HEP_26283_sm_SuppInfo2
advertisement

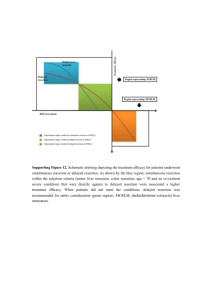
Supporting Mini-Systematic Review and Meta-Analysis on the Establishment of Selection Criteria for Patients Who are Suitable for a Simultaneous Resection Today we know that multiple patients are suitable for simultaneous resection and prognostic factors which significantly influence the short- and long-term outcomes have been reported. However, generally accepted selection criteria have not been established so far. Thus, the establishment of suitable selection criteria would be the most interesting result of the present study. On the basis of the included studies and other evidental documents, a mini-systematic review was conducted, and a meta-analysis of the risk factors for postoperative morbidity was performed on Reddy et al.’s and Luo et al.’s studies with a total of 1015 cases (due to the diverse forms of reporting, simply direct data-pooling analytical method was not appropriate).27,49 Particularly, we also refer to current published prognostic scoring systems for patients with colorectal liver metastasis. These published prognostic scoring systems were not limited to synchronous colorectal liver metastasis, let alone simultaneous resections. However, of note was the observation that the results of the present meta-analysis regarding the intraoperative blood loss, operation time, and complication rate might be influenced by the fact that multiple surgeons prefer a simultaneous resection in patients with a restricted metastatic disease, whereas extended and anatomical difficult resections are rather performed as staged procedures. This could severely influence the perioperative results in both groups and ignore the unrevealed prognostic factors in the groups of simultaneous resections within the included studies. From another point of view, when excluding other risk variables, simultaneous resection is associated with a higher risk of mortality in patients with synchronous colorectal liver metastases (Nordlinger et al. reported an operative mortality of 7% for simultaneous resections, compared with 2% for staged resections based on 1568 patientss9), which is not surprising as simultaneous resection is essentially a more invasive procedure to staged resection. Thus, comprehensive and objective selection criteria would be the aim of this mini-systematic review and meta-analysis. Detailed assessments of the risk factors are listed below. Prognostic Factors for Patients with Synchronous colorectal liver metastases within included studies Patient Demographic Information Patient demographics form the core of epidemiological objective characteristics data of a population in any medical institution. They allow for the identification of a patient and his or her categorization into categories for the purpose of statistical analysis. In the included studies, commonly examined demographics include age and gender, and both distributions and trends of the two variables within Capussotti et al.’s and Thelen et al.’s studies are of interest.38,42 Univariate analysis in Capussotti et al.’s found the sex to be one of the five statistically significant prognostic factors in the synchronous resection group (P = 0.0233), which was further identified by multivariate analysis as independent prognostic factor (P = 0.040).38 The finding that male patients had a statistically significantly worse prognosis than female patients has never been clearly explained up to now. Alternatively, female patients owned a better prognosis after hepatectomy for colorectal liver metastases implied by previous Adson et al.’s and Hohenberger et al.’s studies.s19,s20 It is hypothesized that sex hormonal may influence the prognostic outcome, still, future studies are necessary to confirm and clarify this interesting observation. Thelen and colleagues have found that in patients with simultaneous major liver resection, an age exceeding 70 years correlated significantly with postoperative mortality in the univariate analysis (P = 0.029), whereas in younger patients, the mortality rate was comparable between both groups after major hepatectomy.42 This finding was also in line with Tanaka et al’s that patient age of 70 years or older (P < .05) predicted decreased overall survival by univariate analysis.37 In summary, male sex and patient beyond the age of 70 years may be considered as potential risk factors for simultaneous resection. Extend of Liver Resection A majority of included studies have mentioned the term major hepatectomy (more than three Couinaud segments) which was commonly