Document
advertisement

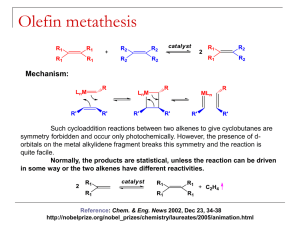
NEW CATALYST SYSTEMS FOR Z-SELECTIVE OLEFIN METATHESIS Alicia Phelps Schomaker Group UW-Madison Literature Seminar March 15, 2012 1 What is Olefin Metathesis? 2005 Nobel Prize in Chemistry awarded for “the development of the metathesis method in organic synthesis". Yves Chauvin Robert Grubbs Vougioukalakis, G.; Grubbs, R. Chem. Rev. 2010, 110, 1746. Richard Schrock 2 Types of Olefin Metathesis Ring Opening Metathesis Polymerization (ROMP) Ring Closing Metathesis (RCM) Cross Metathesis (CM) 3 Vougioukalakis, G.; Grubbs, R. Chem. Rev. 2010, 110, 1746. Mechanism of Olefin Metathesis • Reversible at each step • Equilibrium mixture of products Thermodynamic conditions favor E product! 4 Vougioukalakis, G.; Grubbs, R. Chem. Rev. 2010, 110, 1746 Two Classes of Catalysts Grubbs 1st Generation Schrock Alkylidene Hoveyda-Grubbs 2nd Generation Grubbs 1st Generation Schrock, R. R.; Murdzek, J. S.; Bazan, G. C.; Robbins, J.; DiMare, M.; O’Regan, M. B. J. Am. Chem. Soc. 1990, 112, 3875. Trnka, T. M.; Grubbs, R. H. Acc. Chem. Res. 2001, 34, 18. 5 Other Approaches to Z Olefins • Classic Olefinations • Wittig Reaction (non-stabilized ylides) • Cross-coupling reactions • Peterson Olefination • Partial Hydrogenation of Alkynes • Removable directing groups Fürstner, A.; Mathes, C.; Lehmann, C. Chem. Eur. J. 2001, 7, 5299. Wang, Y.; Jimenez, M.; Hansen, A.; Raiber, E.; Schreiber, S.; Young, D. J. Am. Chem. Soc. 2011, 133, 9196. 6 Substrate Controlled Z Selectivity Sattely, E.; Cortez, G.; Moebius, D.; Schrock, R.; Hoveyda, A. J. Am. Chem. Soc. 2005, 127, 8526. Crowe, W.; Goldberg, D. J. Am. Chem. Soc. 1995, 117, 5162. Hansen, E.; Lee, D. Org. Lett. 2004, 6, 2035. 7 Catalyst Screening X= dr cat A/B/C (%) yield (%) er Z/E Cl 3.0:1 56:22:22 80 95:5 >98:2 Br 2.2:1 62:08:30 85 98:2 >98:2 I 1.7:1 67:04:29 60 >98:2 95:5 F N/A 07:47:46 57 95:5 80:20 Ibrahem, I; Yu, M.; Schrock, R.; Hoveyda, A. J. Am. Chem. Soc. 2009, 131, 3844. 8 Mode of Selectivity • Steric bulk of aryloxide as compared to imido forces reaction to go through all-syn metallocyclobutane Ibrahem, I.; Yu, M.; Schrock, R.; Hoveyda, A. J. Am. Chem. Soc. 2009, 131, 3844. 9 Substrate Scope of ROCM Ar Olefin equiv. mol% cat Time (h) Yield (%) er Z/E p-OMeC6H4 2 1 0.5 80 97:3 95:5 p-CF3C6H4 2 1 1 67 98:2 >98:2 o-BrC6H4 10 2 1 50 99:1 89:11 o-MeC6H4 10 2 1 54 99:1 88:12 10 Ibrahem, I; Yu, M.; Schrock, R.; Hoveyda, A. J. Am. Chem. Soc. 2009, 131, 3844. Z-Selective ROMP and CM • Able to isolate 99% cis, 99% syndiotactic polymers of dicarbomethoxynorbornadiene • Catalyst screens showed “small” imido group is not required for terminal substrates and W catalysts deliver higher % Z product compared to Mo catalysts Flook, M.; Jiang, A.; Schrock, R.; Müeller, P.; Hoveyda, A. J. Am. Chem. Soc. 2009, 131, 7962. Jiang, A.; Zhao, Y.; Schrock, R.; Hoveyda, A. J. Am. Chem. Soc. 2009, 131, 16630. 