of models) of the quantum mechanics. They would continue to look for “a radial sharpening of the opposition between classical and quantum theory”, whilst Bohr and Heisenberg opted for a “sharpening of the correspondence principle”. 2.4 Kramers; “In dieser Nacht von Schwierigkeiten und Unwissenheit ist nun das von Bohr 1917 aufgestellte Korrespondenzprinzip ein Leuchtpunkt“109 In 1919 Kramers had already reached, in his dissertation on the “Intensities of spectral lines, some quantitative expressions for the intensities of the hydrogen lines with the help of the correspondence principle: he took the Fourier components of the electron that was orbiting around its nucleus, to give an estimation of the probability of the corresponding quantum transitions. Kramers summarized his conclusions as follows: “… the results obtained as regards the applications to the Stark effect and to the fine structure of the hydrogen lines must be considered as affording a general support for Bohr’s fundamental hypothesis of the connection between the intensity of spectral lines and the amplitudes of the harmonic vibrations into which the motion of the electron in the atom may be resolved”.110 With help of the very extensive calculations, Kramers had arrived at theoretical values of the relative intensities of the Stark lines, which he modestly described as ‘convincing’. The theoretical values and the experimental data matched so well that it gave a strong support for the correspondence principle of which they were derived. Kramers was in everyway a strong supporter of almost all of Bohr’s theories. He could, with the help of these and his strong mathematical knowledge, derive certain properties of atoms that other physicists couldn’t even dream of. It was Kramers who helped the correspondence principle win in conviction with all his different applications of it. Bohr’s correspondence principle was for Kramers and Bohr a lot of times like a guiding principle, which pointed them in the right direction when looking for answers. This 108 Pauli to Bohr December 31st, 1924; quoted in Heilbron 1983, p. 306 and Serwer 1977, p. 236. ‘In this night of difficulties and ignorance it is only the correspondence principle of Bohr from 1917 that established a bright spot’ [kramers 1923] 110 Kramers, Hendrik Anthony. Intensities of spectral lines on the application of the quantum theory to the problem of the relative intensities of the components of the fine structure & of the Stark effect of the lines of the hydrogen spectrum. AF Host & son, 1919. 109 33 direction usually seemed to be the strong connection between quantum physics and classical theories. Kramers could even prove that the principle was useful even for systems without the original limitation of periodicity. In 1920 Kramers published the first concrete application of the correspondence principle outside of the context of multi-periodic systems. The determination of the effect of a small electric field on the fine structure of the hydrogen spectrum: “that one could expect to be able to determine theoretically, according to the principle of correspondence, a relationship between the motion of the atom and the emitted radiation and the polarization state of these components and that it was possible to draw a conclusion about their intensity”111112. We see that Kramers also expected to be able to calculate the intensity components by using the correspondence principle, although nor he nor Bohr gave a direct description on how to do that. It appeared that there was an inside connection between Kramers and Bohr in understanding the quantum theory that other physicists couldn’t understand. In his book of 1923, coauthored by Helge Holst, Kramers mentioned the correspondence principle and that “It is difficult to explain in what it consists, because it cannot be expressed in exact quantitative laws, and it is, on this account, also difficult to apply.”113 So we see that even the favorite student of Bohr had difficulty grasping the full body of the correspondence theory, although he was himself an acknowledged master of making mathematical sense of the correspondence philosophy. If even Kramers couldn’t fully explain the correspondence principle, then perhaps explaining it should be deemed impossible. 2.5 The reception of the second atomic theory; “Wouldn’t that be grand! But the tragedy is that we understand not a word of how he does it.”114 When the news surfaced of a second atomic theory developed by Bohr, the German physicists responded decidedly positive and with a noticeable lack of a critical attitude. Landé, a German physicist, was among the many enthusiasts, he said: “[the atomic theory] will be of the very greatest significance for the future of the atomic theory […] on the whole it seems to me that until your full and detailed exposition appears there is no sense in doing any more theoretical work in the atomic theory”115. Another follower of Bohr’s work Born wrote to Frank, who at that time was staying with Bohr in Copenhagen: “Wouldn’t that be grand! But the tragedy is that we understand not a word of how he does it. He only says that it is based on the correspondence principle … Dear, good Franck, be a nice chap and write to us about it as well as you understand it, or ask Bohr or Kramers to write something clear. Otherwise, we will explode from curiosity.”116 We see here many positive reactions on a theory from which its basis was actually not fully understood. Many physicists assumed 111 das man auf Grund des nach dem Korrespondenzprinzip zu erwartenden Zusammenhanges zwischen der Bewegung des Atoms und der emittierten Strahlung auch den Polarisationszustand dieser Komponenten theoretisch bestimmen konnte und das es möglich war, gewisse Schlusse über ihre Intensität zu ziehen. 112 Kramers, Hendrik A. "Über den Einfluß eines elektrischen Feldes auf die Feinstruktur der Wasserstofflinien." Zeitschrift für Physik A Hadrons and Nuclei3.4 (1920): 199-223. 113 Kramers to holst 1955, reprinted in Kragh 2012, p.204. 114 Born to Frank, reprinted in Kragh 2012, p.298. 115 Landé to Bohr, 3 march 1921, in Rud Nielsen 1977, p. 722 116 Born to Franck, 22 February 1921, as quoted in Greenspan 2005, pp. 104-5, reprinted in Kragh 2012, p.204. 34 Bohr’s theory to be based on formal calculations and therefore a major step towards the mathematization of the physics. Even Sommerfeld’s response was initially largely positive as he wrote: “I am certainly convinced that your way is the right one; if, as it seems, you can mathematically reconstruct the numbers of the periods 2, 8, 18, … then that is indeed the fulfillment of the most daring hopes in atomic physics.”117 In his third edition of Atombau, he stated how excited he was over ‘Bohr’s consideration based on calculations’. Sommerfeld was not the only physicist that read Bohr’s work as being based on rigorous calculations, although an explicit formulation of these calculations was missing in the article send to Nature. The Finnish mathematical physicist Gunnar Nordström, professor at the University of Helsinki, after having studied Bohr’s latest work wrote: “The great and simple lines that you -in spite of the complex relationships- have found to govern the structure pf atomic systems provide your new theory with a suggestive power which makes it appear convincing even in the absence of compelling proof from quantum theory. You have taken a giant step in understanding the world of atoms, and I heartily congratulate you with this new progress”118. Another positive comment came from Rubinowicz, to whom Bohr had sent a reprint of his first report in Nature, he wrote as follows: “Even if your letter to Nature does not let us see the finer details of the wonder-world which you now again have opened up to us, but only foreshadows them, I believe nevertheless to have gathered so much from it that by your work you once more lead us to a summit from which we now again can view a wide field as a clear and beautiful harmonic unity”119. It is interesting that although Bohr’s theory obviously lacked hard proof and some physicists noted this, it didn’t seem to cause any distress and the theory was received as an important step to the complete mathematization of the sciences of matter. Although it was largely received positively, there were also some physicists that expressed their puzzlement over the foundations of Bohr’s work. For example Ehrenfest, whom had also received a copy of the manuscript before the publication in Nature, wrote: “I have read your Nature letter with great eager. The picture you sketch there is really so much more beautiful than that of the quantum numbers decreasing again toward the outside. Of course it now interests me even more how you saw it all in considerations of the correspondence. It interests me especially: what were the difficulties like that taught you to find the way from the conception of the first Nature letter to that of the second. I am VERY curious to know that.”120 Most of the leading physicist thought Bohr’s work to be open a world for a new kind of quantum mechanics, based on formal mathematics and everyone was very curious to know how this derivation looked like. Very few considered the thought that if there was not an explicit derivation in the original paper of Bohr, that this derivation may not exist. It could have been Bohr’s fame that caused the blind acceptance of his new theory while the full derivation was missing. Also Bohr’s writing style always had some 117 Sommerfeld to Bohr, 25 April 1921, in Rud Nielsen 1977, p. 741. Nordström to Bohr, 2 February 1922, reprinted in Kragh 2012, p. 299. 119 Rubinowicz to Bohr, 1922, reprinted in Kragh, p. 299. 