Nuclear
advertisement

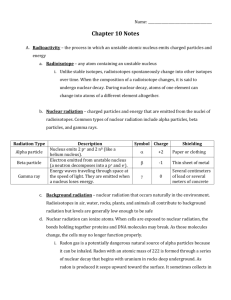
Chemistry - Dr. May Notes Nuclear Chemistry The Nucleus and Radiation The Nuclide is name given to the nucleus of the atom. A beta particle has the same mass as an electron and has a negative charge. It can be designated as β or –10e. An alpha particle is a helium nucleus having 2 protons and 2 neutrons and no electrons. The alpha particle has a mass of 4 and is designated as or 4 He 2 Ionizing radiation has sufficient energy to form ions (gain or lose electrons) and includes alpha, bets, gamma, and X-rays. Nonionizing radiation will not form ions and includes radio waves and visible light. Background radiation amounts to about 100 millirems of ionizing radiation per year. High altitude (airplane) and X-rays can increase this exposure. Types of Nuclear Decay In alpha emission, a new element is formed which has an atomic number of two less and an atomic mass of four less since the alpha particle is a helium nucleus. The helium nucleus contains two protons and two neutrons. An example of alpha () emission is the radioactive decay of Thorium-230 to Radium-226 as shown below. 230 Th 90 4 226 He + 2 Ra 88 The other common type of nuclear decay involves the emission of a beta () particle or an electron. In this case a neutron in neon-23 is converted to a proton to make sodium-23 as shown below. 23 Ne 23 0 –1 e 10 + Na 11 Energy and Mass in Nuclear Reactions 1 The famous equation formulated by Einstein tells us that matter can be converted into energy and vice-versa. E is the energy in joules, m is the mass in kilograms, and c is the speed of light (3.00 x 108 meters/sec). In the alpha decay of radium-226 to form radon-222, 0.0053 grams of mass is converted into energy. 226 88Ra 222 86Rn + 4 2He + energy E = mc2 E = (5.3 x 10–6 kg)(3.00 x 108 m/sec)2 E = 4.8 x 10 kg m2/sec2 (1 joule = 1 kg m2/sec2) (1 kJ = 1000 joules) E = 4.8 x 108 kJ 11 Half-Life The time it takes for one-half of a radioactive sample to decay is known as halflife. The fraction of a sample that remains after a certain amount of time can be expressed as (½)n where n = the number of half-lives in a certain period of time. To determine what fraction of Strontium-90 (half-life of 29 years) from the 1963 nuclear-bomb test will remain in the year 2050 (87 years later): 87 years ÷ 29 years = 3 half-lives. Then (½)3 = 0.125 or 12.5% will remain. Nuclear Decay and the Environment The most harm from nuclear decay is probably the result of man-made isotopes from weapons and power plants. Using Nuclear Reactions for Research Synthetic Elements Transuranium elements do not exist in nature. All of these elements have halflives of a few days or less. Radioactive Dating The half-life of carbon-14 (5730 years) is used to determine the age of many very old items. Carbon-14 is taken into a living plant as a part of the CO2 it breathes. The rate of decay of carbon-14 in a living plant is 15.3 decays per minute per gram of carbon. A plant that died 5730 years ago would have 7.65 decays per minute per gram of carbon. The disintegration rate would be 15.3 x (½)n decays/min/gram where n = the number of half-lives that have passed since the organism died. 2 Radioactive Isotopes as Tracers Radioactive elements react in the same way as non-radioactive elements do since chemical reactions involve the electrons around the nucleus and do not involve the nucleus. Radioactive reactants can be substituted for others and detection instruments can monitor the reaction. Nuclear Reactions for Energy Splitting Nuclei – Nuclear Fission Fission is a nuclear reaction in which a nucleus is broken into two or more new nuclei. The reaction is initiated by the bombardment of the nuclear reactants with neutrons. 235 1 139 U 0n + 94 Kr + 3 10n + energy Ba + 92 56 36 The reaction gives off more neutrons, which split more nuclei thus setting off a Chain Reaction. A certain amount of material is needed to sustain this reaction. This amount is called the Critical Mass. Nuclear Power Plants Nuclear power plants are fission reactions. The large amount of energy given off is used to heat water, which is turned to steam, which turns turbines. There is a great energy loss during these energy transfers. Building Nuclei – Nuclear Fusion Fusion is the nuclear reaction that occurs on the sun. Hydrogen is fused to form helium and energy is the result. 3 H + 1 2 H 1 4 He + 2 3 1 0n + energy When Uranium undergoes fission, one kilogram of Uranium will give energy equal to the combustion of 2 million kilograms of coal. The fusion of one kilogram of hydrogen gives off energy equal to 40 million kilograms of coal. Nuclear Waste The questions to be asked on nuclear waste are: 1. 2. 3. 4. 5. What is the half-life? What is the product? What radiation is emitted? Can the waste be shielded? Will the waste harm the food chain? 4