Light Scattering, Sedimenation, Gel Electrophoresis, Higher Order
advertisement

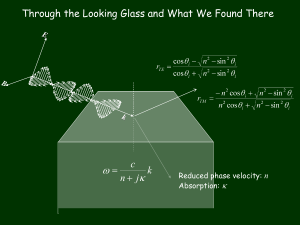
I . Separation Techniques (chapter 5 of V-H and notes from elsewhere) A. General Principles 1. –fv + F = 0 or F = fv (terminal velocity, -Fb = -bv, a = 0, F = eE or “centrifugal + buoyant”). 2. For sphere, Stoke’s law f0 = 6R, where = viscosity (g/cm.s = poise). Water = 1 cp. f f For others, f = f0, where is obtained from table 5.1 The f0 f0 f smallest frictional drag is from a sphere and gives the drag f0 for another type of particle with an equivalent volume (4/3 Re3). e.g. Prolate ellipsoid. 1 1 f P 3 P2 1 2 , f0 = 6Re, Re = (ab2)1/3, P = a/b. 1 f0 ln P P 2 1 2 b a B. Sedimentation 1. Forces Fc = "centrifugal" force, really acceleration, mv2/r. v = r Fc = mr2 Fb = buoyant force = “weight” of displaced fluid = mor2 Fd = frictional force = -fv Ignore gravity Fb Fd a -Fb-Fd = - mv2/r or Fc-Fb-Fd = 0. mr2 - mor2 –fv = 0. mr2 (1- v ) -fv = 0, with v = specific volume ~ volume/mass of substance in solution (V/m). = mo/V, V = volume. Mulitply by Avogadro’s number, R = NA. mNA = M (molecular weight) [m = g/molecule, M = g/mole, NA = # molecules/mole]. M(1- v ) r2 – NAfv = 0 M1 vρ v s *** NAf 2 r 1- v is the buoyancy factor ~ 1- o/ (if o > then it floats). s = sedimentation coefficient, [s] = Svedberg, 1 x 10-13 sec = 1 Svedberg. 2. Determining s a. Analytical centrifuge – absorption b. vb = drb/dt = 2srb, where the subscript b signifies the boundary (so rb is the boundary between solvent and solution). drb r t 2st t *** 2sdt ln b 0 r t b 0 rb So plot ln(rb) vs t and get slope which is equal to 2s. c. Diffusion blurs boundary (discussed in detail below) Simply: D = RT/NAf, ([D] = cm2/s as RT has units of(g cm2)/(s2 . mole) and f has units of g/s) x ~ Dt d. Variance of f w/ T, due to variance of (T). Define w/r to standard conditions: 20oC in water: 1 vρ20, w ηT, b s 20 ,w s T ,b 1 vρT, b η20, w e. s depends on M and f (shape). sphere: f0 = 6R0 and 4/3.R03 = V0 = M v /NA 1 s* = s 020 ,w v 3 1 vρ 2 M 3 2 4π 3 6πηN A3 3 1 , here, 0 is s 020 means 0 concentration, that is low concentrations so you avoid concentration effects See Figure 5.14, variance due to non-sphericity.(only globular proteins fit curve). f. If know M, can use s to get shape factor f = 6Rs, where s is the stokes radius g. Density gradient – use buoyant force, can separate different densities (example sickle red cells). C. Electrophoresis Now have F = ZeE, fv = ZeE. Mobility, U = v/E = Ze/f Sphere: U = Ze/(6R) D. Movement on a gel. (see math supplement) 1. Cx , t 2 Cx , t D Fx Cx , t 2 t x x D = diffusion constant = mobility of molecule F = force of molecule at position x 2. C(x, t=0) = N.(x-x0), where .(x-x0) is the dirac delta function. F(x) = F, constant (ZeE) for electrophoresis Define Ck , t dxe ikx Cx , t dk ikx C(x,t) = e 2π ikx 0 Ck , t C (k,0) = N e Put in differential equation dk 0 e ikx Ck , t Dk 2 Ck , t μFik Ck , t 2π t Since FT of [] is zero then [] 0 0 Ck , t Dk 2 Ck , t μFik Ck , t t dC Dk 2 μFik dt C Ck , t Ck ,0 exp - t Dk 2 μFik 2 Ne ikx 0 e t Dk μFik Transform back Cx, t N 2π N ik x x 0 tDk 2 tμFik e e dke 2π x x 0 μFt 2 tD e u tD k i 2 x x 0 μFt tDk i2 tD dke x x 0 μFt 2 tD du tD dk , e- u du π 2 N Cx , t 2 π x x 0 Ft 2 tD 1 e tD 2 2 This is a gaussian centered around x = xo + Ft*** With rms of tD , velocity = F, = 1/(6r) with r = radius and = viscosity D = kBT Do simple examples of 1D diffusion See Maple animations (gelg and explain, newgelg) E. Running DNA on a gel 1. Closed small plasmids run as rigid particles - See discrete bands (more below) 2. Long DNA (10-50kb) Persistence length = 500-600 angstroms, goes as 1/mass (tunnels) Say #cations = DNA charge Separation causes force – more work to separate - If lots of cations get screnning 1 Xκr μ 1 κr 6ππη Xκr some function 1 8πN o e 2 2 1 I 2 κ 1000ε 0 k B T 1 I C i Zi 2 ionic strength 2 Ci = concentration of ion, Zi = charge of ion 3. 