120 Ehrenfest to Bohr, 27 September 1921, reprinted in Kragh 1979, p. 137 118 35 ambiguity in it that usually dissolved in further papers, but this time the ambiguity remained. Kramers recounted in 1935 how Bohr had, contrary to popular belief arrived at his theory, not on calculations, but rather on qualitative arguments based on the correspondence principle and empirical data. He wrote: “It is interesting to recollect how many physicists abroad thought at the time of the appearance of Bohr’s theory of the periodic system that it was extensively supported by unpublished calculations which dealt in detail with the structure of the individual atoms, whereas the truth was, in fact, that Bohr had created and elaborated with a divine glance a synthesis between results of a spectroscopic nature and of a chemical nature.”121 So we see that Bohr had arrived at his theory ‘with a divine glance’ which came from his renowned physics intuition. Rather than a formal deduction through mathematics the theory was the based on matching empirical data with chemical properties. Physicist how lacked this ‘divine glance’ could not, but fail to understand how this produced the final theory. As soon as it became known that his new atomic theory was indeed not based on formal calculations, the positive response quickly cooled. Sommerfeld stated in a detailed treatment of the theory: “Bohr’s theory is, of course, not based on mathematics but is conceived intuitively”122. The ‘of course’ shows Sommerfeld’s sceptic attitude towards most of Bohr’s, model based, quantum theories. We see that the enthusiasm that had surfaced at the first release of the second atomic theory had mostly evaporated, although eventually the reception of Bohr’s second atomic theory lost its positivism, the original acceptance of it was blind trust and a firm belief in the ability of Bohr to help the quantum theory to a new level. We can see that the name of a physicist has a lot of impact on the original acceptance of his theory. This can also be the reason that even though the correspondence principle was quite ambiguous in nature and most physicists didn’t fully comprehend it, it held ground for a very long time as one of the pillars of the old quantum theory. 2.6 The general reception of the correspondence principle So we have seen three general responses to the correspondence principle; full acceptance, distrust and misunderstanding. In this chapter we saw that even in the time of Bohr there where only a handful of physicists that could ‘successfully’ apply the correspondence principle. Which makes it quite clear that the writings of Bohr where not unambiguous. The correspondence principle had a wide set of applications and therefore received much admiration. These various contributions were reasons that Bohr was awarded the Nobel Prize in 1922. For the Nobel Prize the speaker Svante Arrhenius, renowned physicist himself, emphasized the worth of the correspondence principle of Bohr. Arrhenius illustrated the use of the correspondence principle by explaining its first use as 121 122 Kramers 1935, reprinted in Kragh 2012, p. 300. Sommerfeld 1924b, reprinted in Kragh 2012, p. 300. 36 defining the structure of the helium atom: “It occurs in two different modifications: one is called parhelium, and the other is called ortho-helium – these were supposed at first to be different substances. The principle of correspondence states that the two electrons in parhelium in the track of rest [stationary orbits] run along two circles, which form an angle of 60 degrees to one another. In ortho-helium, on the other hand, the tracks of the two electrons lie in the same plane, the one being circular, while the other is elliptical.”123 We see here that Arrhenius does not try to explain how these successes where derived with help of the correspondence, seeing how this was a very difficult task. The most that the physics community got from the correspondence principle was that it was widely applicable, it solved many, in that time, experimental problems and gave a direct link between quantum physics and classical theories, but how all this came to play in the formal theory was a mystery to most physicists. There was certain flexibility in how the correspondence principle was to be understood and used. Ehrenfest emphasized the need to maintain this adaptability of the correspondence principle: “It is not desirable that, with the most automatic application in view, one already casts in a rigid form the condition of correspondence, which up to now has been variable and groping”124. So the quantum theory had well-established quantum rules, but the correspondence principle was the connecting tool that could be different in each hand. For this reason there were also a lot of extensions on the basics of the correspondence principle. For example van Vleck wrote two articles called; ‘the most sophisticated application of the correspondence principle’ and ‘Some extensions of the correspondence principle’, which included a correspondence theory of absorption. In these articles he looked for new areas in which the correspondence principle could also be used. So the correspondence principle was not only used in its original form, but also extended and formed to be applicable in situations, even outside of the scope of physics. Later the term correspondence principle was remolded to serve as a general principle of the reduction of a new scientific theory to the old one in appropriate limits, although this was not how Bohr had defined it. There were also complete misinterpretations of the correspondence principle in the physics community. Campbell had claimed that the electrons in the Bohr-Sommerfeld theory do not really move on the orbits and, in justification, had cited the correspondence principle. Bohr was completely against such an interpretation; “On the contrary of we admit the soundness of the quantum theory of spectra, the principle of correspondence would seem to afford perhaps the strongest inducement to seek an interpretation of the other physical and chemical properties of the elements on the same line as the interpretation of their series spectra.”125 Where these miscomprehensions caused by the ‘unilluminating’ papers of Bohr or was it the term ‘correspondence principle’ that was misused in the physics community? It was probably a 123 Nobel Prize in Physics 1922 - Presentation Speech. Nobelprize.org. Nobel Media AB 2014. Web. 21 May 2015. <http://www.nobelprize.org/nobel_prizes/physics/laureates/1922/press.html> 124 Ehrenfest 1923a, reprinted in Kragh 2012, p. 142. 125 Bohr, Niels. Atomic structure. Printed in Great Britain by the Cornwall Press, 1921. 37 bit of both, as Bohr was not very clear, physicists could fill in their own understanding on the use of the correspondence principle. In general we can state the correspondence principle was well received. In the beginning of the old quantum theory the correspondence principle was more a formal mathematical application, but as the atomic theory changed and with it the correspondence principle, this mathematical application made room for a more philosophical interpretation, which we shall elaborate in the third chapter of this paper. During the second atomic model we saw how much influence the name Bohr had, because of earlier success there was room for some ambiguity in Bohr´s newer theories. In every hand the correspondence principle could take a new form and in the end it seemed that all connections from former classical theories to quantum theories could be deemed a part of the correspondence principle. A large part of this was fueled by the explanations the correspondence principle provided for experiments, also that it was quite easy to visualize and fitted in the general belief of the existence of one universal theory. 38 3. Bohr as a Philosopher “The significance of science […] is concerned with learning the structures of the world that lie beyond the expressions and concepts of any particular language”126 Niels Bohr was renowned for his contributions to the quantum theory, but also for his philosophical views. Heisenberg even stated that ‘Bohr was primarily a philosopher not a physicist’127. However if one reads Bohr’s work, it is quite clear that Bohr does not consider himself a full-fledged philosopher: “It is not my intention to enter into an academic philosophical discourse for which I would hardly possess the required scholarship”128. Even though Bohr did not consider himself a philosopher, his work in physics was primarily spurred by his philosophical considerations. Rosenfeld also noted this: “epistemological considerations played a decisive part in ... investigations, and that, inversely, the results of the latter led … to a deeper understanding of the theory of knowledge”129. Bohr was convinced that a new mathematical formulation had to come after a physical understanding of the problem. According to Heisenberg: “Bohr would always say, ‘we’ll, first we have to understand how physics works. Only when we have completely understood what it is all about can we then hope to represent it by mathematical schemes’”130. Bohr believed mathematics to be the tool to discover the ‘real’ physics. He did not believe a description of nature based solely on mathematics to be a good one, he wanted a further reaching interpretation linked to reality. This was also the big difference between Bohr and Heisenberg whom “wanted to start entirely from the mathematical scheme of quantum theory…”131, however this was something Bohr would never quite accept, as he also wanted a further reaching interpretation linked to physical world. Bohr’s philosophy can be said to be sui generis, or one of a kind, although when we take a closer look at the works of Bohr we will see hints of Kant’s, Kierkegaard’s, Høffding’s and other philosopher’s thoughts. The relation of the works from these philosophers to Bohr’s theories is, however, far from obvious, as Bohr does not in his work explicitly quote or mentions them. Even though the similarities between Bohr and other philosophers are not quite so obvious, it is interesting to study these similarities, primarily Høffding and Kant, because by comparison and differentiating we will be able to understand Bohr’s work better. The Kantian part of the philosophy of Bohr has been analyzed by a number of historians, among them Hooker 1972, Honner 1982 and recently Brock 2003. I will also address some of the Kantian parts of Bohr’s philosophy in this chapter. Although Bohr himself never explicitly mentions having been influenced by the thoughts of Kant, we can 126 Hesse, Mary. "Comment on Von Weizsäcker." Transcendental Arguments and Science. Springer Netherlands, 1979. 