used to describe liver resections at risk for postoperative complications. As a matter of fact, no standard definition of major hepatectomy exists until Reddy and his colleagues, after reviewing the demographics, diagnoses, surgical treatments and outcomes from 1670 patients underwent a hepatic resection at two high-volume centres, found that resection of four or more liver segments was independently associated with postoperative mortality and severe, overall, and hepatic-related morbidity (all P < 0.01) on multivariable analysis. Thus, consistence with the included studies, a major hepatectomy should be defined as resection of four or more liver segments.s21 In this mini-systematic review, meta-analysis on the risk factors of postoperative morbidity for a total of 1015 cases has revealed that major hepatectomy was associated with a higher risk for simultaneous resection than minor hepatectomy (P = 0.05). In Thelen et al.’s study, four patients (10%) died in the postoperative course. These four patients all had undergone simultaneous major liver resections (two right lobectomies, one extended right lobectomy, and one right trisectionectomy).42 In parallel to our own results, major liver resection was the only independent risk factor responsible for postoperative mortality in simultaneous liver resections in that study (P = 0.041). While in the delayed resection group, none of the investigated parameters revealed statistically significant influence on the postoperative mortality. Also, a 12% postoperative mortality for simultaneous resections and a 24% mortality for simultaneous resections involving major liver resections have been reported by Bolton and Fuhrman.s22 In 2007, the largest multi-center retrospective study on this topic so far has reported that for major hepatectomy, combined severe morbidity (which required operative or nonoperative intervention or contributed to prolongation of hospital stay) after delayed resections was lower compared to simultaneous resections (36.1% versus 17.6%, P = 0.05. Major hepatectomy independently predicted severe morbidity in the group of simultaneous resection, HR = 3.4, P = 0.008), but it was similar for minor hepatectomy (14.1% versus 10.5%, P > 0.05).27 Moreover, intraoperative blood loss, frequency of microscopic positive hepatic resection margins (R1 resection), mortality, overall morbidity, and length of hospital stay after hepatectomy were all greater after simultaneous resections compared to after hepatectomy alone among patients who underwent major hepatectomy. In summary, major hepatectomy might be avoided in simultaneous liver resections for safety consideration from a conservative point of view, and minor liver resections are equally safe as simultaneous and staged resections. Liver Metastasis Half of patients with colorectal cancer have liver metastasis during their illness. In the Western world, metastasis to the liver is more common than primary liver cancer. In Capussotti et al.’s series, for the first time, the presence of more than three metastases was confirmed to be a prognostic factor in the synchronous group, and patients with more than three metastases had a significantly worse survival in synchronous group than in delayed group (3-year survival, 15.0% vs. 34.3%, P = 0.007).38 In multivariable analysis for the total study population by de Hass et al., three or more colorectal liver metastases at diagnosis (RR = 3.5, P = 0.001) was also identified as an independent predictors for disease recurrence.47 Resection of structures infiltrated by liver metastases was associated in eight patients: diaphragm in six patients (five in the delayed group), portal vein in one, and rib in one (both in the synchronous group) (4.2% vs. 8.8%, P = .504) in Capussotti et al.’s study. Univariate analysis found the associated resection of structures infiltrated by liver metastases to be one of the five statistically significant prognostic factors in the synchronous resection group (P < 0.001), which was further identified by multivariate analysis as independent prognostic factor (P < 0.0001).38 In summary, these data suggest that patients with four or more liver metastases synchronous with primary colorectal cancer and metastasis infiltration of neighboring structures have to be carefully evaluated before resection, because they could have a dismal prognosis due to rapidly progressing disease. Primary Tumor Location Two strategies in the colon and rectal cancers have been revealed in an attempt to review the literature initially: probably a majority of simultaneous operations