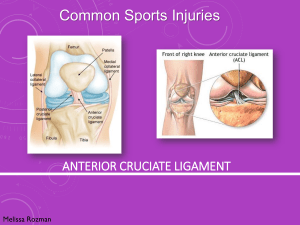
Comparison of Revision Rates in Autograft and Allograft Anterior
advertisement

1 Comparison of Revision Rates in Autograft and Allograft 2 Anterior Cruciate Ligament Reconstruction 3 4 Purpose: The purpose of this study is to compare the rates of revision after allograft and 5 autograft Anterior Cruciate Ligament (ACL) Reconstruction. 6 7 Methods: All ACL Reconstructions performed by a single surgeon (TSP) between 8 1/1/00 and 12/31/06 were identified by retrospective chart review. 231 patients met the 9 inclusion criteria and 181 patients were available for follow-up. 142 patients underwent 10 bone-patella tendon-bone autograft reconstruction (BTB-auto), 31 patients underwent 11 bone-patella tendon-bone allograft reconstruction (BTB-allo) and 8 patients underwent 12 Achilles tendon allograft reconstruction (Ach-allo). Patients were contacted to determine 13 whether their ACL Reconstruction had been revised. In addition, subjective IKDC and 14 Tegner scores were obtained. 15 16 Results: At a mean follow up of 49 months (11-91 months), revision rates were .7% 17 (1/142) in the BTB-auto group, 9.7% (3/31) in the BTB-allo group and 37.5% (3/8) in the 18 Ach-allo group. This difference reached statistical significance when comparing the 19 BTB-auto and BTB-allo groups (p=.02), as well as when comparing the BTB-auto and 20 Ach-allo groups (p=.0004). The difference in revision rates between the BTB-allo and 21 Ach-allo groups did not reach statistical significance (p=.09). Subjective IKDC scores 22 of non-revised (surviving) grafts in the BTB-auto group, BTB-allo group, and Ach-allo 1 23 group were 98.3, 95.2 and 86 respectively (p<.0001). Tegner scores of non-revised grafts 24 in the BTB-auto group, BTB-allo group, and Ach-allo group were 6.2, 6.5 and 5.6 25 respectively (p=.02). 26 27 Conclusions: ACL reconstruction with the use of bone-patella tendon-bone allografts 28 and Achilles tendon allografts is associated with a higher revision rate when compared to 29 bone-patella tendon autograft reconstruction. In addition, when comparing surviving 30 grafts, the subjective IKDC scores and Tegner scores are lower in the Achilles tendon 31 allograft group. 32 33 Clinical Relevance: Surgeons should be aware of the higher revision rate associated 34 with allograft ACL reconstruction when counseling patients on graft options. 35 36 Level of Evidence: III 37 38 Key Words: ACL, Allograft, Achilles, Revision, Autograft, Failure 39 40 41 42 43 44 45 2 46 Ruptures of the Anterior Cruciate Ligament (ACL) continue to be a common and 47 significant source of disability amongst athletes. Throughout the past century, great 48 strides have been made in our understanding of the ACL and our ability to reconstruct it 49 surgically. Most of this progress has come in the form of improved surgical techniques 50 resulting from the development and advancement of arthroscopic surgery as well as a 51 better understanding of the anatomy of the ACL. Surgeons continue to explore ways to 52 improve ACL reconstruction and allow athletes a less painful recovery with a quicker 53 return to function. Some have advocated the use of allografts to achieve these goals and 54 to avoid the peri-operative morbidity associated with the harvesting of autograft tissue. 55 56 Despite the obvious advantages of allografts, there are also many potential 57 disadvantages associated with their use. Allografts carry the risks of viral disease 58 transmission, bacterial contamination and immune reaction.1, 2 Outside of these risks 59 inherent to the use of cadaveric tissue, there is also some evidence that allografts take 60 longer to incorporate, revascularize and remodel than autograft tissue.3-9 This longer 61 period of incorporation and remodeling may, in turn, cause allografts to have weaker 62 structural properties than autografts and potentially lead to a higher rate of failure.10-12 It 63 currently remains unclear whether the potential benefits from the use of allografts in ACL 64 reconstruction outweigh the risks. This study seeks to shed light on this dilemma by 65 comparing the failure rates of autograft and allograft ACL reconstruction. Specifically, 66 we seek to determine and compare the rates of failure for both autograft and allograft 67 ACL reconstructions performed by a single surgeon. We hypothesize, that the failure 68 rate of allograft ACL reconstruction is higher than that for autograft ACL reconstruction. 