Quiz 5

Quiz 5 Key
1. Scientists believe they know the average global temperature over the last 160,000 years. Which property of the ice core samples from Antarctica provides this information?
A.
the thickness
B.
the carbon dioxide concentration
C.
the 1 H/ 2 H ratio
D.
the temperature
Think about which of these would be a component of rain and snow.
2. Although carbon dioxide is the primary compound discussed with respect to global warming; of the common atmospheric components the global warming potential (GWP) of a carbon dioxide molecule is very low. Which statement best explains this pair of facts?
A.
CO
2
molecules are highly symmetrical.
B.
CO
2
has a low molar mass.
C.
CO
2
exists in relatively high concentrations.
D.
CO
2
is formed in the combustion of fossil fuels.
Remember that it is both concentration and GWP which determine what level of warming might come from any one gas.
3. This is the Lewis structure for acetic acid. The bond angles are not drawn to scale.
Based on the electronic configuration shown in the Lewis structure, predict the O C O bond angle, in degrees
.
A.
90
B.
109.5
C.
120
D.
180
Remember that the molecular geometry dictates the bond angles.
4. Scientists have correlated average global temperature with atmospheric carbon dioxide levels for the last
160,000 years using ice cores from Antarctica. How is the carbon dioxide level determined?
A.
by measuring the CO
2
concentration in trapped air bubbles in the ice layers
B.
by measuring the 18 O/ 16 O ratio in the CO
2
molecules
C.
by extrapolating the CO
2
level from the temperature of the ice
D.
by extrapolating the CO
2
level from the average global temperature
Remember that atmospheric gases are trapped in the ice layers.
5. Which practices might be described as carbon sequestration?
I. Planting trees
II. Deforestation
III. Pumping carbon dioxide deep into the ocean
IV. Increasing combustion of fossil fuels
A.
I and II only
B.
II and III only
C.
III and IV only
D.
I and III only
Remember that sequestration means trapping the CO
2
where it cannot enter the atmosphere.
6. Which explanation accounts for the fact that, on average, cloudy nights are warmer than clear nights?
A.
Clouds reflect light incoming from the moon.
B.
H
2
O does not absorb infrared energy.
C.
Clouds are composed of water vapor and H
2
O is a strong greenhouse gas.
D.
Clouds have a high concentration of carbon dioxide from fossil fuel combustion.
Think about what clouds are made of and how that interacts with IR radiation.
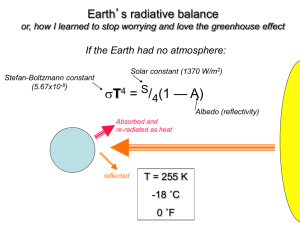
7. The greenhouse effect refers to the process by which
A.
atmospheric oxygen and nitrogen absorb heat radiated by Earth.
B.
atmospheric nitrogen absorbs ultraviolet radiation radiated by Earth
C.
atmospheric gases trap and return infrared radiation radiated by Earth.
D.
greenhouse gases absorb ultraviolet radiation from the Sun, warming the molecules.
Remember that it is the warmth of the earth that is being trapped.
8. What is the coefficient for oxygen in the balanced chemical equation for the combustion of heptane (C
7
H
16
)?
Unbalanced equation: C
7
H
16
+ O
2
CO
2
+ H
2
O
A.
11
B.
8
C.
7
D.
1
Remember to balance CO
2
and H
2
O before the O
2
.
9.
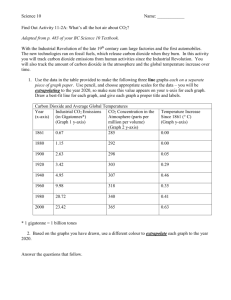
. The top graph shows the mean temperature over the past 160,000 years as determined from ice-core data.
Which of graphs A through D best represents the carbon dioxide concentration in air over the same time period as measured from air bubbles trapped in the ice cores?
A.
Graph A
B.
Graph B
C.
Graph C
D.
Graph D
Remember that temperature and CO
2
concentrations follow the same trends.
10. Which vibrational mode for carbon dioxide is not expected to contribute to the greenhouse effect?
A.
B.
C.
D.
Remember that vibrations that cancel each other out do not contribute to warming.
11. The most abundant greenhouse gas in Earth's atmosphere is:
A.
CO
2
B.
H
2
O vapor
C.
N
2
D.
CH
4
Remember that the earth is ~70% water.
12. What is the mass of exactly 500 sulfur atoms?
A.
1.61 10 4 g
B.
32.1 g
C.
2.66 10¯ 20 g
D.
5.33 10¯ 23 g
Remember that one mole is 6.02 x 10 23 items and one sulfur has a molar mass of 32 g/mole.
13. Tropospheric ozone (O
3
) is a greenhouse gas. Why is O
3 not assigned a Global Warming Potential (GWP)?
A.
Ozone molecules absorb only low energy infrared frequencies.
B.
Ozone molecules have a low molecular weight.
C.
Tropospheric ozone concentrations are not changed by human activity.
D.
Ozone has a relatively short lifetime in the troposphere and its distribution is not uniform around the world.
Remember that ozone in the troposphere varies widely due to anthropogenic pollution.
14. Calculate the mass of CO
2
produced by the combustion of a tank of gasoline. Assume the mass of gasoline is 46.0 kg and that gasoline is pure octane (C
8
H
18
);
A.
142 kg
B.
17.8 kg
C.
32.3 kg
D.
46.0 kg
Remember to write a balanced chemical equation for the combustion of octane forming carbon dioxide and water before doing stoichiometry calculations.
15. Calculate the mass percent of chlorine in CCl
3
F (Freon-11).
A.
77.5%
B.
25.8%
C.
51.6%
D.
13.8%
Remember that you need to take the total mass of chlorine divided by the mass of the entire molecule.