2-D Electrophoresis Pulsed Field electrophoresis DNA snakes its way through Migrates as 1/mass, i.e. 1/length Genome project (was $1 per base pair) Using Saenger method P P P OCH 2O Base H HH H H 3'-GAATCTAGCTC – Primer 5'-CTTAG Mix DNA polymerase I, nucleotides and labeled didexoy base analogs (dATP, dTTP, dCTP, dGTP, each labeled with different fluorescent dye). Used to mix with one didexoy at a time. Run gel. C T A G 4. Running plectonemic helices a. Review from chapter1 L, T, W, L = T + W Supercoiling important in vivo b. For DNA of several Kbp main effect on mobility is radius, as radius decreases it becomes more mobile. Since twist is approximately constant, mobility goes as writhe and hence L which is an integer (for same length). c. If nick DNA they all run together d. For a large number of base pairs, if add a few more, doesn’t make a difference e. If have DNA plectonemic helices of a defined length, and run on gel, get a gaussian distribution of bands due to topoisomers, difference in writhe due to difference in linking number. F. Determination of Helical Repeat of DNA (Wang, 1979) 1. Definitions # base pairs = n + x l0 = distance between bands h = helical repeat 2. Hypothesis a. If x is an integral number multiple of h then no change in pattern L = T + W L+x = T + x + W, W stays same b. If x is not an integral multiple of h then pattern shifted by(x/h) l0 T = integrated twist angle/2if increase T by 1 you have added h base pairs. T T + 22= T + 1, if increase T 90 90 90 Original W L 10 100 11 101 12 102 by P/2 T T + ½ Writhe must go down by ½ , shift = (x/h) l0 = l0/2 3. Example, Say we have Increase x by integer Increase x by non-integer Position T W L Position T W L Position -9 91 9 100 -9 90.2 9.8 100 -8.8 -10 91 10 101 -10 90.2 10.8 101 -9.8 -11 91 11 102 -11 90.2 11.8 102 -10.8 Wang found that h = 10.4 ± 0.1 Slightly different than W-C (crystal) Is twist constant? II. Light Scattering (Chapter 7 (Van Holde) and more) A. Single Particle – Rough Treatment Isotropic small particle P E ; excite molecule and it accelerates and reradiates. 1 d 2P Incident: E E 0 ei k 0 r t , Scattered : E s is propotiona l to and c 2 dt 2 falls off as 1/r. The scattering angle, , is defined w/r transmitted light k0 in the scattering plane. The scattering plane is defined by Sample Light Source the vectors k and k0. dipole For Horizontally polarized light: 4 2 E 0 cos ik r it 16 4 2 cos 2 Es e , IH = Is/I0 = r2 4 r 2 For Vertically polarized light: IV = Is/I0 = 16 4 2 4 r 2 For unpolarized light: Is/I0 = ½ (IV + IH) = 8 4 2 4 2 1 cos 2 r B. Dilute “gas” of small particles. Classius-Mosotti: n2 – 1 = 4N, where n is the index of refraction, N is the number of particles per unit volume. For a gas n ~ 1. Taylor Series expansion: n 1 + (dn/dc)c + … dn/dc ~ n/c – how n changes w/c, c being the mass concentration. n2 = 1 + 2(dn/dc)c + (dn/dc)2c2 + ... 