11 Tungsten Catalysts R n time %conv %cis Me 3 4h 74 >99 Me 5 4d 89 >99 CO2Me 8 2d 45 >99 Ph 1 24 h 35 97 SiMe3 1 4h 45 81 1 2.5 h 6 92 First example of Z-selective metathesis in the presence of E olefins 12 Marinescu, S.; Schrock, R.; Müeller, P.; Takase, M.; Hoveyda, A. Organometallics 2011, 30, 1780. Cross-Metathesis – Enol Ethers Catalyst Time Conv (%) Z:E 1a 2h 85 98:2 1b 2h 47 >98:2 2 2h 37 >98:2 3 2h <2 NA 4 10 min 80 48:52 5 24 h <2 NA • No previous CM of enol ethers reported Meek, S.; O’Brien, R.; Llaveria, J.; Schrock, R.; Hoveyda, A. Nature 2011, 471, 461. 13 Cross Metathesis System Design VS. • Mo-methylidene can cause equilibration • Alkoxy-substituted alkylidene is less reactive • Conclusion: use excess enol ether 14 Meek, S.; O’Brien, R.; Llaveria, J.; Schrock, R.; Hoveyda, A. Nature 2011, 471, 461. Cross-Metathesis – Enol Ethers Meek, S.; O’Brien, R.; Llaveria, J.; Schrock, R.; Hoveyda, A. Nature 2011, 471, 461. 15 Ring Closing Metathesis Cat Mol% cat Conc. Pressure Time (h) Yield (%) Z:E 1 5.0 1.0 mM ambient 16 96 34:66 2 10.0 1.0 mM 1.0 torr 3.0 57 64:36 3 10.0 1.0 mM ambient 1.5 87 85:15 3 10.0 1.0 mM 1.0 torr 1.5 91 90:10 4 10.0 1.0 mM 1.0 torr 2.5 97 96:4 4 3.0 0.05 M 1.0 torr 3.0 97 97:3 4 7.5* 6.0 mM 0.02 torr 4.0 96 94:6 * Catalyst weighed out in air Yu, M.; Wang, C.; Kyle, A.; Jakubec, P.; Dixon, D.; Schrock, R.; Hoveyda, A. Nature 2011, 479, 88. 16 Enol Ethers in ROCM • Enol ethers formed are electronically differentiated, unlike previous reactions with aryl substrates Yu, M.; Ibrahem, I.; Hasegawa, M.; Schrock, R.; Hoveyda, A. J. Am. Chem. Soc. 2012, 134, 2788. 17 Summary of Mo/W Catalysis • Molybdenum and tungsten catalysts can perform olefin metathesis with very high Z selectivity • Ring opening cross metathesis • Ring opening metathesis polymerization • Ring closing metathesis • Cross metathesis • Z-selectivity controlled by steric interaction with aryloxide ligand 18 Ruthenium Catalysis – Catalyst Design • Intramolecular C-H activation of NHC ligand yields Z- selective catalysts Endo, K.; Grubbs, R. J. Am. Chem. Soc. 2011, 133, 8525. 19 Ruthenium Catalyst - Results Cat mol% cat Solvent Temp (°C) Time (min) Yield 1 (%) Z/E 1 Yield 2 (%) Z/E 2 A 2.5 C6H6 23 60 57 31:69 3 17:83 B 5.0 C6H6 70 120 36 88:12 26 94:6 B 5.0 THF Reflux 240 60 81:19 31 96:4 B 5.0 THF/H2O Reflux 240 64 86:14 29 97:3 C 2.5 C6H6 23 30 66 9:91 10 15:85 D 2.5 C6H6 23 30 <1 34:66 NA NA Endo, K.; Grubbs, R. J. Am. Chem. Soc. 2011, 133, 8525. 