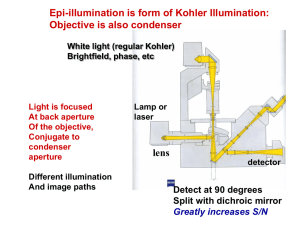
Quantum yield determination using comparative method Use the
advertisement

Quantum yield determination using comparative method Use the following solvent for all experiments: 20 mM Tris/HCl pH 7, 50 mM NaCl. 1) Determine the extinction coefficient of the nucleoside at 260 nm. Prepare three different solutions of your nucleoside of known concentration. Measure first the buffer only as a blank, and then measure the UV/VIS spectra of the nucleosides. Note down the extinction at 260 nm and at the maximum wavelength of absorption. The extinction should not be larger than 1. Calculate the extinction coefficient at a particular wavelength according to Lambert-Beer: =molar extinction coefficient [M-1 cm-1] c= sample concentration [M-1] d=path length of cuvette [cm] 2) Determine the quantum yield. Make three nucleoside and three standard solutions. Standard: quinine sulfate in 0.1 M H2SO4 (quantum yield (Q) = 0.577 for excitation at 350 nm). Measure again the UV/VIS spectra of all solutions. This time all extinctions should be below 0.05 to avoid inner filter effects. Before you start, double-check the wavelength calibration of the fluorimeter. The easiest way to do this is to measure the Raman band of water. Excite at 350 nm. The Raman band should peak at 397 nm. Note that the Raman band shifts as you change the excitation wavelength. It is always 47 nm red-shifted relative to the excitation. Some people mistake the band for fluorescence of their compound. If you examine an unknown nucleoside, the wavelength of the emission maximum must be independent of the excitation wavelength you chose! Use exactly the same solutions you used to record the UV/VIS spectra to measure the fluorescence emission. The excitation wavelength should be close to the absorption maximum and identical for the standard and the nucleoside. Integrate the peak area. If you want to present the data in a paper, you need to show the corrected emission spectra. Usually fluorimeters have an integrated correction option that eliminates distortions of the spectrum that originate from the wavelength dependence of the excitation source, the monochromators and the detector. Use this equation to calculate the quantum yield: Q=quantum yield I=Integrated fluorescence intensity E=Extinction n=refractive index The index R indicates the standard.