chap 5 the carbon cycle
advertisement

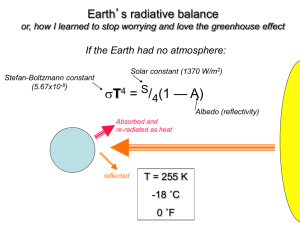
Chapter 5: The carbon cycle Recall: factors that control the climate: S, α and n σ T4 = [(n+1)S(1-)/4] with S = solar constant, α = albedo and n = layers or composition of the atmosphere Q: Are humans perturbing α and/or n? What is the natural value of n? 5.1 Composition of the atmosphere Recall: atmosphere is transparent to visible radiation but absorbs IR In particular, atmospheric greenhouse gases absorb IR Chemical composition of the atmosphere: Nitrogen gas (N2): 78% of the dry atmosphere (basically inert) Oxygen (O2): 21% of the dry atmosphere (only planet with oxygen) Argon (Ar): 1% (noble gas, also inert) Above three gases make up over 99% of our atmosphere by volume Do not absorb IR, do not warm surface - not greenhouse gases Greenhouse gases 1. Water vapour (H2O): [0.2 – 4.0 %] at surface Decreases rapidly with height (0.0005% in the stratosphere) Most abundant greenhouse gas; evaporation from oceans 2. Carbon dioxide (CO2): 0.039% in 2010 (390 parts per million) Animal and plant exhalation, emissions from fossil fuels 3. Methane (CH4): 1.8 ppmv (2010) Emissions from wetlands, ruminant animals, agriculture, fossil fuel emissions 4.Nitrous oxide (N2O): 0.3 ppmv (2010) Fertilizer and natural sources 5.Ozone (O3): 10 ppb (surface)– 10 ppm (stratosphere) UV protection in high atmosphere, pollutant at low atmosphere 6.Halocarbons (CFC, HCFCs): 10 ppb Synthetic industrial chemicals (refrigerants etc) Warming efficiency = [abundance] x [effective molecular absorption] O2, N2 : molecular absorption = 0: not greenhouse gases CH4 : molecular absorption = 20 x CO2: strong greenhouse gas YouTube video: http://www.youtube.com/watch?v=wYLmLW4k4a 5.2 Atmosphere-land exchange Abundance of atmospheric carbon dioxide over the last 50 years Note: long-term increase and seasonal cycle Seasonal cycle Abundance of atmospheric carbon dioxide 1988-1990 Annual cycle of plant growth and decay 1. Plants absorb CO2 from the atmosphere CO2 + H2O + sunlight -> CH2O + O2 Produces carbohydrates and oxygen (photosynthesis) 2. Animals and bacteria consume plant material CH2O + O2 -> CO2 + H2O + energy Produces energy and CO2 (respiration) Cycle: CO2 absorbed in (1) released in (2):O2 produced in (1) released in (2) No net change in carbon dioxide or oxygen (sunlight -> energy) How much carbon is transferred in atmosphere-land exchange? Steady-state values: 740 Gt Carbon in the atmosphere (740 x 1012 kg) 2000 GtC in the biosphere: living plants and animals, soil, permafrost (frozen carbon) Flux: 100 GtC/yr - no net gain or loss Photosynthesis: removes 100 GtC from atmosphere in a year Respiration: transfers 100 GtC back into atmosphere Turnover time for atmosphere to land : (740 GtC)/(100 GtC/yr) = 7 years Turnover time for biosphere to atmos: (2000 GtC)/(100 GtC/yr) = 20 years Global balance not uniform: most land, plants in northern hemisphere During spring and summer, plants growing and trees leafing photosynthesis > respiration → net global loss of CO2 from atmos. During autumn and winter: plants decay, releasing carbon as CO2 respiration > photosynthesis → net global gain of CO2 to atmos. Note : 1 GtC = 3.67 GtCO2 5.3 Atmosphere-ocean exchange Steady-state value Surface (mixed) layer contains 1000 GtC (about 100 m thick) Deep ocean contains about 38,000 GtC Flux: 100 GtC exchanged per year Turnover time for deep ocean is (38,000 GtC)/(100 GtC/yr) = 380 years: dominates Carbon dioxide dissolves in water, forming carbonic acid; ocean becomes more acidic (1) CO2 + H2O -> H2CO3 Carbon is returned to the atmosphere in reverse reaction (2) H2CO3 -> CO2 + H2O degassing In the ocean; carbon goes into plants and fish CO3 + critter -> CaCO3 5.4 Combined atmosphere-land-ocean system Total Flux: 200 GtC exchanged per year Turnover time for atmosphere to land/sea: (740 GtC)/(200 GtC/yr) = 3.7 years Average lifetime of carbon atom in atmosphere = 3.7 years Nearly all of atmospheric carbon replaced from land or ocean after 4 years Turnover time for land to atmos: (2000 GtC)/(100 GtC/yr) = 20 years Turnover time for mixed layer : (1000 GtC)/(200 GtC/yr) = 5 years Turnover time for deep ocean is (38,000 GtC)/(100 GtC/yr) = 380 years: dominates Several centuries for carbon atom to make round trip to deep ocean Atmos. exchanges carbon rapidly with land and mixed layer, slowly with deep ocean 5.5 Atmosphere-rock exchange Most carbon is stored in rocks such a limestone CaCO3 Carbon dioxide is transferred directly to atmosphere via volcanoes Balanced by weathering and burial of carbon in the oceans Flux: (0.1 GtC/yr) slow Chemical weathering: atmospheric CO2 dissolves into raindrops CO2 + H2O -> H2CO3 Acidic rain falls on rocks ; physical impact and chemical reaction Carbonic acid produced CaSiO3 + CO2 -> CaCO3 + SiO2 Calcium carbonate i.e. limestone (CaCO3) produced runs off to ocean CaCO3 deposited on the sea floor; carried deep by plate tectonics over millions of years Deep within the earth, pressure converts limestone to magma. Eventually, magma transferred to surface by volcanoes, releasing CO2 into the atmos Large reservoir, small exchange: many millions of years for turnover Turnover: (40,000 GtC)/(0.1 Gt/yr) = 400,000 years: 5.6 Extra pathway; fossil fuels Fossils formed when plant material buried before it can respire Stored in rock reservoirs; subjected to great heat and pressure Digging up and burning fossilized carbon releases energy - fossil fuels Also releases carbon into the atmosphere Flux from fossil fuels: 6 GtC/yr … much larger than volcano cycle Leads to a buildup in atmospheric CO2 Why associate CO2 increase with fossil fuels? A. Compare CO2 rise with recorded fossil fuel burning Increase in atmospheric CO2 tracks emissions closely Estimated emissions output with CO2 overlay B. Identify % atmospheric carbon associated with fossil fuels: Radioactivity dating: C13 and C14 concentration A + B: strong evidence that humans are responsible for rise in CO2 The CO2 cycle Not all of the CO2 added to the atmosphere stays there (about 50%). The rest goes into the ocean and land About 50% is removed in one year into the land and ocean About 20% is removed over several centuries by transport into the deep ocean Another 20% takes thousands of years by deposition in rock