Reactions Review Answer Key Balancing the following reactions: Zn
advertisement

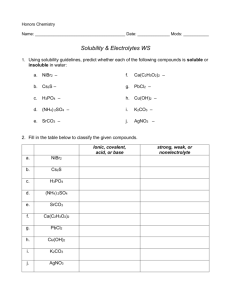
Reactions Review Answer Key Balancing the following reactions: 1. Zn(s) + 2HCl(aq) ZnCl2(aq) + H2(g) 2. 2NO2(g) N2O4(g) 3. ZnCl2+ 2AgNO3(aq) 2AgCl(aq) + Zn(NO3)2(aq) 4. 2KClO3(aq) 2KCl(aq) + 3O2(g) 5. MgCl2(aq) + 2HgNO3(aq) 2HgCl(s) + Mg(NO3)2(aq) Write the balanced chemical equation for each reaction described below. 6. Gaseous hydrogen reacts with gaseous oxygen to form water. 2H2(g) + O2(g) 2H2O(l) 7. Iron metal forms iron (III) oxide in air. 4Fe(s) + 3O2(g) 2Fe2O3(s) 8. A precipitate forms when an aqueous solution of silver(I) nitrate is mixed with hydrochloric acid (HCl) to make silver chloride and aqueous nitric acid (HNO3). AgNO3(aq) + HCl(aq) AgCl(s) + HNO3(aq) 9. Aluminum reacts with oxygen gas to form aluminum oxide. 4Al(s) + 3O2(g) 2Al2O3(s) Identify the types of reactions below: Types of reactions are: -synthesis -decomposition -single replacement double displacement -combustion 7. 4C5H9O(g) + 27O2(g) 20CO2(g) + 18H2O(l) combustion 8. 2NaBr(aq) + Ca(OH)2(aq) CaBr2(aq) + 2NaOH(aq) double displacement 9. 3Pb(s) + 2H3PO4(aq) 3H2(g) + Pb3(PO4)2(s) single replacement 10. Ag2O(s) Ag(s) + O2(g) decomposition 11. 2NH3(g) + H2SO4(aq) (NH4)2SO4(aq) synthesis For the double displacement reactions below, predict the products that will form, balance the equations, and identify the precipitate that form. 12. CuBr2(aq) + Na2CO3(aq) CuCO3(s) + 2NaBr(aq) 13. Na2SO4(aq) + Ca(NO3)2(aq) 2NaNO3(aq) + CaSO4(s) Write the ionic and net ionic equations for the reaction below. 14. 2Na3PO4(aq) + 3PbBr2(aq) 6NaBr(aq) + Pb3(PO4)2(s) Ionic: 6Na+(aq) + 2PO43-(aq) + 3Pb2+(aq) + 6Br-(aq) 6Na+(aq) +6Br-(aq) + Pb3(PO4)2(s) Net Ionic: 3Pb2+(aq) + 2PO43-(aq) Pb3(PO4)2(s) Chemical Reactions Review Balancing the following reactions: 1. Zn(s) + HCl(aq) ZnCl2(aq) + H2(g) 2. NO2(g) N2O4(g) 3. ZnCl2+ AgNO3(aq) AgCl(aq) + Zn(NO3)2(aq) 4. KClO3(aq) KCl(aq) + O2(g) 5. MgCl2(aq) + HgNO3(aq) HgCl(s) + Mg(NO3)2(aq) Write the balanced chemical equation for each reaction described below. 6. Gaseous hydrogen reacts with gaseous oxygen to form water. 7. Iron metal forms iron (III) oxide in air. 8. A precipitate forms when an aqueous solution of silver(I) nitrate is mixed with hydrochloric acid (HCl) to make silver chloride and aqueous nitric acid (HNO3). 9. Aluminum reacts with oxygen gas to form aluminum oxide. Identify the types of reactions below: Types of reactions are: -synthesis -decomposition -single replacement double displacement -combustion 7. 4C5H9O(g) + 27O2(g) 20CO2(g) + 18H2O(l) 8. 2NaBr(aq) + Ca(OH)2(aq) CaBr2(aq) + 2NaOH(aq) 9. 3Pb(s) + 2H3PO4(aq) 3H2(g) + Pb3(PO4)2(s) 10. Ag2O(s) Ag(s) + O2(g) 11. 2NH3(g) + H2SO4(aq) (NH4)2SO4(aq) For the double displacement reactions below, predict the products that will form, balance the equations, and identify the precipitate that form. 12. CuBr2(aq) + Na2CO3(aq) 13. Na2SO4(aq) + Ca(NO3)2(aq) Write the ionic and net ionic equations for the reaction below. 14. 2Na3PO4(aq) + 3PbBr2(aq) 6NaBr(aq) + Pb3(PO4)2(s) Ionic: Net Ionic: