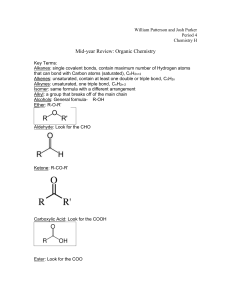
Problema 3 Essential ozone Some hydrocarbon

www.pianetachimica.it
Problema 3 Essential ozone
Some hydrocarbon C
10
H
16
participates in the transformations given in Scheme 1.
1. Determine the structural formulae of the hydrocarbon C
10
H
16
and the molecules A–D accounting for the fact that compounds C and D are isomers of the initial hydrocarbon; the ozonolysis of C followed by the treatment of the reaction mixture with alkaline H
2
O
2
produces a single product while the same transformations of D afford two compounds.
Solution
O OH
1) O
3
2) Zn/H +
EtONa
EtOH
EtONa
EtOH
O
A
O O
C
10
H
16
OH
1) O
3
2) NaBH
4
H
2
SO
4
+
OH
C
10
H
16
B C D
Some other hydrocarbon E (
ω
C
= 90.6%) under ozonolysis (1. O
3
, CH
2
Cl
2
, –78 o
C; 2. Me
2
S) forms three carbonyl compounds – F (C
2
H
2
O
2
), G (C
3
H
4
O
2
), and H (C
4
H
6
O
2
) in a ratio of 3:2:1. Initial hydrocarbon E doesn’t decolorize bromine water.
2. Write down the structural formulae of hydrocarbon E and products of its ozonolysis F – H .
Solution
Hydrocarbon E has 90,6% of C, 9.4% of H. Then its empirical formula is C
4
H
5
. Hydrocarbon E is aromatic because it doesn’t decolorize bromine water, so it is C
8
H
10
ortho-xilene.
O O O O O O
1) O
3
C C C C C C
2) Me
2
S
H H H H H
3
C CH
3
E F F H
E
1) O
3
2) Me
2
S
O
C C
O
H H
F
O
C C
O
H CH
3
G
O
C
H
G
C
O
CH
3
47 IChO 2015 Baku – Azerbaijan Soluzioni preliminari dei problemi preparatori 1
www.pianetachimica.it
Hydrocarbon I having center of symmetry was used as an initial material in the total synthesis of pentalenene (Scheme 2):
The ozonolysis of hydrocarbon I furnishes a single compound P or Q depending on the treatment of the ozonolysis product. Under treatment with I
2 and NaOH, compound Q forms a yellow precipitate containing 96.7% of iodine. Under basic conditions compound Q is transformed into compound R containing 4 types of hydrogen atoms (4 signals in
1
H NMR spectrum with integral intensity of signals
1:1:2:2). Molecular formula of R is C
5
H
6
O. Molecule of compound N has bicyclic framework containing R as a fragment. Molecule of O consists of three rings.
3. Descript the scheme of the synthesis of pentalenene .
Solution
P and Q differ for a single oxygen atom then Q is ketone and aldehyde and Q is ketone and acid.
Q is a methylketone because it gives the iodoform reaction. Q and P are then:
O O
H OH
O O
P
O
OH
−
O
H
O
Q
Assembling P and Q we obtain I. There are two possibilities:
OH
Q
OH
−
O
R
But only the first one is correct because it has a center of symmetry.
Then the synthesis of pentalenene is as follows:
47 IChO 2015 Baku – Azerbaijan Soluzioni preliminari dei problemi preparatori 2
I
1) 9-BBN
2) H
2
O
2
/OH
−
O
O
2
PdCl
2
CuCl
J
L
O
BF
3
-OEt
2
HCOOH +
N
Soluzione proposta da
Mauro Tonellato - ITI Marconi - Padova
M
O
OH
PCC
O
O
NaH
THF
O
K www.pianetachimica.it
O
1) LiN(SiMe
3
)
2
2) CH
2
=CHCH
2
Br
O
OH
NaH
THF
Al(CH
3
)
3
O pentalenene
47 IChO 2015 Baku – Azerbaijan Soluzioni preliminari dei problemi preparatori 3