159-170. 127 Heisenberg on Bohr, reprinted in Plotnitsky, Arkady. Reading Bohr: Physics and Philosophy: Physics and Philosophy. Vol. 152. Springer Science & Business Media, 2006. P.146. 128 Bohr, Niels. "Essays 1958-1962 on atomic physics and human knowledge." (1963). 129 L. Rosenfeld ‘Biographical sketch’, reprinted in Honner, John. "The transcendental philosophy of Niels Bohr." Studies in History and Philosophy of Science Part A 13.1 (1982): 1-29. 130 Interview with Heisenberg by T. S. Kuhn on 19 February 1963. American Institute of Physics, Niels Bohr Library and Archives. 131 Interview with Heisenberg, 25 February 1963, reprinted in Kalckar, Jørgen, ed. Foundations of Quantum Physics I (1926-1932). Vol. 6. Elsevier, 2013. 39 find some of the same thought patterns in both works. Honner stated that Bohr’s philosophical views can be seen as transcendental, which means that there is a certain allowance for paradoxes in the conditions to obtain knowledge of the system. We will later see how this transcendental part of Bohr’s philosophy relates to Kant. In this chapter I will not try to label the philosophy of Bohr as being either positive or pragmatic, for I think Bohr’s philosophic thoughts are too subtle for that. Moreover, his thoughts and theories changed quite a bit over the course of time, making it more difficult to label them. Seeing how Bohr is sometimes called primarily a philosopher, I intend to take a closer look at how these philosophies correlate to his work in physics, and perhaps stimulated the first expressions of his atomic model and with it the correspondence principle. I think it is necessary to thoroughly analyze the philosophical considerations of Bohr in order to understand the origin of the correspondence principle. During this analysis I will make extensive use of his later work, as Bohr articulates his philosophical thoughts more clearly then. While using these works I will keep in mind that Bohr’s ideas could have altered significantly compared to the time of the first formulation of the correspondence principle. There are however a few leading considerations made by Bohr that can be found starting from his earlier works until his final papers. 3.1 Inconsistencies in the quantum theory and empirical efficiency Until 1925 when Heisenberg came up with the new formulation of the quantum theory, the quantum theory contained some significant inconsistencies. The Bohr model, which was then primarily used to describe the quantum system, was in its founding not fully consistent, some of its concepts came from classical theory, while they applied in a quantum environment. Even though Bohr was aware of these inconsistencies it seemed he allowed them. With regard to these inconsistencies, Heisenberg and Thomas Kuhn expressed their opinions in 1963. Kuhn was of the opinion that Bohr was usually eager to get rid of the inconsistencies although there was a sort of ‘higher inconsistency’, which he was willing to except. Heisenberg, however, pointed out that whilst reading Bohr’s work one should distinguish between ‘paradox’ and ‘inconsistency’, and that Kuhn’s ‘higher inconsistency’ might belong to the first class.132 Bohr, according to Heisenberg, strongly believed that certain paradoxes would remain even in the final version of quantum theory: “Because he was so much impressed by these paradoxes which were apparently unavoidable he counted always on the possibility, ‘Well, these paradoxes may even, in the long run, mean some kind of inconsistency which cannot be avoided’. On the other hand, in the course of the development, Bohr realized that an inconsistency is something still much worse than a paradox because inconsistency means that you talk just nonsense- that you do not know what you are talking about. A paradox may be very disagreeable but still you can make it work. An inconsistency can never be made to work”133. This comment shows us that Bohr expected the quantum theory, or theories in physics in general, to always keep some form of inconsistency, or in his words paradoxes. According to Bohr these inconsistencies did not come from nature, but from our own observations of nature. He claims that as our means to make observations will never be perfect and always create a superficial context, a certain extent of inconsistency is to be expected. That is not to say that Bohr did not believe that 132 133 Kragh 2012, p. 366. Interview by T. S. Kuhn of 25 February 1963. American Institute of physics. 40 nature could not be fully explained by physical laws, he only stated that he believed everything was context related. We cannot create a frame in which our observation methods do not alter the object that we are observing, so it is only possible to a certain extent to give a full representation of nature in the form of physical laws, based on measurements. The unfinished manuscript in 1927 ‘Philosophical Foundations of Quantum theory’ summarizes one of the main points Bohr was trying to make within his philosophical considerations: “Due to the contrast between the principles underlying the ordinary description of natural phenomena and the element of discontinuity characteristic for quantum theory, we must be prepared that every concept used in accounting for the experimental evidence will have only restricted validity when dealing with atomic phenomena.”134 He stresses once again that we must keep in mind that we use classical descriptive words for the observations made on a quantum level. This cannot be unambiguously done and must therefore always be taken into account when trying to define a new theory on atomic level. These reflections between words and reality, classical and quantum will remain a reoccurring problem in the works of Bohr and this Bohrisch sense of objectivity will be more extensively explained later in this chapter. Although sometimes a theory turns out to be wrong, as was the case with Bohr’s theory at the end of 1924, Bohr believed that a future theory needed to contain the same strong values as the previous one. In the case of Bohr’s atom model, its strong empirical record should still agree with the future theory. He wrote to Born: “It is necessary for us to regard the usual formulation of the quantum theory, as it is elaborated for atomic structure, as a limiting case of a more general theory.”135 According to Bohr the connection to the classical theory was one of the important features of the old theory that the new theory should also contain. According to Darrigol, Bohr “did not regard logical completeness as prior to empirical efficiency. He believed instead that progress was possible within a manifestly incomplete theory, under the guidance of organizing principles like the correspondence principle”136. From this we can conclude that if, before describing the physical reality, a theory can explain the experimental data, Bohr would consider it sufficiently correct. According to him the theory does not have to be a reflection of the ‘real’ world. The Borhish approach to physics was largely empirically driven. He used well-defined phenomena as a good starting point and as new quantum effects were introduced, he altered the theory to satisfy these effects. He attempted to determine the atomic structure from the experimentally found quantum properties. He used the spectral evidence without having a real understanding of the physics behind the emission of this radiation. Bohr sometimes referred to conclusions reached through this method as “empirical deductions from the spectral evidence”.137 Tanona said this process explicitly required a tool to allow one to bridge the gap between phenomena and theory. The correspondence principle was 134 N. Bohr, ‘Philosophical foundations of quantum theory’, "Essays 1958-1962 on atomic physics and human knowledge." (1963). 135 Bohr to Born, 9 December 1924, in Stolzenburg 1984, p.73. 136 Darrigol, Olivier. From c-numbers to q-numbers: the classical analogy in the history of quantum theory. University of California Press, 1992. 137 Bohr, Niels, Hendrik Anton Kramers, and John Clarke Slater. "LXXVI. The quantum theory of radiation." The London, Edinburgh, and Dublin Philosophical Magazine and Journal of Science 47.281 (1924): 785-802. 41 this tool, which together with Bohr’s frequency condition acted as the bridge that allowed him to make connections between classical and quantum theory based on empirical phenomena. 