CHEM2720-121912
advertisement

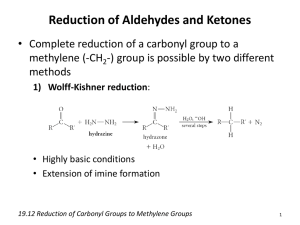
HIBBING COMMUNITY COLLEGE COURSE OUTLINE COURSE TITLE & NUMBER: Organic Chemistry 2: CHEM 2720 CREDITS: 5 (4 Lec/1 Lab) PREREQUISITES: CHEM 2710: Organic Chemistry 1 CATALOG DESCRIPTION: Organic Chemistry 2: CHEM 2720 is a continuation of Organic Chemistry 1. The chemistry of carbon compounds emphasizing the theories and mechanisms which account for their physical and chemical properties is again studied. Techniques of purification, separation, and synthesis are practiced in the laboratory. Organic Chemistry 2 is meant for students planning to major in biology, chemistry, chemical engineering, pharmacy, and certain medical fields. OUTLINE OF MAJOR CONTENT AREAS: I. II. III. IV. Conjugated unsaturated systems A. Allyic substitution and the allyl radical B. Alkadienes and polyunsaturated hydrocarbons C. The stability of conjugated dienes D. The Diels-Alder reaction Spectroscopic methods A. IR spectra B. NMR spectra Aromatic compounds A. Structure and properties of benzene B. Nomenclature C. Huckel's Rule D. Other aromatic compounds E. Electrophilic aromatic substitution reactions 1. Halogenation 2. Nitration 3. Sulfonation 4. Friedel-Crafts alkylations 5. Friedel-Crafts acylation Aldehydes and ketones A. Nomenclature B. Physical properties C. Synthesis of aldehydes 1. Oxidation of primary alcohols CHEM2720 1 Hibbing Community College, a technical & community college, is an equal opportunity educator & employer V. VI. 2. Reduction of acyl chlorides, esters, and nitriles 3. Ozonolysis of alkenes D. Synthesis of ketones 1. Oxidation of secondary alcohols 2. Friedel-Crafts acylation 3. Ozonolysis of alkenes 4. Reaction with lithium dialkylcuprates E. Reactions of aldehydes and ketones 1. Addition of organometallic compounds 2. Addition of hydride ion 3. Addition of hydrogen cyanide 4. Addition of ylides 5. Addition of alcohols 6. Addition of derivatives of ammonia 7. Aldol condensations Carboxylic acids A. Nomenclature B. Physical properties C. Synthesis 1. Oxidation of aldehydes and primary alcohols 2. Oxidation of alkenes 3. Oxidation of alkylbenzenes 4. Oxidation of methyl ketones 5. Carbonation of Grignard reagents 6. Hydrolysis of nitriles D. Reactions 1. Salt formation 2. Reduction 3. Conversion to acyl chlorides 4. Conversion to acid anhydrides 5. Conversion to esters 6. Conversion to lactones 7. Conversion to amides and imides 8. Conversion to lactams 9. α - halogenation 10. Decarboxylation E. Acyl chlorides F. Acid anhydrides Esters A. Nomenclature B. Physical properties C. Synthesis 1. Esterification 2. From acid chloride or anhydrides D. Reactions 1. Hydrolysis CHEM2720 2 Hibbing Community College, a technical & community college, is an equal opportunity educator & employer VII. VIII. IX. X. XI. 2. Transesterification 3. Conversion to amides 4. Reaction with Grignard reagents 5. Reduction Amides A. Nomenclature B. Physical properties C. Synthesis D. Reactions 1. Hydrolysis 2. Conversion to nitriles 3. Conversion to imides β - dicarbonyl compounds Amines A. Nomenclature B. Physical properties C. Synthesis 1. Gabriel synthesis 2. By Reduction of alkyl azides 3. By Amination of alkyl halides 4. By reduction of nitrorenes 5. By reductive amination 6. By reduction of amides, nitriles and ozimes 7. Through the Hofmann and Curtis rearrangements D. Reactions 1. As Bases 2. Diazotization 3. Conversion to sulfonamides 4. Conversion to amides 5. Hofmann and Cope eliminations Phenols A. Nomenclature B. Physical properties C. Synthesis 1. Via arenediazonium salts 2. Dow process 3. From sodium benzenesulfonates 4. Via cumene hydroperoxide D. Reactions 1. As acids 2. Williamson synthesis 3. Acylation 4. Electrophilic aromatic substitution Aryl halides A. Nomenclature B. Physical properties CHEM2720 3 Hibbing Community College, a technical & community college, is an equal opportunity educator & employer C. D. Synthesis 1. By electrophilic aromatic substitution 2. Via arenediazonium salts Reactions 1. Electrophilic aromatic substitution 2. Nucleophilc aromatic substitution COURSE GOALS/OBJECTIVES/OUTCOMES: The student will 1. recognize reactions that are enhanced by resonance stabilization of the intermediates. 2. predict the products of Diels-Alder reactions. 3. identify the key features of IR and NMR spectra. 4. describe the physical properties of aromatic compounds, aldehydes, ketones, carboxylic acids, esters, amides and amines. 5. describe synthesis routes for aromatic compounds, aldehydes, ketones, carboxylic acids, esters, amides, and amines. 6. describe the chemical reactions of aromatic compounds, aldehydes, ketones, carboxylic acids, esters, amides, and amines. 7. use the IUPAC system to name aromatic compounds, aldehydes, ketones, carboxylic acids, esters, amides, and amines. 8. obtain IR spectra of laboratory samples. 9. identify unknown organic compounds in the laboratory using basic chemical procedures. HCC COMPETENCIES MET: Thinking Creatively & Critically STUDENT ASSESSMENT SHALL TAKE PLACE USING INSTRUMENTS SELECTED/DEVELOPED BY THE COURSE INSTRUCTOR. SPECIAL INFORMATION: (SPECIAL FEES, DIRECTIVES ON HAZARDOUS MATERIALS, ETC.) Hazardous chemicals will routinely be used in laboratory. Students will be supplied with pertinent information relating to these materials at the appropriate time. AASC APPROVAL DATE: December 19, 2012 REVIEW DATE: December 2017 CHEM2720:so 121912 CHEM2720 4 Hibbing Community College, a technical & community college, is an equal opportunity educator & employer