20 Homodimerization with Ruthenium Substrate Time (h) Yield (%) Z:E Allylbenzene 1 81 92:8 Methyl undecenoate 5.5 >95 73:27 Allyl acetate 4 62 89:11 1-octene 4 79 83:17 Allyl trimethylsilane 3 54 >95:5 Allyl pinacol borane 4 74 >95:5 4-penten-1-ol 1 72 72:28 2-(allyloxy)ethanol 1 73 66:34 N-allylaniline 2 67 71:29 21 Keitz, B.; Endo, K.; Herbert, M.; Grubbs, R. J. Am. Chem. Soc. 2011, 133, 9686. Screening for Improved Ru Catalysts • Adamantyl group is critical for selectivity • Monodentate ligands have no CM reactivity • Nitrato groups were more active than carboxylate groups • Changes to aryl group on NHC did not affect reactivity 22 Keitz, B.; Endo, K.; Patel, P.; Herbert, M; Grubbs, R. J. Am. Chem. Soc. 2012, 134, 693. Improved Ru Catalysts Substrate Time (h) %Z Yield (%) Methyl undecenoate 12 91 85 1-octene 12 92 83 4-penten-1-ol 12 81 67 Allyl trimethylsilane 9 >95 14 Allyl pinacol borane 3 >95 36 Allyl acetate 12 >95 8 2-(allyloxy)ethanol 12 67 30 N-allylaniline 12 90 12 • Z selectivity highest at low conversions • Yield poor in comparison to previous catalyst Keitz, B.; Endo, K.; Patel, P.; Herbert, M; Grubbs, R. J. Am. Chem. Soc. 2012, 134, 693. 23 Ruthenium Catalysis in ROMP Monomer Catalyst % cis Yield (%) 1 2 58 88 88 94 1 2 <5 75 93 88 1 2 81 91 95 78 1 2 78 61 95 40 • High cis selectivity, but no long-range tacticity 24 Keitz, B.; Federov, A.; Grubbs, R. J. Am. Chem. Soc. 2012, 134, 2040. Selectivity of Ruthenium Catalysts • Free energy calculations show side-bound pathway favored with Z- selective catalysts by 3.1 kcal/mol (B3LYP/LANL2DZ-6-31G(d)) 25 Liu, P.; Xu, X.; Dong, X.; Keitz, B.; Herbert, M.; Grubbs, R.; Houk, K. J. Am. Chem. Soc. 2012, 134, 1464. Transition States 26 Liu, P.; Xu, X.; Dong, X.; Keitz, B.; Herbert, M.; Grubbs, R.; Houk, K. J. Am. Chem. Soc. 2012, 134, 1464. Transition States Side-bound transition state d → π* (NHC) d → π* (alkylidene) Bottom-bound transition state Backdonation to NHC and alkylidene involves same Ru d orbital 27 Liu, P.; Xu, X.; Dong, X.; Keitz, B.; Herbert, M.; Grubbs, R.; Houk, K. J. Am. Chem. Soc. 2012, 134, 1464. Transition States – E/Z Selectivity ΔG‡ = 16.1 kcal/mol ΔG‡ = 14.6 kcal/mol ΔG‡ = 18.8 kcal/mol ΔG‡ = 14.4 kcal/mol • Steric interaction favors Z selectivity (B3LYP/LANL2DZ-6- 31G(d)) Liu, P.; Xu, X.; Dong, X.; Keitz, B.; Herbert, M.; Grubbs, R.; Houk, K. J. Am. Chem. Soc. 2012, 134, 1464. 28 Summary of Ru Catalysis • Ru catalysts perform homodimerization and ROMP with comparable selectivity to Mo/W catalysts • Catalyst system is more functional group and moisture tolerant than Mo/W systems • Z-selectivity controlled by side bound metallocyclobutane transition state 29 Nakadomarin A Nilson, M.; Funk, R. Org. Lett. 2010, 12, 4912. Yu, M.; Wang, C.; Kyle, A.; Jakubec, P.; Dixon, D.; Schrock, R.; Hoveyda, A. Nature 2011, 479, 88. 