3 69 70 71 METHODS 72 73 Patient Evaluation 74 75 All primary ACL reconstructions performed by a single surgeon (TSP) from 9/1/2000 to 76 5/31/07 were identified. 231 patients were identified of which 50 patients could not be 77 contacted and were considered lost to follow-up. Of the 181 patients remaining in the 78 study, 142 patients underwent bone-patella tendon-bone autograft reconstruction (BTB- 79 auto), 31 patients underwent bone-patella tendon-bone allograft reconstruction (BTB- 80 allo) and 8 patients underwent Achilles tendon allograft reconstruction (Ach-allo). 81 Patients were contacted to determine whether their ACL reconstruction had been revised. 82 In addition, subjective IKDC and Tegner scores were obtained. Demographic data for 83 the 3 groups was compared (Table 1). Mean follow-up for the groups was 49 months 84 (11-91 months). 85 86 Surgical Technique 87 88 Bone-patella tendon-bone autograft 89 A longitudinal incision was made over the medial aspect of the patella tendon exposing 90 the patella, patella tendon and tibial tubercle. A double-bladed #10 scalpel was used to 91 harvest a 10mm wide patella tendon graft. Patella and tibial bone plugs were harvested 4 92 with an oscillating saw at a length of 20-25 mm and a depth of 8-10 mm. Grafts were 93 prepared and fashioned on the back table while routine arthroscopy was performed along 94 with any concomitant meniscal or articular cartilage treatment. A femoral tunnel was 95 created at the 2 o’clock position (Left knee) or 10 o’clock position (Right knee). The 96 femoral tunnel was reamed to a depth to match the patella bone plug (20-25 mm) and 97 reamed to a diameter to accommodate the patella bone plug (9-10mm). The patella bone 98 plug was then advanced into place and secured with a 7 X 23 mm bioabsorbable 99 interference screw (Arthrex, Naples, FL). The tibial bone plug was then fixed with the 100 knee in 0-10 degrees of flexion using a 9 X 23 or 10 X 23 mm bioabsorbable interference 101 screw (Arthrex, Naples, FL). 102 103 Bone-patella tendon-bone allograft 104 Allografts were inspected and thawed to room temperature in a warm saline bath. They 105 were subsequently prepared and secured with identical technique to the bone-patella-bone 106 autograft group described above. All of the grafts were fresh frozen and 14 of the 31 107 grafts (45%) had undergone low dose irradiation (.8-1.5 MRad). 108 109 Achilles tendon allograft 110 Allografts were inspected and thawed to room temperature in a warm saline bath. The 111 calcaneal bone block was prepared to a length of 20-25mm, a width of 10 mm and a 112 depth of 8-10 mm. A double-bladed #10 scalpel was used to prepare the tendon to a 113 width of 10 mm. The soft tissue end of the grafts was whip-stitched with #2 non- 114 absorbable suture. The graft was fixed to the femoral tunnel with a 7 X 23 mm 5 115 bioabsorbable interference screw (Arthrex, Naples, FL) and the tendinous portion of the 116 graft was fixed to the tibial tunnel with a 10mm bioabsorbable delta taper screw (Arthrex, 117 Naples, FL). Graft fixation was performed with the knee in 0-10 degrees of flexion. All 118 of the Achilles grafts were fresh frozen and had undergone low dose irradiation (.8-1.5 119 MRad). 120 121 Rehabilitation 122 123 Immediate weight bearing as tolerated was allowed with crutches to be used for comfort. 124 All knees were placed in a hinged knee brace locked in extension until sufficient 125 quadriceps control was demonstrated. Early rehabilitation focused on regaining motion 126 and incorporated mostly closed chained exercises. Sports specific activity was 127 introduced at 4 months and a full return to sports was allowed at 6 months if sufficient 128 strength and one leg hop were demonstrated. All three groups underwent the same 129 rehabilitation protocol. 