3.2 Formal through the eyes of Bohr It is sometimes difficult to understand Bohr’s subtle physics and philosophical theories as he had his own set of definitions. One of them was the word formal; a formal principle according to Bohr: ‘has significance because of the role it plays in a theory, rather than because of any direct physical reality’138. The definition of the word formal actually gives quite a good summary of Bohr’s general striving. Instead of giving physics the role of describing the physical world as it is, he wants it to give a descriptive approximation that is as close to reality as possible. In this manner he does not consider mathematical laws to give a representation of the real physical world, but an idealization in formal laws. Darrigol said that ‘Analogical thinking usually works with a touch of blindness: formal relations of a given theory are tentatively applied to new objects, and if the operation is empirically successful, the concepts originally underlying these relations are assumed to extend to these objects.’139 So if the theory is confirmed by the experiment, it is as close as we can get to a good description of the physical world. The correspondence principle was not, as many think, a way for Bohr to secretly include the classical theories in the new quantum ones. It was primarily formal and was not meant to give a reflection of reality, but a relation to it. In quantum mechanics Bohr still used the same concepts as momentum, energy and frequency as formulated in the classical theory, related through the correspondence principle. This led to quite some discussion and confusion, as it seemed that the quantum theory was self-contradictory and, although for its heuristics value, its foundations were unclear, as these were largely based on classical concepts. Bohr realized that certain fundamental concepts from the classical theory could still be used in the quantum theory, within a range of certain quantum numbers (although Bohr made some contradicting statements about this). He used new empirical data to support the use of classical concepts in quantum physics and his theories evolved around this process. Here we must again remember that although Bohr was stated to be a philosopher he placed strong emphasis on the empirical data and was always ready to reconsider his theories if they did not match the experiments. This is a classical characteristic of a physicist, but sometimes overlooked in the case of Bohr. 3.3 The relation to the classical theories and the unity of science 138 MacKinnon, Edward. "Niels Bohr on the Unity of Science." PSA: Proceedings of the Biennial Meeting of the Philosophy of Science Association. Philosophy of Science Association, 1980. 139 Darrigol, Olivier. From c-numbers to q-numbers: the classical analogy in the history of quantum theory. Univ of California Press, 1992. 42 Although Bohr called the relation between quantum theories and classical theories primarily formal, that is not to say that he considered the relation to classical theories dispensable. Everything in our daily lives is lived through the laws of classical physics. Honner stated that Bohr saw classical physics and quantum physics as ‘mutually complementing each other, despite their inherent contradiction.’140 It is possible to predict with classical physics the state of a system at present and future time, if the necessary parameters are provided, however for a quantum system this is not as simple, as the system is altered by the measurement. Bohr repeatedly pointed out that through the measuring apparatus there would exist an indispensable link between quantum physics and classical physics. Heisenberg commented on this view of Bohr: “Bohr has rightly pointed out on many occasions that the connection with the external world is one of the necessary conditions for the measuring apparatus to perform its function, since the behavior of the measuring apparatus must be capable of being … described in terms of simple (classical) concepts, if the apparatus is to be used as a measuring instrument at all. And the connection with the external world is therefore necessary”141. Heisenberg rightfully summarised Bohr’s view in the following manner: that quantum systems are, in the end, part of a bigger system, and through the interaction with this bigger system, classical concepts come into play. The bigger system is the unknown factor in the quantum equations and gives a ‘new statistical element into the description’142. It was, according to Bohr, therefore not possible in the description of quantum systems to completely rid oneself of classical concepts. A. Bokulich also noticed the strong emphasis Bohr placed on classical concepts and therefore states that Niels Bohr’s general approach to physics is in many ways more that of a continuity theorist than a revolutionary. He is a continuity theorist in the sense that he tries to maintain and emphasize those features of the predecessor theory that are preserved in the transition to the successor theory. I agree that Bohr does try to maintain a link to the classical theories, which he deems necessary, but not because he thinks that a theory cannot evolve. He states that our perception of reality cannot be broken from the classical view of the world. It must be emphasized here that Bohr thought the use of the classical concepts indispensable to the quantum theory. So it was not, as for example Heisenberg said, just a case of we do use the classical concepts, but a case of we must us the classical concepts. This emphasis on the use of the classical concepts was because Bohr thought these were necessary to provide an unambiguous communication in physics. He stated: “the unambiguous interpretation of any measurement must be essentially framed in terms of classical physical theories, and we may say that in the sense the language of Newton and Maxwell will remain the language of physics for all time”143. When reading this passage of Bohr it does bring one question to mind; why can we describe observations only in a classical matter? One time Schrodinger posed the question, but Bohr’s reply simply restated “the seemingly obvious fact that the functioning of the measuring apparatus must 140 Honner, John. "The transcendental philosophy of Niels Bohr." Studies in History and Philosophy of Science Part A 13.1 (1982), p 21. 141 Heisenberg, Werner. "The development of the interpretation of the quantum theory." Niels Bohr and the development of physics 1 (1955): 12, pp. 26-7. 142 Heisenberg, 1989, pp. 121-2 143 Bohr 1931, reprinted in Greenberger, Daniel, Wolfgang L. Reiter, and Anton Zeilinger, eds. Epistemological and experimental perspectives on quantum physics. Vol. 7. Springer, 1999. 43 be described in space and time”144, which obviously does not suffice as an answer to the question; why this is to be done so? Aage Petersen, a Danish professor and assistant of Bohr, argued that “Bohr’s remarks on the indispensability of classical concepts are based on his general attitude to the epistemological status of the language and to the meaning of unambiguous conceptual communication, and that they should be interpreted in that background”145. In order for a measurement to be able to be done in the quantum domain and conveyed, we must consider that at least part of the instrument must be described according to the classical rules. Honner said that objectivity according to Bohr “lay in the refinement of unambiguous communication based on a well-defined use of pictures and ideas referring to the events of daily lives”146. This also sums up the relation that Bohr assumed necessary between classical and quantum theory, because all our descriptive methods were based on objects described in a classical nature, it was not possible according to Bohr to, even in the realms of quantum theory, completely let loose of these terms and definitions, because of the need for unambiguous communication. Bohr did not think full objectivity in physics was possible, because people were already pre-instructed on the situation and properties of the experiment just by daily life. It is therefore impossible to achieve objectivity such as ‘the eye of God’, or, as Bohr calls it: “… a radical revision of our attitude toward the problem of physical reality. In fact, as we shall see, a criterion of reality like that proposed by the named authors contains – however cautious its formulation may appear—an essential ambiguity when it is applied to the actual problems with which we are here concerned.”147 Bohr did not view this lack of objectivity as a limit in physics alone, but a general limit in all sciences. The framework we live in will always present a certain context that will limit our objectivity. He stated this key idea of conceptual frameworks in 1955, where he wrote: “The main point to realize is that all knowledge presents itself within a conceptual framework adapted to account for previous experiences and that any such frame may prove too narrow to comprehend new experiences … . When speaking of a conceptual framework, we refer merely to unambiguous logical representation of relations between experience.”148 Especially the last sentence gives a good overview of Bohr’s general concern when involved with physics. One should not strive to have the finest objectivity, but strive to give the best unambiguous relationships between experience and theory. Physics is a science of relationships, according to Bohr, and physicists should aspire to find these relationships and present them in comprehensible theories. He explicitly stated this task for physicists in 1934: “… our task must be to account for such experience in a manner independent of individual subjective judgment and therefore objective in the sense that it can be 144 Bohr 1996, pp. 511-2, reprinted in Schlosshauer, Maximilian, and Kristian Camilleri. "The quantum-toclassical transition: Bohr's doctrine of classical concepts, emergent classicality, and decoherence." arXiv preprint arXiv:0804.1609 (2008). 145 A. Peterson, Quantum Physics and the Philosophical Tradition, MIT Press, Cambridge, 1968, p. 179. 