30 Conclusions • High Z selectivity in olefin metathesis, previously unattainable, has been observed • Both molybdenum/tungsten and ruthenium catalyst systems have been developed to accomplish major classes of olefin metathesis • Methods have already been applied to total synthesis of epothilone C and nakadomarin A Future Work: Metathesis of tri-and tetra-substituted alkenes Moisture and air tolerant catalyst systems 31 Acknowledgements • Prof. Jennifer Schomaker • Kat Myhre • Schomaker Group • Practice Talk Attendees • Rob Risi • Alex Clemens • Jackie Brown • Dr. Doris Pun • Janelle Steves • Megan Cismesia • Dr. Dan Wherritt 32 Synthesis of C18(plasm)-16:0(PC) Meek, S.; O’Brien, R.; Llaveria, J.; Schrock, R.; Hoveyda, A. Nature 2011, 471, 461. 33 Synthesis of KRN7000 34 Meek, S.; O’Brien, R.; Llaveria, J.; Schrock, R.; Hoveyda, A. Nature 2011, 471, 461. Mechanism of Ruthenium Catalysts • Side bound pathway (blue) is energetically favored over bottom bound pathway (green) 35 Transition State Computational Models 36 NBO Models 37 NBO Models 38 Mechanism of Mo Catalysts • Both diastereomers of catalyst 1 were isolated and crystal structures obtained 39 Meek, S.; Malcomson, S.; Li, B.; Schrock, R.; Hoveyda, A. J. Am. Chem. Soc. 2009, 131, 16407. Mechanism of Schrock Catalysts Mechanistic Observations: • R-cat promotes reaction slowly, but with same sense of enantioinduction • Initial enantioselectivity is low but increases during the reaction Cat. Time (min) Conv (%) er (S:R) S-1 2 4 76:24 S-1 2.5 7 87.5:12.5 S-1 3 13 93.5:6.5 S-1 30 >98 96.5:3.5 R-1 30 5 85.5:14.5 R-1 45 15 94:6 R-1 60 23 95:5 R-1 180 >98 96:4 40 Meek, S.; Malcomson, S.; Li, B.; Schrock, R.; Hoveyda, A. J. Am. Chem. Soc. 2009, 131, 16407 Mechanism of Schrock Catalysts • Reaction requires 3 metathesis steps to get to product • Should give net inversion of Mo, lowering er 41 Meek, S.; Malcomson, S.; Li, B.; Schrock, R.; Hoveyda, A. J. Am. Chem. Soc. 2009, 131, 16407 Mechanism of Schrock Catalysts • Mechanism without off-cycle isomerization Problems: • R center in 2nd step should react slowly • Other enantiomer of product is observed Meek, S.; Malcomson, S.; Li, B.; Schrock, R.; Hoveyda, A. J. Am. Chem. Soc. 2009, 131, 16407 42 Mechanism of Mo Catalysts Mechanism of degenerate ethylene isomerization • Under steady-state conditions, inversion of metal center via degenerate metathesis with ethylene is faster than product conversion • Stereochemistry is independent of the starting alkylidene Meek, S.; Malcomson, S.; Li, B.; Schrock, R.; Hoveyda, A. J. Am. Chem. Soc. 2009, 131, 16407. 43 Synthesis of Natural Products - CM Meek, S.; O’Brien, R.; Llaveria, J.; Schrock, R.; Hoveyda, A. Nature 2011, 471, 461. 44 Cross-Metathesis – Allylic Amides Meek, S.; O’Brien, R.; Llaveria, J.; Schrock, R.; Hoveyda, A. Nature 2011, 471, 461. 45