1 + 2(dn/dc)c c dn M dn 2c(dn/dc) = 4N, = , with M = molecular 2N dc 2N A dc weight and NA = avogadro’s number. [c/N = M/NA: (mass/volume)/(moles x NA/volume) = (mass/mole)/NA] Is 2 2 M 2 1 cos 2 dn (unpolarized) I0 dc N 2A 4 r 2 2 Intensity molecular weight 2 scattered light/volume is is Is N A c Is 2 2 1 cos 2 dn N cM , so goes as M. 4 2 I0 I0 M I0 dc NA r 2 C. Macromolecules in solution. 1. Without interaction n2 – 1 n2 – no2, where no is the index of refraction of the medium. Take n no n2 – no2 = (n + no)(n – no) (n+no)dn 2 no(dn/dc)c Note: If n = no no scattering See demo of disappearing beaker. i 2 2 n o 2 1 cos 2 dn cM 4 2 I0 dc NA r 2 i 2 2 n o 2 r2 dn cM R = Rayleigh ratio = I 0 1 cos 2 dc N A 4 R = KcM 2 2 n o 2 dn K= N A 4 dc 2 2 2. Add small interaction Kc/ R = 1/M Kc/ R = 1/M + 2Bc B = 2nd virial coefficient – fudge factor. D. Large Macromlecules s > 25 nm (polymers) Can get size and molecular weight Phase difference k particle ko At = 0, get no phase difference – have scattering from different parts of the particle but the difference in spatial phase within the particle is made up by temporal phase at = 0. At large have big phase difference. 4 sin Remember, phase difference is q. r and q 2 So we get lots of forward scattering Define P() = ratio of scattering of extended particle to an isolated small particle (dipole) where interference is ignored. 1 N N sin qR ij P() = with N = number of scattering points, Rij = N 2 i 1j1 qR ij distance between them and q sin qR ij qR ij 4 sin . 2 1 qR ij 2 qR ij 4 ... 6 120 For small particles, or small , qRij 0 and P() = 1 so we have Raleigh scattering (small particle means we have max Rij << ) q2 N 2 First two terms: P() = 1 R ij 6 N 2 ij Define radius of gyration Rg2 = 1 N 2 R ij 2 N 2 ij Shape Sphere Rg 3 5 Rs Ellipsoid a 2a 2 b 2 / 5 L 12 Rod q 2R g 2 16 2 R g 2 1 sin 2 2 2 3 3 thus the angular dependence gives Rg. P() = 1 Now have Kc 1 1 2Bc R P M Kc 1 1 2Bc 2 2 R M 16 R g 1 sin 2 2 32 Use 1/(1-x) 1 + x 2 2 Kc 16 R g 2 1 1 sin 2Bc 2 2 R M 3 Get Rg and M from Zimm Plot – Remember, for a given concentration, c, we measure ias a function of angle and calculate R. Then calculate Kc/R. Curve A shows what you would get at a constant concentration, cA. One would need to extrapolate to = 0. Do this for several concentration Kc 1 2Bc (straight line) R 0 M Intercept = 1/M, slope 2B Curve B, , keep fixed, extrapolate to C=0 2 2 Kc 1 16 R g 1 sin 2 2 R c0 M 32 slope gives Rg, intercept gives 1/M E. Polarized Light Scattering (and a more rigorous formulation of scattering). 1. The First Born Approximation and the coupled dipole approximation In general, k 2 e i k .r i k .r ˆ ˆ E( r ) E 0 r (1 kk ) dVe V .E r r V is the polarizability per unit volume ˆkˆk involves the outer product. Tij A i B j for Tij A B A B A x B y A x Bz x x T A y B x A y B y A y Bz A z B x A z B y A z Bz 1 ˆkˆk is the transversality condition. Example. If ˆk ˆi , show ( 1 kˆ kˆ )E is perpendicular to k̂ . Work out in class Born Approximation s k 2eik .r i k k o .r 0 E (r) V r' .E dV' e r V' , integral is over particle – no interaction (trasversality condition is implicit). Coupled-Dipole Approximation. k 2eik .r N ik .r j Es (r ) e j .E j r j1 where the sum is taken over each point polarizable group (dipole) and the field at the ith dipole is N 0 ik .r E i E i e 0 i a ij j .E j bij ( jE j .