146 Honner, John. "The transcendental philosophy of Niels Bohr." Studies in History and Philosophy of Science Part A 13.1 (1982). 147 Bohr, Niels. "Can quantum-mechanical description of physical reality be considered complete?." Physical review 48.8 (1935): 696. 148 Bohr, Niels 1958b, pp. 6-7, "Essays 1958-1962 on atomic physics and human knowledge." (1963). 44 unambiguously communicated in common human language”149. So a physicist should be an objective observer who tries to make an unambiguous account of that which he observes. The analysis that came after the observation was something that Bohr had made an art out of. Or as said by his brother Harald “His intuitive grasp of the situation was overwhelmingly convincing notwithstanding the necessary subsequent farther-reaching quantummechanical analysis”150. Bohr was equally infamous for this intuitive grasp, as it allowed him to penetrate the situation, but not to explain it, in a well-rounded manner, to his maybe ‘less’ talented peers. As already stated before, Bohr was not a reality searcher in the sense that he believed that physics should reflect reality or objects as they actually exist in nature. As Heisenberg explains: “Bohr has expressed himself in discussions somewhat as follows; classical physics and the quantum theory, taken as descriptions of nature, are both caricatures; they allow us, so to speak, to asymptotically represent actual events in two extreme regions of nature” 151 We see here that Bohr in general perceived theories to be more asymptotically caricatures of reality. Bohr describes the quantum theory as ‘a rational generalization’ of the classical theories. He sees the new matrix mechanics as defined by Heisenberg as a continuous development of the correspondence principle, which laid the link between the classical theories and the quantum theories with the help of the Fourier development. He emphasizes this point in 1929 when he writes, “We are dealing here with an unbroken development . . . which, beginning with the fundamental works of Planck on black body radiation, has reached a temporary climax, in recent years, in the formulation of a symbolic quantum mechanics”152. He states here that there is a continuity in the sense that the old theory, the classical theory, is incorporated in the new one, the quantum theory, where the former is a special case only allowed in a limited context of the preceding theory. We see here once again the search for relationships between nature and reality, but also between classical theory and quantum theory. In his quest to generalize classical theories, Bohr primarily used the correspondence principle as his tool. For example he writes: “The correspondence principle expresses the tendency to utilize in the systematic development of the quantum theory every feature of the classical theories in a rational transcription appropriate to the fundamental contrast between the postulates and the classical theories”153. Bohr tries to bring quantum theory and classical theory together into one coherent whole. It is interesting to note the change in his philosophical views concerning the correspondence principle. In the first formulations of it Bohr emphasizes multiple times that it was mainly a heuristic tool, but we can see in his later work that Bohr takes the generalization to classical mechanics to be rational. This insistence on the 149 Bohr, Niels, Philosophy of Science Vol. 37 (1934), p. 157, and in The Truth of Science : Physical Theories and Reality (1997) by Roger Gerhard Newton, p. 176 150 R. Courant, ‘Fifty years of Friendship, in Rozental, Niels Bohr, p. 304’ 151 Heisenberg, Physics and Beyond, p. 210 152 Dingle, Herbert. "Atomic Theory and the, Description of Nature 1: Four Essays, with an Introductory Survey." Nature 133 (1934): 962-964. P. 92. 153 Bohr 1925, reprinted in Kragh 2012, p.218. 45 rationality of the correspondence principle was also quite different from how it was perceived by most physicists, who took it at best as a “clever bricolge”154. In his later writings however we continue to see this pursuit to a consistent whole theory that incorporates both classical and quantum concepts. Bohr begins to see that classical theories and quantum theory are complementary in the sense that they fill the holes in the foundations of both theories, despite the inherent contradiction of both theories. There is certain ‘circularity’ in the connection between the two theories, as the classical theory provides the fundamentals of the quantum theory, with help of the correspondence principle, and the quantum theory will then, within certain limits, give the same solution as the classical theory. Bohr sensed wholeness in the two theories that could not exist independent of each other. One was the generalization of the other: “At the same time, it affords some indication of how we may perceive in the quantum theory, in spite of the fundamental character of this departure, a natural generalization of the fundamental concepts of the classical electrodynamic theory.”155 Bohr was listed as the first member of the advisory committee of the unity of science movement. A movement that tried to promote the idea that every science is in a way linked to the other, however it must be said that Bohr only contributed a one-page paper to the movement. It is certain though that Bohr did believe, to some extent, in the unity of nature and always emphasized the relationships to be found in different theories. 3.4 Bohr’s correlation to Høffding Before beginning on the similarities between Bohr’s and Høffding’s philosophy, I first want to clarify their relationship, as some state that Bohr was, throughout his carrier, extensively influenced by Høffding’s reasonings and through him by other philosophers such as Soren Kierkegaard and William James. Høffding was one of Bohr’s professors during his time at the Copenhagen University and also an old friend of the Bohr family. He wrote two, at that time, renowned books called ‘Outlines of Psychology’ and ‘A History of modern Philosophy’. In his expression of his philosophies Kant and Kierkegaard primarily influenced him, among others. Although Bohr is assumed to have attended Høffding lectures in his early scholar carrier, after this initial relationship almost all contact ceased. Only a few letters from the correspondence between him and Høffding were found, most being about matters outside of philosophy or physics. We can state that Høffding was important for the forming of Bohr’s general stand in philosophy, but he did not aid to the specific formulations of Bohr’s theories on philosophy.156 One of the main ideas of Høffding was that one cannot regard an individual on its own. There will always remain or exist a context that directly influences the individual and should therefore be taken into account when regarding the individual or in Høffding’s own words: “Research may and must admit that, as regards the individual, it does not succeed in giving every detail, that there is always something which escapes it, -that the individuality appears in consequence an irrational whole, which admits of only approximate determination”. This emphasis on context is also found in the works of Bohr. The idea that there is no such thing as a free mind and unspoiled context is one of the main debate 154 Darrigol uses this phrase to describe the physics community’s perception of Bohr’s old quantum theory. Bohr, Niels. "The structure of the atom." Nobel lecture 11 (1922): 14. 156 Favrholdt, D., ed. Complementarity Beyond Physics (1928-1962). Vol. 10. Elsevier, 2013. 155 46 questions in the more philosophical papers of Bohr. However a difference must be pointed out: Høffding only applied this context based reasoning to problems within psychology, biology and philosophy, but he never extended it to chemistry or physics. So although the reasonings are similar the, area in which they are applied are different. Bohr believed that all definitions pre-assumed continuity, for both ordinary language and quantum theory. In the case of ordinary language words in isolation naturally contain a form of ambiguity. They need context to acquire their precise meaning. Various meanings of the same word can only be compared if there is a continuous connection between the various contexts in which the word plays a part. For Bohr’s theories the atomic model also contains a certain form of ambiguity as it can be found in different stationary states; the various states can only be compared if there is a continuous deformation of the system connecting the two states. Bohr saw this condition as an essential part of the foundations of his theories, but the condition could not be fulfilled in the later stages of his atomic model. On this problem he wrote to Høffding; “In my personal opinion these difficulties [surrounding the atom model] are of such nature that they hardly allow us to hope that we shall be able, in the world of the atom, to carry through a description in space and time of a kind which corresponds to our ordinary sensory images. Under these circumstances one must naturally constantly bear in mind that one is operating with analogies, and this step, in which the application of these analogies is delimited in each case, is of decisive significance for progress ”157. Bohr claims that we cannot expect the concepts used to describe physics in the observable physical world to also apply in the world of the atom, therefore all measurements and theories done on atoms are idealizations of nature. We cannot expect terms such as momentum and time, which are defined through our sensory experiences, to apply in the same manner in the quantum theory. Bohr did discuss some of his overthinking with Høffding, but it is not quite clear how much Høffding’s work influenced him, but Bohr’s context related ideas did show quite some similarities to Høffding. These reasonings also have quite a Kantian feeling, as we shall see below. 