Rate of reaction depends upon concentration of compounds
advertisement

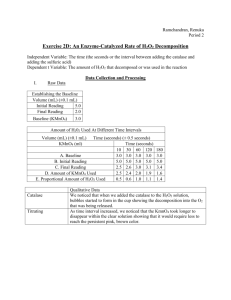
Student Studies CATALASE http://aris.gusc.lv/BioThermodynamics/CATALASE.doc Enzyme CATALASE radical reaction prevent non-enzymatic H2O2 chain reaction provocation Āris Kaksis Riga Stradin’s University 2015 Human Physiology & Biochemistry CAT increase velocity constant toxic radical 2H:-••:O-:-O:••-:H conversion to O2aqua +2H2O +Q Three H2O2 hydrogen peroxide sources from nature at presence of oxygen in water medium O2aqua+H2O, since 500 M-year’s atmospheric oxygen concentration is 20.95% volume per cents. 1. Heavy metal compounds having high oxidation number iron(III) Fe3+, cooper(II) Cu2+ , manganesium(IV) Mn4+, as well pollutant lead(IV) Pb4+ . 2. Ionization radiation: ultraviolet UV, x-ray, gamma radiation, beta particles ( 01 un 01 ), alpha particles 42 . Human body 70 kg mass each second chemical elements carbon-14 146 C and potasium-40 40 19 K isotopes irradiate 7000 beta particles 01 un 01 and high radiation energy absorbed forming in O2aqua+H2O medium of tissues 1013 peroxide H2O2 molecules per second. 3. Enzymes aldehyde oxidoreductases in water medium at presence of oxygen O2aqua + H2O gives products hydrogen peroxide H2O2. Hydrogen peroxide decomposition reaction is spontaneous as is exothermic enthalpy decreases Hr<0 evolving the heat Q in surrounding and entropy growth Sr>0 as from two H2O2 + H2O2 molecules form tree molecules O2aqua + H2O + H2O free energy decreases and its change is negative Hr-TSr= Gr<0 in thermodynamically favorable reaction. High activation energy Ea=79 kJ/mol due to absence of catalyst and low geometric factor A=0.01 M-1s-1 make the Arrhenius velocity constant expression negligible small: k =A e 79000 Ea RT =0.01 e 8 , 314 298 =0.011.41910-14=1.41910-16 M-1s-1 Velocity of reaction makes very small v = k [H2O2]2=1.41910-16[H2O2]2 Ms-1 ;if [H2O2]=1 M ; v = k [H2O2] =1.19110-8 one molar peroxide [H2O2]=1 M solution each second converts of 1.191108 molecules to products only one 1 peroxide molecule, that all would be converted we should wait 1.191108 seconds, 3.78 years. One mol H2O2 contains Avogadro number of molecules NA = 6.0211023 particles/mol and expected time complete converting is larger as 3.78 years 6.0211023/1.191108 = 51015 times and are 3.7851015 =1.91016 years. Universe age is 13.7 billion years, which is million times shorter period. Catalyst CATALASE (CAT) is 1. involved into reaction 2. decreasing activation energy Ea and 3. improve geometrical factor A from worse value zero A => 0 to maximally optimized one A => 1 , that each collision is geometrically active and effective 100% with geometric factor A = 1 favorable conversion to products. 2H2O2 + CAT H2O2CATH2O2 O2aqua+2H2O+Qexothermic+ CAT toxic transition state biological goods compound active complex oxygen+water+heat + CATALASE Biocatalyst enzyme-CATALASE according Cambridge University professor Alana Fersht shows great catalytic v enz = activity: k cat KM [E][H2O2]=3.6107[E][H2O2] , Usual catalase concentration is small and v enz =0.36[H2O2] s-1 . Activation energy of catalase Ea=29 J/mol and geometric factor A=0.1311 active collision fraction is 0.988 and velocity constant value is 0.1296 M-1s-1 , 98.8% of total collisions are active and converting to products O2aqua + 2H2O + Q: [E]=10-8M k v = =A e C AT k 29 Ea RT = A e 8 , 314 298 = A0.988=0.1296 M-1s-1 , v = k [H2O2]2 =A0.988[H2O2]2 C AT [H2O2] =0.36[H2O2] ; k k = 0,36 1,19 10 8 4.) 3010 times greater velocity constant increase =( 1 6 Student Studies CATALASE http://aris.gusc.lv/BioThermodynamics/CATALASE.doc Advanced Studies of Chromo Protein CATALASE Heme pocket active site Experimental research: http://aris.gusc.lv/06Daugavpils/Research/CATALASE.doc H O M= 616.51 g/mol O H H2C Asn147 His74 CH3 O O H3C H2C :N N Fe H3C 3+ CH3 O Fe CH3 :N N Fe 3+ CH3 O O :N N CH3 O H2C M= 616.51 g/mol CH3 O O O :N O H O H3C O H2C C34H32N4O4 H2C O Asn147 + His74 CH3 O M= 616.51 g/mol 3+ N H2C H O :N N O O :N N Asn147 His74 CH3 + O H O H CH3 C34H32N4O4 C34H32N4O4 Tyr357 Tyr357 Tyr357 CH2 CH2 CH2 Singlet •::O-:-O::• iron(III) Fe3+ on Heme center in to water medium conversed to triplet oxygen •:O≡:::≡O:• Heme pocket with 28 hydrophobic amino acids – in waterless medium oxidation-reduction reaction absent: Val72,Val73,Ala75,Val115,Ala116,Pro128,Gly130,Val145,Gly146,Phe153,Ile154,Ala157,Leu159,Phe160, Pro161,Phe163,Ile164,Leu198,Leu298,Ala332,Phe333,Pro335,Met349,Leu350,Gly352,Ala356,Pro358,Ala434 Singlet •::O-:-O::• ————→ triplet •:O≡:::≡O: Asn147 His74 CH3 O H O H O M= 616.51 g/mol H2C O H3C N H2C H3C 3+ H O H :N N O O H Asn147 His74 + CH 3 N O O CH3 H O H :N O H2C CH3 O CH3 O O HEM_.tgf O N O O :N Fe 3+ H2C H O H :N O O O CH3 CH3 M= 616.51 g/mol C34H32N4O4 C34H32N4O4 Tyr357 CH2 H N CH3 C34H32N4O4 H2C H3C 4+ Asn147 His74 CH3 H O HEMO2.tgf :N Fe O O N O H2C O H O HEM-O2.tgf :N Fe M= 616.51 g/mol Tyr357 CH2 Tyr357 CH2 H2O2 + H2O2 + CAT H2O2CATH2O2 O2aqua + H2O + H2O + Q + CAT toxic transition state biological goods compound active complex oxygen + water + heat + CATALASE Conclusions CATALASE 1. Catalyst CATALASE is involved.......... .......... 2. Catalyst decrease .......... 3. Catalyst improve .......... 4. Catalyst increase k .................... 2