ˆn ij )ˆn ij j i and a ij e ik .rij rij ik .r 1 ik e ij 3 3ik 0 ( k 2 2 ) and b ij (k 2 2 ) , Ei rij rij rij rij rij is the incident electric field, n ij is a unit vector pointing from the ith to the jth dipoles and rij is the distance between these dipoles. When interaction between the subunits can be ignored (when the particle is weakly polarizable) then only the first term need be included and we get the first Born approximation. 2. The Stokes Vector (see polarization Bkgd) the intensity and polarization state of light is fully described by the four elements of the Stokes vector, I E 2h E 2v Q 2 2 E E h v U E 2 E 2 2 2 V E r E l where v, h, +, -, r, and l refer, respectively, to the vertical linear, horizontal linear, positive diagonal linear, negative diagonal linear, and right and left circular polarization components for light of arbitrary polarization, and I, Q, U, and V are, respectively, the total intensity, the difference between vertical and horizontal polarized intensities, the difference between diagonal intensities, and the difference between the circularly polarized intensities. The Stokes’ vector is usually normalized and can be defined operationally (eg. I is the intensity with no filters, Q is the difference in intensity when a vertical vs horizontal polarizer is used, etc.) 1 1 1 0 1 0 Examples , , : what do these represent? How would 0 0 1 0 0 0 we represent right circularly polarized light? Work out in class. 3. Mueller Matrices. a. Definition The effect of the optical properties of a substance or optical element on the Stokes vector can be written in terms of the Mueller matrix, M 11 M M 21 M 31 M 41 M 12 M 22 M 13 M 23 M 32 M 42 M 33 M 43 M 14 M 24 M 34 M 44 I I Q M Q U U V f V i b. Mueller matrices for optical elements. Linear Polarizer: cos 2 sin 2 0 1 cos 2 2 cos 2 cos 2 sin 2 0 1 P sin 2 2 0 2 sin 2 cos 2 sin 2 0 0 0 0 For vertical = 90, horizontal = 0, and diagonal = 45. Circular polarizer: 1 0 0 1 0 0 0 0 , right is upper, left is lower 0 0 0 0 1 0 0 1 Retarder at ±45o: 0 0 0 1 0 Cos( ) 0 Sin ( ) , where is the strain. 0 0 1 0 0 Sin ( ) 0 Cos( ) Examples: Show that a vertical, horizontal, circular polarizer do what they are supposed to for incident un- and linearly polarized lights. Show that a vertical polarizer + 90 degree retarder at 45o can act as a circular polarizer, with the polarizer first. Work out in class. (See Maple). Significance of Muller Matrices i) Each element describes a change in polarization Eg. Ir = ½ (M11 + M14) Io IL = ½ (M11 - M14) Io M14 = (Ir – IL)/Io Where Ir is the intensity of the light you get 1 0 when the incident light is Io 0 1 A ln 10 ( Ah Av ) / 2, LB 2 ( nh nv )l / , and IL is the intensity of the light you get when 1 0 the incident light is Io. Thus M14 is CD in 0 1 absorption and CIDS in transmission (absorption). In general, in absorption, each element has a direct interpretation: LD LD' CD 1 LD 1 CB LB' A Me LD' CB 1 LB 1 CD LB' LB where LB' 2 ( n n )l / , CB 2 ( nl nr )l / , A LD ln 10 ( Ah Av ) / 2, LD' ln 10 ( A A ) / 2, CD ln 10 ( Al Ar ) / 2, denotes an absorbance, LB refers to linear birefringence, LD refers to linear dichroism, CB denotes circular birefringence (or ORD) and , n, and l are the wavelength of light, index of refraction, and path length of the sample. The subscripts h, v, l, r, + and - refer to horizontal, vertical, left circular, right circular, +45o, and –45o polarizations. In scattering, The Mueller scattering matrix elements, for a randomly oriented sample, can be written (Tian and McClain, 1989) A B B C h j f g h j D k f g . k E The symmetry of this scattering matrix, which disappears for samples containing