were done in patients with a colon primary tumor and conversely more delayed hepatectomies were done in patients with a rectal primary. As rectal surgical procedures have more morbidity than colonic surgery with more bleeding dissection, it would be expected, according to this hypothesis, that blood loss and hospital stay can have been increased in the rectal surgery and therefore in the delayed strategy. From our integrative analysis for the location of colorectal primary, Supporting figure 1, primary location section, has showed that right colon resection was 26% and 14% respectively in simultaneous and delayed hepatectomies, almost was twice as large as delayed group. In Vassiliou et al.’s series, right colon resection was more performed in simultaneous group than that in the delayed group (67% vs. 18%, P < 0.005).46 In Reddy et al.’s multi-centre study, right colectomy was also more often performed in simultaneous patients whereas more staged patients were treated with left colectomy and low anterior resection.27 It is a common sense that bilateral subcostal incision could provide excellent liver exposure and facilitate colon resection from the cecum up to the descending colon. In contrast, malignancies located in the sigmoid colon downward necessitate a midline incision and an extension to the right. Simultaneous resection is an acceptable option in patients with resectable synchronous colorectal metastasis by avoiding a second laparotomy, which could reduce the overall hospital stay, and with no difference in morbidity and mortality rates, based on the distribution that right hemicolectomy was more common in simultaneous group, whereas more left hemicolectomies performed in staged group in Martin et al.’s study published in 2009 (Although a subset analysis of patients according to location of primary tumor did not reveal a particular group of patients who are placed at unacceptably high risk by the performance of simultaneous colon and liver resection in their previous 2003 study.23).41 Further, rectal cancer was significantly more frequent in the staged resection group in Thelen et al.’s study, and in their own practice, major liver resections were considered only in combination with right or left colectomy, whereas simultaneous resection of rectal primaries were only performed with minor liver resections.42 In summary, simultaneous resection should be preferred for most of the right colon lesions as well as small liver lesions that can be approached from a midline incision, regardless of the colonic tumor location. Co-Morbidity Symptoms of colorectal cancer are numerous and nonspecific, usually not obvious within several years of disease progression, and may be variously presented according to where in the large intestine the tumor is located. Typically, right-sided cancers cause iron deficiency anemia due to the slow loss of blood over a long period of time, whereas cancers of the left colon are more likely to cause partial or complete bowel obstruction. In Thelen et al.’s series, simultaneous liver resections were generally not performed in patients with complications of the colorectal tumor such as bowel obstruction or colonic perforation.42 Further, for other co-existent conditions as severe coronary heart disease, reduced general constitution or severe chronic obstructive pulmonary disease, simultaneous resection was not the preference. In clinical practice, a lack of concern for the impact of coexistent diseases might have on the outcome of care is common, which is partly due to the lack of a validated, practical method for measuring these co-morbidities. In the present systematic review, a majority of included studies did not select patient preoperative co-morbidity as potential prognostic factor neither in univariate nor multivariate analysis. Thus for evaluating the effectiveness of timing selection for SCRLM, future clinical and health services research and audit should take comorbidity into account. In addition, blood transfusion was found to be significant on both univariate and logistic regression (odds ratio 2.98, P = 0.001) as a predictor of complication by Martin et al.41 In summary, the measurement of co-existent or comorbid conditions preoperatively should not be neglected. TNM Stages The most common staging system used for an estimate of the amount of penetration of colorectal cancer is the TNM (for tumors/nodes/metastases) system, from the American Joint Committee on Cancer. In simultaneous resection group, only the univariate analysis in Capussotti et al.’s had found T4 stage of primary