130 131 Statistical Methods 132 133 Revision rates between the three groups were compared using the Fisher test. A series of 134 one way ANOVA tests were used to compare IKDC scores and Tegner scores between 135 the groups. Statistical significance was set at p < .05. Tukey post-hoc analysis was 136 performed to determine specific differences between groups (GraphPad Prism Software, 137 version 5.01). 6 138 139 140 RESULTS 141 142 Revision Rates (Figure 1) 143 144 The primary endpoint of this study was the performance of revision ACL surgery. At a 145 mean follow up of 49 months (11-91 months), revision rates were .7% (1/142) in the 146 BTB-auto group, 9.7% (3/31) in the BTB-allo group and 37.5% (3/8) in the Ach-allo 147 group. This difference reached statistical significance when comparing the BTB-auto and 148 BTB-allo groups (p=.02), as well as when comparing the BTB-auto and Ach-allo groups 149 (p=.0004). The difference in revision rates between the BTB-allo and Ach-allo groups 150 did not reach statistical significance (p=.09). The single revised graft in the BTB-auto 151 group represented an acute traumatic re-rupture. Two of the 3 revisions in the BTB-allo 152 group represented acute traumatic re-ruptures while the other was a graft that was 153 symptomatically lax without any acute event. One of the 3 revisions in the Ach-allo 154 groups represented an acute traumatic re-rupture while the other two were lax grafts 155 resulting in instability without any particular acute event. 156 157 Outcome Scores (Figures 2 and 3) 158 159 Subjective IKDC scores of non-revised (surviving) grafts in the BTB-auto group, BTB- 160 allo group, and Ach-allo group were 98.3, 95.2 and 86 respectively (Table 2). 7 161 Statistically significant differences were found between all three groups (p<.0001). 162 Tegner scores of non-revised grafts in the BTB-auto group, BTB-allo group, and Ach- 163 allo group were 6.2, 6.5 and 5.6 respectively (Table 2). A statistically significant 164 difference group difference was found and Tukey post-hoc analysis demonstrated a 165 difference between the BTB-allo and Ach-allo groups (p=.02). 166 167 DISCUSSION 168 169 The use of allografts during ACL reconstruction remains a popular choice amongst 170 patients and surgeons. There are clearly many advantages to using an allograft: 171 decreased morbidity from tissue harvest, less pain and improved cosmesis.2, 13, 14 172 However, there are also many potential disadvantages: risk of disease transmission, cost, 173 immune reaction and slower incorporation of the graft.1, 2, 9, 11, 15-21 174 175 Concern over slower incorporation, revascularization and remodeling of allograft 176 tissue has been well documented in the orthopaedic literature.22 Jackson et al. performed 177 allograft ACL reconstruction on 11 goats using freeze dried bone-ACL-bone allograft. 178 At 1 year the grafts demonstrated histological properties of a normal ligament and 179 complete incorporation of the bone blocks. However, they also demonstrated inferior 180 biomechanical properties versus the native ACL.21 Zhang et al. performed bilateral ACL 181 reconstructions on dogs, with one knee receiving an allograft and the other an autograft. 182 They found inferior incorporation in the allograft group at 6 months post-operatively.23 A 183 recent sheep retrieval study also demonstrated delayed remodeling and inferior 8 184 biomechanical properties when comparing allografts to autografts at 1 year post- 185 operatively.16 There are few human studies addressing the concerns over incorporation, 186 remodeling and revascularization of allograft ACLs. Muramatsu et al. examined the MRI 187 characteristics of allograft versus autograft bone-patella tendon-bone ACL reconstruction 188 in humans. They found a slower onset and rate of revascularization of the allografts 189 when compared to the autografts.24 Malinin et al. examined ACL allografts that were 190 retrieved at the time of future arthroplasty.9 They found that at two years post- 191 implantation, the central portions of the graft remained acellular and complete 192 incorporation did not occur. If allografts do not incorporate and remodel as well as 193 autografts, they may be more susceptible to re-rupture during physical therapy or when 194 patients return to physical activity. There have been several reports of increased failure 195 rates with the use of allograft tissue during ACL reconstruction.12, 25, 26 Most of these 196 reports are case series or case reports. The data is somewhat conflicting as there are also 197 many studies demonstrating equal rates of success with the use of allografts.3, 27-30 This 198 study represents an attempt to compare a single surgeon’s experience and results with 199 both autograft and allograft ACL reconstruction using similar fixation techniques. As 200 such, it provides particularly useful insight into the ongoing controversy surrounding the 201 use of allografts. 