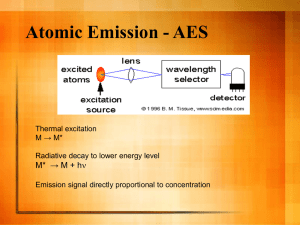
pmic7330-sup-0001-si
advertisement

SUPPORTING INFORMATION Combined use of irreversible binding and multiple reaction monitoring technology for low- and ultra-low copy-number protein detection and quantitation. Arthur T. Kopylov, Victor G. Zgoda, Andrew V. Lisitsa, Alexander I. Archakov Human blood 320 ml Human plasma 200 ml Concentration 61.5 mg/mL (BCA assay) Concentrating of human plasma 150 ml Concentration 80 mg/mL (BCA assay) Aliquotes of human plasma 10 tubes x 15 mL Total protein amount 12 g Depleting of human plasma on MARSTM column 2 parallel MARS (10 x 100 mm) systems 2 systems x 5 tubes x 60 runs = 300 runs Depleted human plasma I 10 tubes x 50 mL of depleted human plasma I = 500 mL Desalting / Concentrating on 5 KWCO spin columns 10 tubes x 4 mL of desalted depleted human plasma = 40 mL of depleted desalted concentrated human plasma II Dilution of human plasma II to the original volume of human plasma I 10 tubes x 4 mL of human plasma II 10 tubes x 50 ml of human plasma III Measurement of protein concentration in depleted desalted diluted human plasma III 10 tubes x 50 mL of depleted desalted diluted human plasma III = 500 mL of human plasma III Concentration 7.81 mg/mL Supporting Information Figure 1. General scheme of preparation and depleting human plasma on Agilent MARSTM Hu-14 multiaffinity removal system. Supporting Information Figure 2. Workflow of determination of LLOD in presence of depleted human plasma proteins without application of non-specific enrichment. Concentration of spiked CYP102 and BSA proteins in the dilutions is varied from 10 -9 M to 10-15 M. Total amount of on-column peptides loaded in each dilution after tryptic digestion is 10 µG in volume 10 µL Supporting Information Figure 3. Workflow of determination of LLOD of CYP102 and BSA proteins in presence of depleted human plasma proteins after application of non-specific enrichment on CNBr-activated sepharose 4B. Ten µL of concentrated sample were loaded onto the column to perform calibration in range of initial concentrations of targeted proteins, CYP102 and BSA, from 10 -18 M to 10-12 M. Supporting Information Table 1. CYP102 peptides and parameters used for MRM method development Peptide ID CYP102P1 CYP102P2 CYP102P3 Sequence Charge state GFAPQVATLDSHAGNLPR 617.65 3+ LWPTAPAFSLYAK 732.88 2+ SPHEDYLYQEELENAQSEGIITLHTAFSR 845.16 4+ 855.89 2+ 875.92 2+ CYP102P4 DFAGDGLFTSWTHEK CYP102P5 Precusor ion m/z QFVDWLDQASADEVK Fragment ion m/z Fragment ion type 851.454 788.419 739.894 731.381 675.863 626.328 556.323 525.948 426.231 382.714 1165.62 967.524 896.49 583.313 484.262 448.751 381.214 300.17 1005.42 994.022 937.468 932.496 718.356 581.301 567.248 888.428 724.865 514.259 506.184 413.215 391.152 334.132 276.155 962.449 847.419 719.362 659.287 561.292 490.245 472.223 357.201 Y8 Y152+ Y142+ Y14-NH32+ Y132+ Y122+ Y5 Y153+ Y82+ Y72+ Y11 Y9 Y8 Y112+ Y92+ Y82+ Y3 B2 B8 Y182+ Y172+ Y8 Y6 Y5 B92+ Y7 Y132+ Y4 B5 Y3 B4 B3 Y2 Y9 Y8 Y7 B5-NH3 Y5 Y4 B4-H2O Y3-H2O Collision energy, eV Retention time, min 29.50 21.7 23.05 34.7 27.32 40.1 27.72 16.33 31.50 31.5 Supporting Information Table 2. BSA peptides and parameters used for MRM method development Peptide ID Sequence Precusor ion m/z Charge state BSAP2 DAFLGSFLYEYSR 784.36 2+ BSAP3 LFTFHADICTLPDTEK 925.95 2+ BSAP6 LGEYGFQNALIVR 740.39 2+ BSAP7 TVMENFVAFVDK 700.38 2+ Fragment ion m/z Fragment ion type 1121.494 717.299 1234.578 1064.473 977.436 830.382 554.238 334.129 1342.631 323.6801 471.1967 814.3861 869.4127 1019.5089 795.8698 631.8197 1017.569 300.148 375.666 571.391 685.422 813.482 960.542 1180.635 571.382 500.348 1199.557 1068.517 825.435 939.474 579.3 508.267 520.248 361.195 Y9 Y5 Y10 Y8 Y7 Y6 Y4 B3 Y13 B52+ Y122+ B7-H20 Y152+ Y9 Y14 B112+ Y9 B3 A7-NH32+ Y5 Y6 Y7 Y8 Y10 Y5 Y4 Y10 Y9 Y7 Y8 Y5 Y4 B92+ Y3 Collision energy, eV Retention time, min 25.01 29.5 32.38 22.1 23.33 24.3 21.81 18 Supporting Information Figure 4. Base peak MS chromatograms of CYP102 (a) and BSA (b) peptides after digestion with trypsin. The signals were measured using a high resolution Q-TOF mass spectrometer (resolution 18200 at m/z 400, 4GHz mode) calibrated with an error of measurement of ±0.8 ppm. The peptides used for further analysis are indicated with arrows. Supporting Information Figure 5. Q-TOF MS/MS spectra of the precusor ions m/z 617.653+ (a) and m/z 875.922+ (c) of CYP102 and m/z 740.392+ (b) and m/z 784.362+ (d) of BSA with indicated fragment ions. The concentration of each protein made 0.05 μM and 1 μl of the sample was subjected onto the column Supporting Information Figure 6. MRM chromatograms of collision-induced dissociation products of CYP102 (m/z 617.65 3+675.82+ and m/z 875.922+962.51+) and BSA (m/z 740.392+685.41+ and m/z 784.362+ 1121.51+) peptide ions at 10-16 M concentration, corresponding to detection limit of the pure protein standards. Supporting Information Figure 7. MRM chromatograms of human plasma performed with method established for CYP102 and BSA. Most of the plasma protein peptides with a sort of resemble transitions are eluted between 12 and 18 minutes and no peptides corresponding to BSA or CYP102 were observed between 21 and 32 minutes. Supporting Information Figure 8. Dependence of the peaks areas of pure standards CYP102 (a) and BSA (b) after enrichment on CNBr-activated Sepharose 4B by non-specific irreversible binding from a sample volume of 10 ml. Detection limit for both the proteins was determinedto be 10 -18 M of original (before enrichment) concentration. The calibration curves plotted for transitions of two CYP102 peptides (precursor m/z 617.653+ solid line “a”/ m/z 875.922+ dotted line “a”) and two BSA peptides (precursor m/z 740.392+ solide line “b”/ m/z 784.362+ dotted line “b”) in five replicates. * * * * * * * Supporting Information Figure 9. Scan spectra obtained on QqQ (scan range m/z 500 - 1100) of synthetic peptides mixture CYP102 (QFVDWLDQASADEVK, Mr 1750.88 Da, m/z 876.452+; GFAPQVATLDSHAGNLPR, Mr 1851.05 Da, m/z 617.023+/926.532) and BSA (DAFLGSFLYEYSR, Mr 1567.72, m/z 784.862+; KVPQVSTPTLVEVSR, Mr 1639.91 Da, m/z 547.64+2/820.963+) recorded at direct infusion into the ion source at concentration 10-9 M (a), 10-10M (b), 10--11 M (c), 10-12 M (d), 10-13 M (e), 10-14 M (f) and blank infusion (g). *- The peptide KVPQVSTPTLVEVSR from bovin serum albumin was synthesized instead of the peptide LGEYGFQNALIVR with Mr 1479.7 Da. The reason is failed to synthesize the peptide LGEYGFQNALIVR due to problems with the peptide synthesis and purification. The replaced peptide, LGEYGFQNALIVR, from BSA is 84% identical to the peptide LGEYKFQNALLVR from HSA.