colorectal tumor to be one of the five statistically significant prognostic factors in the synchronous resection group (P = 0.0216), which was further identified by multivariate analysis as independent prognostic factor (P = 0.0035).38 Alternatively, although concomitant extrahepatic disease was identified as one of the prognostic factors in the univariate analysis (P = 0.005) for both simultaneous and delayed resections, it was not an independent one showed by the multivariate analysis (P = 0.098) in de Haas et al.’s study.47 For the other studies, none of the analyzed staging parameters showed significant influence on survival after simultaneous hepatectomy. In summary, due to the fact that multiple surgeons prefer a simultaneous resection in patients with a less severe disease, the association between tumor staging and patient prognosis on the statuses how far the cancer has grown into the wall of the intestine, whether or not it has reached nearby structures, and whether or not it has spread to the lymph nodes or distant organs may be covered in the included studies. Neoadjuvant Chemotherapy Neoadjuvant chemotherapy prior to hepatectomy may facilitate the resectability of the liver lesions and treat occult metastasis but may also lead to hepatic parenchyma damage in patients with resectable colorectal liver metastases. Five studies reported the use of preoperative chemotherapy before resections. In de Hass et al.’s, Luo et al.’s and Reddy et al.’s studies, simultaneous patients were less often treated with chemotherapy before hepatic resection.27,47,49 This may explain the higher recurrence rate with the simultaneous resection strategy found in de Haas et al.’s study.47 However, of note was the observation that the drop-out in the delayed hepatectomy group of patients with progressive intrahepatic and/or extrahepatic disease after resection of the primary colorectal tumour may have selected a residual group with a more favourable prognosis, which may be the reason why preoperative chemotherapy was not an independent predictor of recurrence in de Haas et al.’s study. Generally, it is hypothesized that in delayed resection group, time-test strategy and neoadjuvant chemotherapy probably led to avoid surgery in patients with rapidly progressing disease and to perform hepatectomy in selected patients who could really benefit from surgery. A recently published systematic review of randomized and nonrandomized trials of the clinical response and outcomes of neoadjuvant systemic chemotherapy for 3728 patients with resectable colorectal liver metastases has demonstrated that, for objective radiological surveillance, a median of 4% (range 038%) and 52% (range 10-90%) of patients had complete and partial response respectively after neoadjuvant chemotherapy. Pathologically, a median of 9% (range 2-24%) and 36% (range 20-60%) had complete and partial response, respectively. Moreover, of patients, 41% (range 0-65%) had stable or progressive disease whilst on neoadjuvant chemotherapy. Median disease-free survival was 21 (range 11-40) months, and median overall survival was 46 (range 20-67) months.s23 In summary, there is argument over the oncologic benefit of neoadjuvant chemotherapy in patients who would already be suitable for a curative hepatectomy. Current evidence suggests that objective response to this practice may be achieved with improvement in disease-free survival in patients with resectable colorectal liver metastases, not overall survival. Review of the published prognostic scoring systems for colorectal liver metastases Hepatic resection provides the best chance for cure in patients with colorectal liver metastasis. Over the past decade, a variety of factors that might influence survival and recurrence have been identified and multiple prognostic scoring systems have been proposed. Relevant articles were reviewed from the published literature using the Pubmed and Embase databases. As shown from Supporting Table 4, a total of 7916 patients from 18 studies were identified, published between 1996 and 2011.s1-s18 Seven of these originated from European institutions, seven from Asian and four from North American centres. The number of patients per study ranged from 81 to 1568 with the median follow-up ranged from 16 to 54. Four were multi-centre studies, and five were prospective. Cox proportional hazards regression was used for multivariate analysis to identify the factors in all studies. Generally speaking, 25 independent prognostic factors were identified (Supporting Table 5). Of these, 11 were identified by two or