preferred orientation, was first demonstrated by Perrin (1942). The uppercase elements have been called dipole elements because they do not disappear in the dipole limit (Harris and McClain, 1985). That is, these elements are non-zero even for small particles of low polarizability where the electric field within the scattering particle is essentially equal to the incident electric field. The lower case elements are called retardation elements or non-dipole elements because of their greater sensitivity to polarizability. The elements h, j, and k are identically zero in the orientation average unless the induced electric field, due to interactions within the particle, is accounted for (Harris and McClain, 1985). The elements f and g are not necessarily zero in the dipole limit, but they are very small and hence have large contributions resulting from interactions within the particle (McClain and Ghoul, 1986). Perrin showed that the elements h, f, j, and g are zero in the orientation average unless the particles have some inherent chirality. Thus, these off-diagonal block elements are called helicity elements (Tian and McClain, 1989). The element M11 describes the total intensity of scattered light. This element is most commonly measured to provide general size information. The other dipole elements can also be used for this purpose. In addition, the deviation of M22 from unity and M33 from M44 is indicative of the non-spherical symmetry of the particle (Quinby-Hunt et al., 1989). The element M14 is also known as circular intensity differential scattering (CIDS) and has been used to probe the chiral nature of particles. The other helical elements can also be used for probing chiral structures. In addition, these elements are useful to ascertain whether the scattering medium has any preferred orientation, as they are zero unless the particles in the medium are chiral or have some preferred orientation. M34 can give especially sensitive information regarding particle size and polarizability, since it is the only retardation element that is not also a helical element. The first and most difficult part of calculating the Mueller matrix for a particle is to calculate the scattered electric field for a given incident electric field. Once the parallel and perpendicular components of the scattered field are calculated for incident parallel and perpendicular electric fields, the scattering amplitude matrix is calculated which relates these components of the incident and scattered fields as follows (Shapiro et al., 1994b): E sh' 1 J11 s exp(ikr) E v' kr J 21 J12 E ih J 22 E iv where i and s are now superscripts referring to scattered or incident. The prime qualifiers for h and v are necessary to distinguish the vertical and horizontal directions for the scattered light from those of the incident light. If the particle is not spherically symmetric, the orientation average of the scattering amplitude matrix elements is calculated by integrating them in terms of Euler angles over all possible orientations. It is straightforward to calculate the Mueller matrix elements from the scattering amplitude elements, Jij (Bohren and Huffman, 1983). 