3.5 The transcendental part of Bohr’s philosophy I get to think about my own thoughts of the situation in which I find myself. I even think that I think of it, and divide myself into an infinite regressive sequence of ‘I’s who consider each other. I do not know at which ‘I’ to stop as the actual, and in the moment I stop at one, there is indeed again an ‘I’ which stops at it. I become confused and feel dizziness as if I were looking down into a bottomless abyss, and my ponderings result finally in a terrible headache.158 One of the first philosophical problems that Bohr countered was the problem of context and how it affected objectivity within experiments; solutions are not universal, but only accountable in a specific frame. He already mentions his overthinking in a letter from 1910 written to his brother: “sensations, like cognitions, must be arranged in planes that cannot 157 Bohr to Høffding 22 September 1922. Kragh 2012 p. 353. Adventures of a Danish student by Poul Martin Møller, one of the favorite books of Bohr and often quoted by him during lectures and in papers. 158 47 be compared”159. Later this slight remark on the planes of objectivity would be worked out in full-fledged papers. Bohr assumed that full objectivity, understood as not being influenced by prejudice, independent of thought and part of reality, was impossible within preforming and describing experiments. He did not think it was a problem of the measurement apparatus, as can be observed clearly in the following statement: “The very fact that quantum phenomena cannot be analyzed on classical lines thus implies the impossibility of separating a behavior of atomic objects from the interaction of these objects with the measuring instruments which serve to specify the conditions under which the phenomena appear.”160 It was not the experiment that was the problem. The problem is that we cannot find an independent reality that is free of any context. In the quantum theoretical case, quantum concepts and theory will always remain, to a certain degree, dependent on the classically described context, which places limitations on our objectivity. Both Kant and Bohr were interested in exploring these limits of objectivity. Kant was one of the first philosophers that explicitly stated the dependency of knowledge on the knowledge seeker: “Thus far it has been assumed that all our cognition must conform to objects. Let us try to find out by experiment whether we shall not make better progress, if we assume that objects must conform to our cognition.”161 According to Kant we can state that the form of objects, as seen by the observer, is predetermined by a set of cognitive conditions from said observer. Objectivity according to Kant is therefore an intersubjective agreement between multiple observers and not a direct reflection of a ‘real’ truth. We recognize some of Bohr’s thinking in these statements, as Bohr said: “The very fact that quantum phenomena cannot be analyzed on classical lines thus implies the impossibility of separating a behavior of atomic objects from the interaction of these objects with the measuring instruments which serve to specify the conditions under which the phenomena appear.”162 According to Bohr there is a limit to our objectivity in theorizing atomic physical processes, because we cannot measure them in a definite descriptive quantum context. Only with the help of our experimental tools can we make certain assumptions about quantum processes and these experimental tools have to be described in classical terms. This is not to say that they cannot be described according to the quantum rules, but, in the end, to describe the phenomena classical terms have to be used. Bohr stated; “… In spite of their limitations, we can by no means dispense with those forms of perception which color our whole language and in terms of which all experience must ultimately be expressed”163. So it was not an ontological problem that gave rise to the need of classical terms, but an epistemological problem, because our whole framework of thought is colored by the classical context. In classical physics we can at the end of our experiment subtract the influence of the measuring instruments, but this cannot be done in quantum theory, as said measuring 159 Bohr, N. "Collected Works V. 1. Early Work (1905-1911)/Ed. J. R Nielsen." (1972) P. 96. Niels Bohr, ‘On the notions of causality and complementarity’, dialectica 2 (1948): 312-319. 161 Pluhar, Werner, and I. Kant. "Critique of pure reason." (1996): A820B848, p.21. 162 Bohr, Niels. ‘On the notions of causality and complementarity’, dialectica 2 (1948): 312-319. 163 Bohr, Niels. "The quantum of action and the description of nature." Bohr (1934)(1929): 92-101. 160 48 instruments are described in classical terms. Even though describing the measuring instrument in classical terms forms an ambiguity in the description of quantum processes, Bohr stubbornly held on to the use of classical concepts, even within the new quantum theory. He states that: “... it would be misconception to believe that the difficulties of the atomic theory may be evaded by eventually replacing the concepts of classical physics by new conceptual forms... It continues to be the application of these concepts alone that makes it possible to relate the symbolism of quantum theory to the data of experience.”164 Bohr stated that even if we do measurements on non-classical objects ‘the account of all evidence must be expressed in classical terms’165. As noted earlier, Bohr considered this an epistemological problem. In a recent essay on Bohr’s philosophy, Henry Folse noticed this distinct trait of Bohr’s epistemology: “his constant concern with the problem of the applicability of individual concepts to the description of phenomena and his deep conviction that one of the principal tasks of science was to develop as it went along ‘a conceptual framework adequate for such a description”166. For Bohr, in the physical world, this conceptual framework has to be described using classical concepts, as our ordinary language is full of the classical notions. He deems it impossible to be completely objective; in the sense that the context is already predetermined by our use of language. According to Kant it is impossible to subtract the contribution of the knower from what is known; ‘pure’ objectivity does not exist. He stated: “Whatever [characteristics] we are acquainted with in matter are nothing but relations (what we call its intrinsic determinations is intrinsic only comparatively); among these relations there are independent and permanent ones, through which a determinate object is given to us”167. Bohr also noticed this “Impossibility of a strict separation of phenomena and means of observation”168. We can call this consideration the ‘relational status of attributes’. The knower is not known in the act of knowing is Kant’s reasoning, which tells us that there are certain preconditions of experience that shape the experience without being part of it. Bohr used the same kind of reasoning in stating that the instrumental preconditions of a quantum description cannot be described quantum-mechanically. According to Bohr when doing an experiment the measuring object is part of the knower and not of the known. This is not to say that the measuring object has in sense different intrinsic properties, but because of the way we epistemologically use the measuring objects classically, we have to describe them classically. According to Bohr there has to be intersubjective agreement about the result, which means that we must use the same language to describe the properties. That way there is only a practical boundary, not an absolute boundary, between items described classically and objects described quantum mechanically. Bohr stated this as the “impossibility of any sharp separation between the behavior of atomic objects and the interaction with the measuring instruments which serve to define the very conditions under 164 Idem, p.16 Bohr, Niels. Quantum physics and philosophy: causality and complementarity, p.39. 166 Folse, Henry J. "The philosophy of Niels Bohr: The framework of complementarity." (1985). 167 Kant, Immanuel. "Critique of Pure Reason, 1781." Translated by Norman Kemp Smith,. 9291nallimcaM: nodnoL (1908). 168 Bohr, Niels. "The quantum of action and the description of nature." Bohr (1934)(1929): 92-101. 