more studies. Although there was a tendency towards the number and maximum size of metastases, lymph nodes spread from colorectal cancer, preoperative high CEA level and presence of extrahepatic disease as representing independent risk factors, the existing predictive models are diverse and there has no well-accepted ideal prognostic scoring system been established. Selection Criteria Establishment for Simultaneous Resection It is hypothesized that simultaneous resection should be associated with more prognostic factors to be found as to the overall procedures, because the surgical strategy is more drastic in simultaneous resection itself, whereas patients underwent various kinds of interventions to treat the liver metastases regardless of strategies applied when analysis is for the overall procedures. As shown by Supporting Figure 11, representative region for prognostic factors detected in overall resectable colorectal liver metastases should be smaller than that restricted in simultaneous group in SCRLM patients under the precondition of strictly balanced patient characteristics (as indicated by the area). In our study, a majority of patients included were in less severe diseases in simultaneous resection group compared to those in delayed group, thus some potential factors may not be found if review patients underwent simultaneous resection alone. Therefore, the real form of expression of distribution for the prognostic factors in simultaneous group should be smaller, and tilt to the less severe direction for risk assessment. Also, this hypothesis has been confirmed by this mini-systematic review and meta-analysis (Supporting Table 3). In addition, the Zone A without overlapping the representative region for overall resectable colorectal liver metastases was exclusive to simultaneous resection group; factors in it were viewed as the selection criteria for simultaneous patients directly against delayed resection. While the overall prognostic factors in Zone A+B+C were considered as the whole prognostic factors for CLM. Finally, based on the included studies and current published prognostic models, several factors were considered as the selection criteria for simultaneous resection directed against delayed resection: liver resection no more than 3 segments, colon resection (especially the right-sided colectomy), age less than 70 years old and coexistent severe conditions. These factors were exclusive to simultaneous resection group. From Supporting Figure 12, future large and well desired randomized controlled trials would be conducted under these selection criteria to conform the conclusions. Comment As demonstrated by the main meta-analysis of timing of hepatectomy for patients with synchronous liver metastases, long-term outcomes of overall survival and recurrence-free survival were similar between simultaneous and delayed groups; short-term outcome of postoperative morbidity was less in simultaneous group. Therefore, the safety of simultaneous resection was more considered when establishing the selection criteria. It was true that patients in simultaneous group had less severe disease compared with those in delayed group because of the nature of the included study types, but this was not the main concern in the present study. The most interesting result of the current study would be the establishment of selection criteria for patients who are really suitable for simultaneous resection, besides simply compare the safety and effectiveness of the two resection strategies. Limitation In this mini-systematic review and meta-analysis only observational studies were included, thus the main limitation was the selection bias for patients allocated to each group. Initially, a scoring-system was planned for SCRLM patients, but the applicability of the scoring system recommended by the current study for the postoperative risk assessment after simultaneous hepatectomy could not be validated. It is a common sense that the best method to establish the selection criteria should be based on the individual patient data analysis (IPD meta-analysis), however this is not always be available, and the diverse reporting forms from the included studies could reduce the reliability of the conclusion. Supporting References s1. Fong Y, Fortner J, Sun RL, Brennan MF, Blumgart LH. Clinical score for predicting recurrence after hepatic resection for metastatic colorectal cancer: analysis of 1001 consecutive cases. Ann Surg 1999;230:309-318. s2. Iwatsuki S, Dvorchik I, Madariaga JR, Marsh JW, Dodson F, Bonham AC, et al. Hepatic resection for