Name: Variations in a Salty Ocean Background Anyone who ever
advertisement

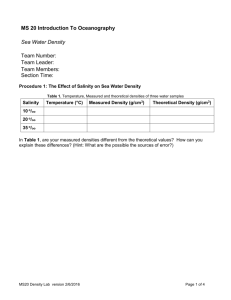
Name: _______________________ Variations in a Salty Ocean Background Anyone who ever tasted seawater knows that it is truly salty. The salty residue left on swimsuits after they’ve been in the ocean is further evidence of the ocean’s salinity. Salinity is a measure of how much salt is dissolved in water. The average salinity of seawater for all Earth’s oceans is about 35 grams of dissolved salts per kilogram of seawater. This is shown as 35 parts per thousand, or 35‰. The symbol ‰ is read "permill." It is similar to percent, but it refers to parts per thousand instead of parts per hundred. The Red Sea and the Mediterranean Sea contain some of the saltiest water on Earth. In some areas, these bodies of water show salinity measurements as high as 40‰. Objective In this investigation, you will examine the relationship between temperature and salinity in the oceans and how both of these qualities affect the density of seawater. Ultimately, you’ll estimate densities for water masses and use them to predict how water will mix in the oceans. Procedure Use the maps labeled “Temperature” and “Salinity” on mrsvanvliet.weebly.com to answer the following 3 questions 1. Describe some of the general patterns you observe for temperature and salinity ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ 2. Describe the location of at least three places in the world’s oceans that have high temperatures but low salinity ________________________________________________________________________ ________________________________________________________________________ ________________________________________________________________________ 3. What do you think might cause low salinity in these areas of warm ocean water? ________________________________________________________________________ ________________________________________________________________________ Name: _______________________ Background Both salinity and temperature affect the density of seawater. A liter of seawater has a higher mass than a liter of freshwater due to the added mass of the dissolved salts. As density measures mass per unit volume, saltwater has a higher density than freshwater. Increasing the amount of salt dissolved in water increases the water’s density. Temperature affects the density of seawater by changing the volume of the water itself. Higher temperatures actually cause water to expand, increasing the amount of space it takes up. The mass of warmer water is spread over a larger volume than a similar amount of cooler water, so the warmer water has a lower density. Increasing the temperature of water results in a decrease in the water’s density. The graph allows you to estimate the density of any mass of seawater based on its salinity and temperature. To estimate density, find the point where temperature and salinity values for the water intersect. The blue line on the graph nearest the point of intersection indicates the density of the water. Mixing in the oceans depends on the densities of water masses. When water masses of differing density meet, the mass with the higher density will sink to the bottom and the mass with the lower density will float on top of it. Procedure Use the maps labeled “0, 50W Temperature”, “0, 50W Salinity”, “20N, 90E Temperature” and “20N, 90E Salinity” to answer the next two questions. 4. Determine the temperature, salinity, and density of ocean water at the following locations. A. 0O, 50OW Temperature: ________________ Salinity: ________________ Density (use the density graph): _____________________ Name: _______________________ B. 20ON, 90OE Temperature: ________________ Salinity: ________________ Density (use the density graph): _____________________ 5. What do you think is responsible for the unusually low salinity of these warm waters? (If you need a hint, look at the land near these locations on a world map) ________________________________________________________________________ ________________________________________________________________________ Use the maps labeled “Mixing at the Panama Canal Temperature” and “Mixing at the Panama Canal Salinity” to answer the next two questions. 6. Determine the temperature, salinity, and the density of water on the Caribbean and the Pacific sides of the Panama Canal (The Panama Canal is at 10ON, 80OW) A. Caribbean Side Temperature: ________________ Salinity: ________________ Density (use the density graph): _____________________ B. Pacific Side Temperature: ________________ Salinity: ________________ Density (use the density graph): _____________________ 7. Predict what would happen to water that moves from the Caribbean Sea into the Pacific Ocean ________________________________________________________________________ ________________________________________________________________________ Use the maps labeled “Strait of Gibraltar Temperature” and “Strait of Gibraltar Salinity” to answer the next two questions. 8. Determine the temperature, salinity, and the density of water on the Mediterranean and the Atlantic sides of the Strait of Gibraltar (The Strait of Gibraltar is at 35 ON, 5OW) A. Mediterranean Side Temperature: ________________ Salinity: ________________ Density (use the density graph): _____________________ B. Atlantic Side Temperature: Salinity: ________________ ________________ Name: _______________________ Density (use the density graph): _____________________ 9. Predict what would happen to water that moves from the Mediterranean Sea into the Atlantic Ocean ________________________________________________________________________ ________________________________________________________________________