202 203 In this study we find that the use of allografts during ACL reconstruction is 204 associated with a higher risk of revision ACL surgery. This was found to be true with 205 both bone-patella tendon-bone allografts and Achilles tendon allografts. The reason 206 behind the higher rate of revision surgery was not the subject of this study and remains 9 207 unclear. Certainly, many possibilities exist. The slower rate of incorporation, 208 remodeling and revascularization of the allograft tissue may lead to a higher rate of re- 209 rupture as many authors have previously speculated. While this seems like a logical 210 explanation, other possibilities must be considered. The sterilization techniques used 211 (e.g. irradiation) may affect the structural properties of the allograft tissue thereby 212 causing more failures.31-34 Alternatively, it may be that patients feel better soon after 213 surgery allowing them to rehab more aggressively than desirable and leading to failure. 214 Differences in fixation may also account for some of these failures. This would not 215 explain the differences found in this study between the BTB-auto and BTB-allo groups 216 but could potentially be a cause of increased failures in the Ach-allo group. Inferior 217 fixation associated with Achilles tendon grafts has been clearly demonstrated.35 While 218 the exact etiology of the difference in failure rates between autograft and allograft ACL 219 reconstruction remains unclear, it appears relatively certain from this study that there is a 220 lower incidence of revision ACL reconstruction when autograft tissue is used. 221 222 This study has several weaknesses. This is a retrospective study and as such is 223 subject to the weaknesses and potential bias that accompanies any retrospective study. In 224 addition, the demographics of the 3 groups (BTB-auto, BTB-allo, Ach-allo) were not 225 identical. While the 3 groups had equivalent pre-operative IKDC and Tegner scores, they 226 were of different ages. The Ach-allo group represented a population that was 227 significantly younger than the other groups. This was due to the author’s preference to 228 use Achilles allografts on patients with open physes. This is a potential source of bias 229 and must be considered. However, the findings of increased IKDC and Tegner scores 10 230 and a lower revision rate was also found when comparing the BTB-auto and BTB-allo 231 groups, which have comparable ages. With these weaknesses in mind, this study also has 232 several strengths. It compares the results of a single surgeon and thereby eliminates the 233 problems of comparing results of allografts done by one surgeon with the results of 234 autografts done by another surgeon. Also, the techniques and fixation used in the BTB- 235 auto and BTB-allo groups are identical, allowing a fair direct comparison. Lastly, this 236 study involves a large sample size and thereby allows small differences in revision rates 237 and outcome scores to be detected. 238 239 Despite the findings of this study, it is unwise to abandon the use of allograft 240 tissue during ACL reconstruction. There may be instances when allograft tissue is a 241 better option than the use of autograft. There are certainly instances in revision and 242 multi-ligament surgery where standard autograft options may not be available. It is also 243 still the authors’ practice to use allograft tissue in patients who have limited athletic 244 aspirations and desire a quick return to their daily activities. In other words, allografts 245 may be a reasonable option for the middle-aged executive who desires to ski several 246 times a year, but a less desirable option for the competitive college basketball player with 247 professional aspirations. 