165 49 which the phenomena appear”169. This condition of separability was quite vague, as it was not fully clear how one should choose where to place the separation. Bohr explained that the separation was bendable depending on the context. Bohr stated in his paper Atomic Theory and the Description of Nature: “Ultimately every observation can, of course, be reduced to our sense perceptions. The circumstance, however, that in interpreting observations use has always to be made of theoretical notions entails that for every particular case it is a question of convenience at which point the concept of observation involving the quantum postulate with its inherent irrationality is brought in.” In order to make an observation to a quantum mechanical system one has to permit a certain interaction with the surroundings, which are unfortunately described by classical theories. If a description of the quantum system is made with these difficulties in mind, one allows for a certain ‘idealization of observation and definition’ according to Bohr. “The relation between subject and object”, as said by Bohr, “forms the core of the problem of knowledge”170 and the reason for the irrationality. Strawson described this problem as: “The investigation of that limiting framework of ideas and principles, the use and application of which are essential to empirical knowledge, and which are implicit in any coherent conception of experience which we can form”171. According to Bohr there was no such thing as an independent reality, as it would, in the physical world, always be dependent on the agencies of observation. The problem with quantum mechanics was that these agencies were classically described while the measured objects contained quantum elements. With this separation of measured objects and measuring devices, one has to keep in mind that this vaguely defined separation was able to shift. In order to do any experiments on a quantum system we must allow for an interaction between the two separated systems. The systems being the object and measuring instruments, where one is described in quantum terms and the other in classical terms. This concern with the necessary conditions in accumulating knowledge and giving an unambiguous report about the foundings can be described as transcendental. Honner described the term transcendental as ‘signifying a fundamental concern with the necessary conditions for the possibility of (experiential) knowledge’ According to Honner it is a word that can “describe the character of Bohr’s philosophy […] The term ‘transcendental’ is a key: and understanding of Bohr lies behind the door that unlocks it”172. This transcendentalthought was one of the doctrines founded by Kant. If we consider the transcendental part of Bohr’s philosophy, we can understand his stress on the need for classical concepts in the quantum theory better. Someone who also made this observation was Weizsäcker, who said: “Niels Bohr is the only physicist in our time who-as far as I know, without having been influenced by Kant-proceeded from fundamental insights similar to Kant’s … I join in the conjecture that these principles, to put it in Kant’s terminology, will be neither transcendent nor empirical, but transcendental in nature. In other words, they will formulate neither metaphysical hypotheses nor particular experiences, but merely the preconditions of the possibility of experience as such. Only in this framework will physicists be able to do justice 169 Bohr, Niels. "Discussion with Einstein on epistemological problems in atomic physics." Quantum theory and measurement (1949): 1, p.210. 170 Niels, Bohr, Atomic Theory and the Description of Nature (Cambridge: Cambridge University Press, 1934), p.117. 171 Strawson, Peter Frederick. The Bounds of Sense: An Essays on Kant's Critique of Pure Reason. Methuen, 1966. 172 Honner, John. "The transcendental philosophy of Niels Bohr." Studies in History and Philosophy of Science Part A 13.1 (1982): 1-29. 50 to Bohr’s doctrine of the indispensability of classical concepts.”173. If one chooses to do a measurement on a quantum system there will always be preconditions of the context in which it is applied and both Bohr and Kant have made the important point on unambiguous communication in the language. There are, however, also differences to be spotted in the work of Bohr and Kant and I would not like to claim that Bohr is considerably Kantian in nature. For example an important difference noted by Henry Folse: “this (Bohr’s philosophy) … has a certain Kantlike appearance, for it issues in statements about the logical requirements for the proper use of concepts in experience. But such an appearance is revealed to be mistaken when we recall that these claims have nothing to do with how experience is obtained, as the Kantian statements concerning the proper use of concepts imply, but only has to do with the communication of an observation or an experience once that experience has already come to pass. In this sense, Bohr’s reference to the use of concepts is tellingly non Kantian.”174 I fully agree with the point being made here and I do not have the intention to push Bohr over to Kant’s side, but I am of the opinion that there is a similar style to their philosophies. Bohr is not explicitly Kantian, but there are several features of Bohr’s philosophy of physics that are very isomorphic to Kant’s philosophy of knowledge. Their general framework of thought shows quite some similarities and I hope that with the comparison of these two great minds, we came to understand the work of Bohr better. 3.5 The position of Bohr within philosophy There are two general themes that are repeated over and over again in the philosophical considerations of Bohr, which we have covered in this paper. First there is the search of relationships between theories. A new theory does not replace the other, but is a new revised version of the old one, where the old theory is true in a limited context. Secondly there is the focus on concepts, and then especially the use of unambiguous language. There must be a complete awareness of the presumptions that are made when doing experimental research and when describing the discovered data from the researches. This awareness is crucial for a clear communication in physics. Bohr strongly emphasized this need for unambiguous communication within physics, even at the boundaries of human experience. The quantum realm was one of these boundaries, as we cannot directly interact with the system. Only with the help of measuring mechanisms can we become more knowledgeable on the quantum systems, but these can only be described in classical terms. Even quantum systems are in the end ‘tied to the concepts-or, much more, the wordswhich are used in our ordinary description of nature’175. He believed that classical concepts were indispensable for this description of nature, because these concepts have adapted to the way humans describe the world. If we want to provide logical rules in common language 173 Weizsäcker in E. W. Bastin (ed.), Quantum Theory and Beyond (Cambridge, Cambridge University Press, 1971), p. 372. 174 Folse, H. J. (1985). The philosophy of Niels Bohr: The framework of complementarity. 175 Letter to Einstein, 13 April 1927, BSC:10, reprinted in Honner 1982. 51 to all men, we must use these concepts. We need these conceptual representations, for else we would not be able to understand how we should perceive what happens on quantum level. From his elaborate philosophical reasoning within quantum theory, Bohr realized that we could never be only the spectators, seeing how context always has an effect on the view of the experiment or as Bohr said: 'We are both onlookers and actors in the great drama of existence: this is 'the old truth' of which 'the new situation in physics... has so forcibly reminded us'176. Pauli once wrote to Bohr to congratulate him on having ‘omitted all physics’ and that he could now fully focus on philosophy. I do not think, however, that Bohr viewed it as his task to solve general philosophical problems. In most of his work he focused primarily on quantum and atomic problems and from there extended them to wider subjects, such as biology and psychology. I consider, although not everyone credits him in this name, Bohr one of the major philosophers of science of the twentieth century. He has influenced his peers in physics and also other sciences to rethink their conduct in science and look beyond the context presented by the framework of instruments. If we want to describe Bohr as a philosopher it is best to do so in his own words: “The task of philosophy may be characterized as the development of conceptual means appropriate for communication of human experience”177. In this quote Bohr gives an overview of what is to be the relationship between philosophy and science, but it is also a good self-assessment and description of the position Bohr holds within philosophy. Conclusion While doing the research on the correspondence principle of Bohr it can be noticed that it is called one of the most important theories in the old quantum theory. It was named the ‘cornerstone of Bohr's philosophical interpretation of quantum mechanics’ by A. Bokulich. The fact that this cp is so well-known is quite interesting, as it is largely an ambiguous principle. How is it that such an indefinable principle became one of the ‘cornerstones’ of the quantum theory? The quantum theory is said to be primarily formal and set on clear quantum rules, even though the correspondence principle represents a grey area in the theory, were also some flexibility is allowed. In chapter three, we saw that Bohr usually considered quantum problems to be connected with the physical world. He not only desired to describe the quantum world according to mathematical formulas, but also wanted to describe a further reaching connection to the physical world. From his aspirations towards physics it is clear why he deemed the correspondence principle indispensable to the quantum theory, as it was the only remaining link with the classical theories, which color our language and perspective of the world. So it is epistemologically needed, in the sense that we cannot describe quantum effects without the use of classical concepts, because 176 177 Bohr, Niels. "The quantum of action and the description of nature." Bohr (1934)(1929): 92-101. Note, 4 January 1958, reprinted in Honner 1982. 