4. Measurement of the Mueller Matrix Many elements are very small, including interesting ones like CIDS. One method of detecting small signal in large background is modulating the signal. The method described here works for CD and CIDS. Light source 90o Polarizer PEM at 45o Sample Filter? detector PEM Quartz (strain) Apply ac voltage – piezoelectric crystal Polarization State: The Mueller matrix of PEM is the same as that of a retarder at 45o with = Sin(t) where is the frequency of modulation (usually 50 kHz), and A is the amplitude of the modulation. The amplitude of modulation is wavelengthdependent, = 2ds/, with d being the width of the crystal, s is a strain factor that depends on the material and the applied voltage, and is the wavelength. We get 1 1 1 a Polarizer 1 PEM cos b 0 0 c 0 sin M11 M12 cos M14 sin sample M 21 M 22 cos M 24 sin M M 32 cos M 34 sin 31 M M cos M sin 42 44 41 We can expand the cosine and sine terms as Bessel Functions: Cos(Asin(wt)) = J0(A) + J2(A)cos(2wt) + J4(A)cos(4wt) + … Sin(Asin(wt))= J1(A)sin(wt) + J3(A)cos(3wt) + … A lock-in amplifier can select signals that are oscillating either at the same or twice the frequency of a reference signal, thereby selecting either the sine or cosine terms. A feedback servo circuit is used to keep the DC current constant, effectively normalizing by M11. So if no final filter is used, The DC current is M11 The 1f signal is M14/M11 The 2f signal is M12/M11 Ex – How would you get M22 and M13? Work out in class. F. Octopus Sperm Chromosome. Used PLS to show DNA is perpendicular to the fiber axis. See SEM and TEM of fiber a. TEM shows thick helical fiber with a 200 nm radius, 100 nm thick. b. Model r' = rh cos(h) i+ rh sin(h) j + (Ph/(2 k pp pp tt nn nn tt The polarizability tensor is defined by its magnitude along the principle axes, tangent (t), normal (n), and binormal (p) to the polymer. Thus, where the strengths along each principle axis have a real and imaginary component. Normally, t = derivative, normal = 2nd derivative and binormal is ˆt ˆn . Show these are perpendicular for r'. Used to move between these. C. Results – Figures 7 and 8 – DNA is perpendicular to fiber. D. Consistent with cholesteric crystal model. 30 nm fiber G. Higher order DNA structure and function 1. Eukaryotic structure (Prokaryotes probably have some proteins too). a. DNA is highly condensed (mitosis: 5000 times shorter). b. As degree of condensation increases, knowledge of structure decreases. 2. Nucleosome a. Evidence Maurice Wilkins’ X-Ray structure gave 100 angstrom helical repeat Histone proteins found in chromatin Cut by DNAse every 10 nucleotides wrapping b. Structure 146 bp wrapped around core particle (110 x 55 angstroms) with H1 have 166 bp involved in double wrap Recently the amount of DNA around core particle has been found to vary between 100 and 170 bp c. 30 nm fiber (see pictures) Solenoid model - 6-7 nucleosomes/turn - nucleosomes parallel to helix axis - space in center Crossed linker model - Alternating sequence of nucleosomes - Variable diameter – depends on linker length - 26 nm pitch - filled center d. Protein scaffold 30 nm fiber attached to protein matrix e. Function efficient compaction Txn control - Domains: genetically distinct regions - In dipterian polytene chromosomes see band structure: compacted vs condensed regions, number of bands corresponds to number of genetic functions - Lampbrush chromosomes – unlooped active regions - IN other eukaryotes have differential DNAse sensitivity - Eg globin gene Early embryo – adult and embryo region sensitive Later inactive embryo region is insensitive Replication and Topoisomerases Knots and Catanes distinguished by electrophoresis Necessary enzymes: Topo I SS DNA and Topo II for ds DNA.