248 249 In summary, we have demonstrated a higher rate of revision ACL reconstruction 250 associated with the use of allograft during the primary ACL reconstruction. Surgeons 251 should be aware of these findings and counsel patients appropriately when discussing 252 graft options prior to ACL reconstruction. 11 253 254 255 256 257 258 259 260 261 262 263 264 265 266 267 268 269 270 271 272 273 274 275 276 277 278 279 280 281 282 283 284 285 286 287 288 289 290 291 292 293 294 295 296 297 REFERENCES 1. Cohen SB, Sekiya JK. Allograft safety in anterior cruciate ligament reconstruction. Clin Sports Med 2007;26:597-605. 2. Strickland SM, MacGillivray JD, Warren RF. Anterior cruciate ligament reconstruction with allograft tendons. Orthop Clin North Am 2003;34:41-7. 3. Indelli PF, Dillingham MF, Fanton GS, Schurman DJ. Anterior cruciate ligament reconstruction using cryopreserved allografts. Clin Orthop Relat Res 2004:268-75. 4. Zhang CC, Zhou JS, Pan GP. [Ultrastructure of anterior cruciate ligament after transplantation]. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi 2003;17:370-3. 5. Gorschewsky O, Browa A, Vogel U, Stauffer E. [Clinico-histologic comparison of allogenic and autologous bone-tendon-bone using one-third of the patellar tendon in reconstruction of the anterior cruciate ligament]. Unfallchirurg 2002;105:703-14. 6. Lomasney LM, Tonino PM, Coan MR. Evaluation of bone incorporation of patellar tendon autografts and allografts for ACL reconstruction using CT. Orthopedics 2007;30:152-7. 7. Lee CA, Meyer JV, Shilt JS, Poehling GG. Allograft maturation in anterior cruciate ligament reconstruction. Arthroscopy 2004;20 Suppl 2:46-9. 8. Goertzen MJ, Buitkamp J, Clahsen H, Mollmann M. Cell survival following bone-anterior cruciate ligament-bone allograft transplantation: DNA fingerprints, segregation, and collagen morphological analysis of multiple markers in the canine model. Arch Orthop Trauma Surg 1998;117:208-14. 9. Malinin TI, Levitt RL, Bashore C, Temple HT, Mnaymneh W. A study of retrieved allografts used to replace anterior cruciate ligaments. Arthroscopy 2002;18:16370. 10. Siebold R, Buelow JU, Bos L, Ellermann A. Primary ACL reconstruction with fresh-frozen patellar versus Achilles tendon allografts. Arch Orthop Trauma Surg 2003;123:180-5. 11. Grafe MW, Kurzweil PR. Anterior cruciate ligament reconstruction with Achilles tendon allografts in revisions and in patients older than 30. Am J Orthop 2008;37:302-8. 12. Singhal MC, Gardiner JR, Johnson DL. Failure of primary anterior cruciate ligament surgery using anterior tibialis allograft. Arthroscopy 2007;23:469-75. 13. Fuchs R, Wheatley W, Uribe JW, Hechtman KS, Zvijac JE, Schurhoff MR. Intraarticular anterior cruciate ligament reconstruction using patellar tendon allograft in the skeletally immature patient. Arthroscopy 2002;18:824-8. 14. Sadovsky P, Musil D, Stehlik J. [Allograft for surgical reconstruction of the cruciate ligaments of the knee - part 1]. Acta Chir Orthop Traumatol Cech 2005;72:2936. 15. Kaminski A, Gut G, Marowska J, Lada-Kozlowska M, Biwejnis W, Zasacka M. Mechanical properties of radiation-sterilised human Bone-Tendon-Bone grafts preserved by different methods. Cell Tissue Bank 2008. 16. Scheffler SU, Schmidt T, Gangey I, Dustmann M, Unterhauser F, Weiler A. Fresh-frozen free-tendon allografts versus autografts in anterior cruciate ligament reconstruction: delayed remodeling and inferior mechanical function during long-term healing in sheep. Arthroscopy 2008;24:448-58. 12 298 299 300 301 302 303 304 305 306 307 308 309 310 311 312 313 314 315 316 317 318 319 320 321 322 323 324 325 326 327 328 329 330 331 332 333 334 335 336 337 338 339 340 341 17. Menetrey J, Duthon VB, Laumonier T, Fritschy D. "Biological failure" of the anterior cruciate ligament graft. Knee Surg Sports Traumatol Arthrosc 2008;16:224-31. 18. Ozer H, Selek HY, Turanli S, Atik SO. Failure of primary ACL surgery using anterior tibialis allograft via transtibial technique. Arthroscopy 2007;23:1026. 