52 classical concepts are the only ones we can really comprehend in a world classically described. The correspondence principle was also deemed indispensable and used extensively by other physicists, even though not all physicists comprehended it entirely. Why did the physics community accept and aspire to use a principle that was considered ambiguous? It could have been that other physicists were impressed by the explanations Bohr could give with help of the correspondence principle for the found experimental data done on atomic systems. They therefore also tried to apply and understand it. A fault in this argument was, however, as we saw, that the correspondence principle also extended to the new quantum theory, which actually explained these experimental data without direct need for the correspondence principle. In the final stages of the correspondence principle, it could be argued that it was actually dispensable. The correspondence principle gave a bridge between the new quantum theory and the classical theory and was applied according to the old quantum rules, but in the new theory correspondence was already included in the theory itself. Or as Born, Heisenberg and Jordan said in their famous Dreimännerarbeit: “This similarity of the new theory with the classical theory also precludes any question of an independent correspondence principle in addition to the theory; rather the theory itself can be regarded as an exact formulation of Bohr’s correspondence ideas [Korrespondenzgedanken].”178 So as the correspondence principle lost direct application with the change of the quantum theory, only the concept remained. The term correspondence principle is still used though, in today’s literature for example, Joseph Ford and Giorgio Mantica in their article, “Does Quantum Mechanics Obey the Correspondence Principle? Is It Complete?” quote the correspondence principle and state it to be understood as; “any two valid physical theories which have an overlap in their domains of validity must, to relevant accuracy, yield the same predictions for physical observations”179. It could be deemed that only the general concept of correspondence between theories is the one that is debated in today’s literature. The correspondence principle as first articulated in 1913 did not survive the transition from the old quantum theory to the new one, but the general concept of correspondence between classical mechanics and the new quantum theory did survive. So although the correspondence principle is still a subject of today’s literature, we can state that it is different from the original version. The correspondence principle and its formulation changed according to the context it is used in and it is therefore a good reflection of the general consensus, in a certain time period, on how the atomic theory should be perceived. We saw that with the fall of the old atomic model the correspondence principle remained as a bridge between the classical and 178 Max Born, Werner Heisenberg, and Pascual Jordan. Zur Quantenmechanik II. Zeitschrift für Physik, 35(89):557–615, 1926. English translation in [46], pp. 321–385. 179 Ford, Joseph, and Giorgio Mantica. "Does quantum mechanics obey the correspondence principle? Is it complete?." American journal of physics 60.12 (1992): 1086-1098. 53 quantum theory, but the direct application became lost. If we follow the correspondence principle, we get a good notion of the changes the quantum theory went through in the course of time. The correspondence principle gives a good overview of Bohr’s general thinking and shows beautifully the changes to his concept of quantum physics and the atom theories throughout the years. I do believe that if anyone wants to understand Bohr theory of physics, one must try to begin with understanding the correspondence principle, as it was the link that Bohr saw between the classical mechanics, the old quantum theory and new quantum mechanics. Bibliography I. II. III. IV. V. VI. VII. VIII. IX. X. XI. XII. XIII. XIV. XV. XVI. XVII. Petersen, Quantum Physics and the Philosophical Tradition, MIT Press, Cambridge, 1968. Beller, Mara. "Jocular Commemorations: The Copenhagen Spirit." Osiris(1999) Bohr 1918, “The quantum theory of line-spectra”, Det kongelige Danske videnskabernes Selskab, Matematiske-fysike Meddelser, 4(1): 1–36; reprinted in Bohr (1976), pp. 67–102 Bohr 1920, “On the series spectra of the elements”, Lecture before the German Physical Society in Berlin (27 April 1920), translated by A. D. Udden, in Bohr (1976), 241–282. Bohr 1920, "Über die Serienspektra der Elemente." Zeitschrift für Physik A Hadrons and Nuclei 2.5. 1 Bohr 1921a, “Application of the quantum theory to atomic problems in general”, unpublished manuscript in Bohr (1976) Bohr 1922, Seven lectures on the theory of atomic structure”, unpublished, in Bohr (1977), pp. 341– 419. Bohr [1923] 1924, “On the application of the quantum theory to atomic structure,” Proceedings of the Cambridge Philosophical Society (supplement), Cambridge: Cambridge University Press, pp. 1–42. Bohr 1925, “Atomic theory and mechanics,” Nature (Supplement), 116: 845–852. Reprinted in Bohr (1984), pp. 273–280. Bohr 1931, reprinted in Greenberger, Daniel, Wolfgang L. Reiter, and Anton Zeilinger, eds. Epistemological and experimental perspectives on quantum physics. Vol. 7. Springer, 1999. Bohr 1934, Philosophy of Science Vol. 37, p. 157, and in The Truth of Science : Physical Theories and Reality (1997) by Roger Gerhard Newton, p. 176. Bohr 1949, "Discussion with Einstein on epistemological problems in atomic physics." Quantum theory and measurement. Bohr, Niels 1958b, pp. 6-7, "Essays 1958-1962 on atomic physics and human knowledge." (1963). Bohr 1996, pp. 511-2, reprinted in Schlosshauer, Maximilian, and Kristian Camilleri. "The quantum-toclassical transition: Bohr's doctrine of classical concepts, emergent classicality, and decoherence." (2008). Bokulich, Alisa, "Bohr's Correspondence Principle", The Stanford Encyclopedia of Philosophy (Spring 2014 Edition), Edward N. Zalta (ed.). Bokulich, Alisa. "Bohr's correspondence principle." (2010). Bokulich, Alisa. "Three Puzzles about Bohr's Correspondence Principle." (2009). 54 XVIII. XIX. XX. XXI. XXII. XXIII. XXIV. XXV. XXVI. XXVII. XXVIII. XXIX. XXX. XXXI. XXXII. XXXIII. XXXIV. XXXV. XXXVI. XXXVII. XXXVIII. XXXIX. XL. XLI. XLII. XLIII. XLIV. XLV. XLVI. XLVII. XLVIII. Darrigol, Olivier. From c-numbers to q-numbers: the classical analogy in the history of quantum theory. University of California Press, 1992. De Broglie, Louis. Recherches sur la théorie des quanta. Diss. Migration-université en cours d'affectation, 1924. Dingle, Herbert. "Atomic Theory and the, Description of Nature 1: Four Essays, with an Introductory Survey." Nature 133 (1934): 962-964. Dirac, Paul AM. "The fundamental equations of quantum mechanics."Proceedings of the Royal Society of London. Series A, Containing Papers of a Mathematical and Physical Character (1925): 642-653. Favrholdt, D., ed. Complementarity Beyond Physics (1928-1962). Vol. 10. Elsevier, 2013. Faye, Jan, "Copenhagen Interpretation of Quantum Mechanics", The Stanford Encyclopedia of Philosophy (Fall 2014 Edition), Edward N. Zalta (ed.), Folse, Henry J. "The philosophy of Niels Bohr: The framework of complementarity." (1985). Ford, Joseph, and Giorgio Mantica. "Does quantum mechanics obey the correspondence principle? Is it complete?." American journal of physics 60.12 (1992): 1086-1098. Heisenberg, W. "The Physical Principles of the Quantum Theory (1930)."Scientific Review Papers, Talks, and Books Wissenschaftliche Übersichtsartikel, Vorträge und Bücher (2013): 118., p. 83 Heisenberg, Werner. "The physical principles of quantum mechanics." U. Chicago Press, Chicago (1930): 21. Hesse, Mary. Transcendental Arguments and Science. Springer Netherlands, 1979. 159-170. Honner, John. "The transcendental philosophy of Niels Bohr." Studies in History and Philosophy of Science Part A 13.1 (1982), p 21. Jammer, Max. The conceptual development of quantum mechanics. Tomash, 1989. Kant, Immanuel. "Critique of Pure Reason, 1781." Translated by Norman Kemp Smith, (1908). Kragh, Helge. "Bohr—Kramers—Slater Theory." Compendium of Quantum Physics. Springer Berlin Heidelberg, 2009.. Kragh, Helge. Niels Bohr and the Quantum Atom: the Bohr model of atomic structure 1913-1925. Oxford University Press, 2012. Kramers, Hendrik A. "Über den Einfluß eines elektrischen Feldes auf die Feinstruktur der Wasserstofflinien." Zeitschrift für Physik A Hadrons and Nuclei (1920). L. Rosenfeld ‘Biographical sketch’, reprinted in Honner, John. "The transcendental philosophy of Niels Bohr." Studies in History and Philosophy of Science Part A 13.1 (1982): 1-29. MacKinnon, Edward. "Niels Bohr on the Unity of Science." PSA: Proceedings of the Biennial Meeting of the Philosophy of Science Association. Philosophy of Science Association, 1980. Margenau, Henry. "The nature of physical reality: A philosophy of modern physics." (1950). Nielsen, J. Rud, ed. The correspondence principle (1918-1923). Elsevier, 2013. Nobel Prize in Physics 1922 - Presentation Speech. Nobelprize.org. Nobel Media AB 2014. Web. 21 May 2015. <http://www.nobelprize.org/nobel_prizes/physics/laureates/1922/press.html> Nordström to Bohr, 2 February 1922, reprinted in Kragh 2012, p. 299. Pauli, Wolfgang. "Remarks on the History of the Exclusion Principle." Science103.2669 (1946). Pluhar, Werner, and I. Kant. "Critique of pure reason." (1996): A820B848, p.21. Post, Heinz R. "Correspondence, invariance and heuristics: in praise of conservative induction." Studies in History and Philosophy of Science Part A 2.3 (1971). Rynasiewicz, Robert. "The Correspondence Principle." (2013). Sommerfeld, A. "Atombau und Spektrallinien. Friedrich Vieweg und Sohn, Braunschweig (1919)." English see Atomic Structure and Spectral Lines, translated from the third German edition by HL Brose, Methuen (1923). Stachel, John. "Bohr and the Photon." Quantum Reality, Relativistic Causality, and Closing the Epistemic Circle. Springer Netherlands, 2009. Stolzenburg, Klaus, ed. The Emergence of Quantum Mechanics (Mainly 1924-1926). Elsevier, 2013. Strawson, Peter Frederick. The Bounds of Sense: An Essays on Kant's Critique of Pure Reason. Methuen, 1966. 55 56