Clinical Trial Protocol Outline
advertisement

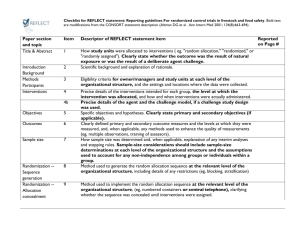
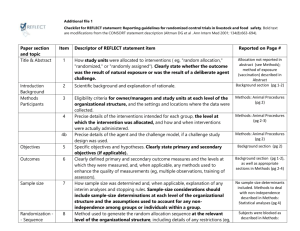
Clinical Trial Protocol Outline I. II. III. IV. V. Background/justification/rationale A. Description of disease being studied and problem being addressed B. Description of current therapy and any shortcomings C. Description of drug and its activity D. Description of any comparator drugs and justification for its use as the control E. Summaries of studies conducted to date Objectives A. Primary B. Secondary Study Design A. Single center or multicenter B. Phase C. Number and definition of arms D. Blinding E. Randomization if multiple arms F. Indication and patient population G. Sample size H. Duration and number of sites Study population A. Inclusion B. Exclusion Schedule of visits/assessments VI. A. Screening/eligibility determination B. Consent C. Enrollment/randomization D. Baseline evaluation E. Treatment schedule F. Follow-up schedule Ethical and Regulatory Considerations A. Regulatory document collection B. IRB review C. Informed consent administration VII. Study Procedures VIII. Clinical assessment IX. A. Special procedures (QOL, bronchoscopy, biopsy, PK, genetic sampling, etc.) B. Laboratory testing Study drug and comparator A. Drug shipment/receipt B. Drug packaging C. Drug storage D. Drug preparation E. Drug dispensing and accountability F. Drug administration G. Contraindications/concomitant medications H. Breaking the blind I. Drug destruction J. X. Adverse Experiences (AEs) A. Documentation of AEs B. Coding of AEs C. Serious Adverse Events D. XI. XII. Medication compliance assessment 1. Definition 2. Reporting Expected AEs/toxicities Subject Completion and Early Withdrawal A. Procedures for completion B. Procedures for early withdrawals C. Reasons for withdrawals Data Collection A. Method of data collection B. Procedures for data collection C. Data submission XIII. Study Monitoring/Audits XIV. Data Analysis A. Sample size determination B. Efficacy analyses C. Safety analyses D. Other analyses (if applicable) E. Interim analyses (if applicable) F. Data Safety Monitoring Board (if applicable) XV. Confidentiality XVI. Publication Policy XVII. References XVIII. Appendices A. Declaration of Helsinki B. Template informed consent form C. Toxicity criteria D. Health status measurement scale E. Laboratory sample preparation and shipment F. Standardized clinical procedure methodology G. Standardized questionnaire instructions H. Standardized measurement instructions