19. Victor J, Bellemans J, Witvrouw E, Govaers K, Fabry G. Graft selection in anterior cruciate ligament reconstruction--prospective analysis of patellar tendon autografts compared with allografts. Int Orthop 1997;21:93-7. 20. Vasseur PB, Stevenson S, Gregory CR, et al. Anterior cruciate ligament allograft transplantation in dogs. Clin Orthop Relat Res 1991:295-304. 21. Jackson DW, Grood ES, Arnoczky SP, Butler DL, Simon TM. Freeze dried anterior cruciate ligament allografts. Preliminary studies in a goat model. Am J Sports Med 1987;15:295-303. 22. Dustmann M, Schmidt T, Gangey I, Unterhauser FN, Weiler A, Scheffler SU. The extracellular remodeling of free-soft-tissue autografts and allografts for reconstruction of the anterior cruciate ligament: a comparison study in a sheep model. Knee Surg Sports Traumatol Arthrosc 2008;16:360-9. 23. Zhang CL, Fan HB, Xu H, Li QH, Guo L. Histological comparison of fate of ligamentous insertion after reconstruction of anterior cruciate ligament: autograft vs allograft. Chin J Traumatol 2006;9:72-6. 24. Muramatsu K, Hachiya Y, Izawa H. Serial evaluation of human anterior cruciate ligament grafts by contrast-enhanced magnetic resonance imaging: comparison of allografts and autografts. Arthroscopy 2008;24:1038-44. 25. Krych AJ, Jackson JD, Hoskin TL, Dahm DL. A meta-analysis of patellar tendon autograft versus patellar tendon allograft in anterior cruciate ligament reconstruction. Arthroscopy 2008;24:292-8. 26. Baer GS, Harner CD. Clinical outcomes of allograft versus autograft in anterior cruciate ligament reconstruction. Clin Sports Med 2007;26:661-81. 27. Poehling GG, Curl WW, Lee CA, et al. Analysis of outcomes of anterior cruciate ligament repair with 5-year follow-up: allograft versus autograft. Arthroscopy 2005;21:774-85. 28. Sun K, Tang JW, Xu Q, et al. [A prospective study of the anterior cruciate ligament reconstruction: allograft versus autograft]. Zhonghua Wai Ke Za Zhi 2004;42:989-92. 29. Kustos T, Balint L, Than P, Bardos T. Comparative study of autograft or allograft in primary anterior cruciate ligament reconstruction. Int Orthop 2004;28:290-3. 30. Nyland J, Caborn DN, Rothbauer J, Kocabey Y, Couch J. Two-year outcomes following ACL reconstruction with allograft tibialis anterior tendons: a retrospective study. Knee Surg Sports Traumatol Arthrosc 2003;11:212-8. 31. Goertzen MJ, Clahsen H, Burrig KF, Schulitz KP. Sterilisation of canine anterior cruciate allografts by gamma irradiation in argon. Mechanical and neurohistological properties retained one year after transplantation. J Bone Joint Surg Br 1995;77:205-12. 32. Goertzen MJ, Clahsen H, Schulitz KP. Anterior cruciate ligament reconstruction using cryopreserved irradiated bone-ACL-bone-allograft transplants. Knee Surg Sports Traumatol Arthrosc 1994;2:150-7. 13 342 343 344 345 346 347 348 349 350 351 352 353 354 355 356 357 358 359 360 361 362 363 364 365 366 367 368 369 370 371 372 373 374 375 376 377 378 379 380 381 382 383 384 385 386 387 388 33. Rappe M, Horodyski M, Meister K, Indelicato PA. Nonirradiated versus irradiated Achilles allograft: in vivo failure comparison. Am J Sports Med 2007;35:16538. 34. Rihn JA, Irrgang JJ, Chhabra A, Fu FH, Harner CD. Does irradiation affect the clinical outcome of patellar tendon allograft ACL reconstruction? Knee Surg Sports Traumatol Arthrosc 2006;14:885-96. 35. Fogel H PD, Verma N, Bach B Jr, Wang V, Provencher M. Biomechanical Analysis of Soft Tissue ACL Tibial Fixation: Comparison of One-,Two-, and Four-Stranded Allograft Constructs. AOSSM Annual Meeting 2008. 14 Table 1 Mean Age Female (years) Male (%) (%) BTB-auto 30.2* 74 (51%) 71 (49%) BTB-allo 35.5* 17 (55%) 14 (45%) Ach-allo 16.6* 4 (50%) 4 (50%) *Differences in mean age reached statistical significance between all groups 389 390 391 392 Table 1: Patient demographics by type of graft Table 2 IKDC post pre post BTB-auto 43.9 98.3 2.2 6.2 BTB-allo 42.7 95.2 2.5 6.5 Ach-allo 44.1 86.0 2.2 5.6 0.08 0.02* p value 0.62 <.0001* *Denotes statistically significant difference 393 394 Tegner pre c Table 2: Subjective IKDC and Tegner scores pre and post ACL reconstruction 395 396 397 Figure 1: Revision Rates 15 398 399 Figure 2: Subjective IKDC Scores 400 401 Figure 3: Tegner Scores 16