metastatic colorectal adenocarcinoma: a proposal of a prognostic scoring system. J Am Coll Surg 1999;189:291-299. s3. Konopke R, Kersting S, Distler M, Dietrich J, Gastmeier J, Heller A, et al. Prognostic factors and evaluation of a clinical score for predicting survival after resection of colorectal liver metastases. Liver Int 2009;29:89-102. s4. Lee WS, Kim MJ, Yun SH, Chun HK, Lee WY, Kim SJ, et al. Risk factor stratification after simultaneous liver and colorectal resection for synchronous colorectal metastasis. Langenbecks Arch Surg 2008;393:13-19. s5. Lise M, Bacchetti S, Da Pian P, Nitti D, Pilati P. Patterns of recurrence after resection of colorectal liver metastases: prediction by models of outcome analysis. World J Surg 2001;25:638-644. s6. Malik HZ, Prasad KR, Halazun KJ, Aldoori A, Al-Mukhtar A, Gomez D, et al. Preoperative prognostic score for predicting survival after hepatic resection for colorectal liver metastases. Ann Surg 2007;246:806-814. s7. Minagawa M, Yamamoto J, Kosuge T, Matsuyama Y, Miyagawa S, Makuuchi M. Simplified staging system for predicting the prognosis of patients with resectable liver metastasis: development and validation. Arch Surg 2007;142:269-276. s8. Nagashima I, Takada T, Matsuda K, Adachi M, Nagawa H, Muto T et al. A new scoring system to classify patients with colorectal liver metastases: proposal of criteria to select candidates for hepatic resection. J Hepatobiliary Pancreat Surg 2004;11:79-83. s9. Nordlinger B, Guiguet M, Vaillant JC, Balladur P, Boudjema K, Bachellier P, et al. Surgical resection of colorectal carcinoma metastases to the liver. A prognostic scoring system to improve case selection, based on 1568 patients. Association Française de Chirurgie. Cancer 1996;77:1254-1262. s10. Rees M, Tekkis PP, Welsh FK, O’Rourke T, John TG. Evaluation of long-term survival after hepatic resection for metastatic colorectal cancer: a multifactorial model of 929 patients. Ann Surg 2008;247:125-135. s11. Schindl M, Wigmore SJ, Currie EJ, Laengle F, Garden OJ. Prognostic scoring in colorectal cancer liver metastases: development and validation. Arch Surg 2005;140:183-189. s12. Settmacher U, Dittmar Y, Knösel T, Schöne U, Heise M, Jandt K, AltendorfHofmann AK. Predictors of long-term survival in patients with colorectal liver metastases: a single center study and review of the literature. Int J Colorectal Dis 2011;26:967-981. s13. Tan MC, Castaldo ET, Gao F, et al. A prognostic system applicable to patients with resectable liver metastasis from colorectal carcinoma staged by positron emission tomography with [18F] fluoro-2-deoxy- D-glucose: role of primary tumor variables. J Am Coll Surg 2008;206:857-868. s14. Tanaka K, Shimada H, Fujii Y, et al. Pre-hepatectomy prognostic staging to determine treatment strategy for colorectal cancer metastases to the liver. Langenbecks Arch Surg 2004;389:371-379. s15. Ueno H, Mochizuki H, Hatsuse K, Hase K, Yamamoto T. Indicators for treatment strategies of colorectal liver metastases. Ann Surg 2000;231:59-66. s16. Wang Y, Liu YF, Cheng Y, Yi DH, Li P, Song WQ, Fu DZ, Wang X. Prognosis of colorectal cancer with liver metastasis: value of a prognostic index. Braz J Med Biol Res 2010;43:1116-1122. s17. Yamaguchi T, Mori T, Takahashi K, Matsumoto H, Miyamoto H, Kato T. A new classification system for liver metastases from colorectal cancer in Japanese multicenter analysis. Hepatogastroenterology 2008;55:173-178. s18. Zakaria S, Donohue JH, Que FG, Farnell MB, Schleck CD, Ilstrup DM, et al. Hepatic resection for colorectal metastases: value for risk scoring systems? Ann Surg 2007;246:183-191. s19. Adson MA. Resection of liver metastases—when is it worthwhile? World J Surg 1987;11:511–520. s20. Hohenberger P, Schlag PM, Gerneth T, Herfarth C. Pre- and postoperative carcinoembryonic antigen determinations in hepatic resection for colorectal metastases. Predictive value and implications for adjuvant treatment based on multivariate analysis. Ann Surg 1994;219:135-143. s21. Reddy SK, Barbas AS, Turley RS, Steel JL, Tsung A, Marsh JW, et al. A standard definition of major hepatectomy: resection of four or more liver segments. HPB (Oxford) 2011;13:494-502. s22. Bolton JS, Fuhrman GM. Survival after resection of multiple bilobar hepatic metastases from colorectal carcinoma. Ann Surg2000;231:743-751. s23. Chua TC, Saxena A, Liauw W, Kokandi A, Morris DL. Systematic review of randomized and nonrandomized trials of the clinical response and outcomes of neoadjuvant systemic chemotherapy for resectable colorectal liver metastases. Ann Surg Oncol 2010;17:492-501.