Synthesis and Antibacterial Activity of Isoindoline
advertisement
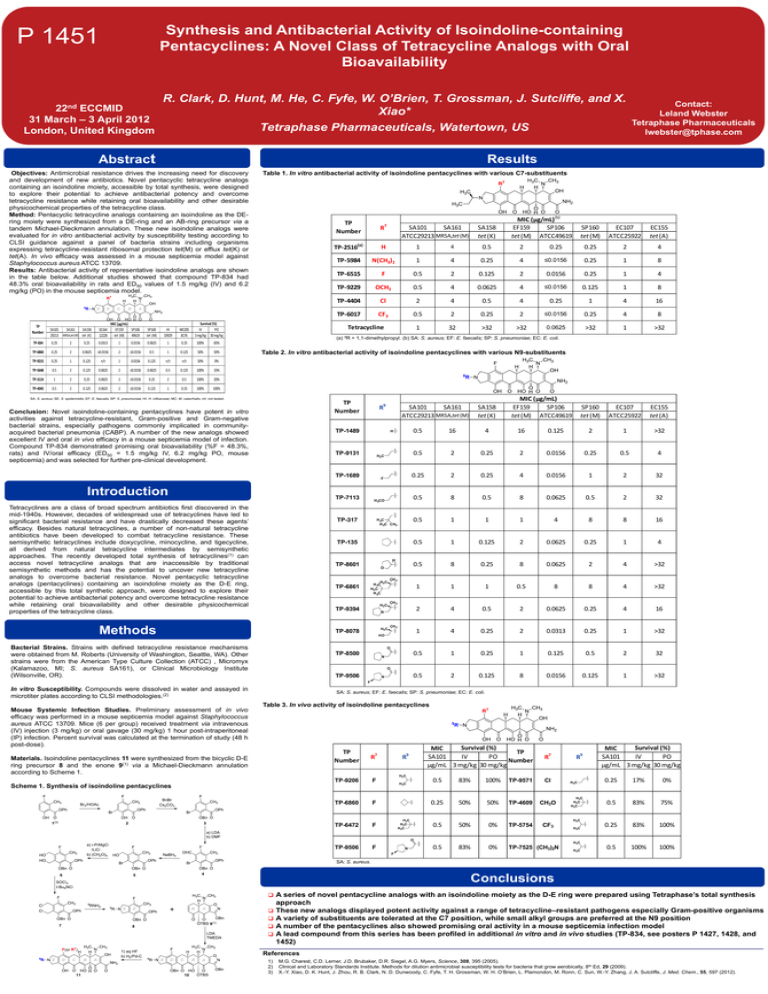
Synthesis and Antibacterial Activity of Isoindoline-containing Pentacyclines: A Novel Class of Tetracycline Analogs with Oral Bioavailability P 1451 R. Clark, D. Hunt, M. He, C. Fyfe, W. O’Brien, T. Grossman, J. Sutcliffe, and X. 22nd ECCMID Xiao* 31 March – 3 April 2012 Tetraphase Pharmaceuticals, Watertown, US London, United Kingdom Abstract Results Objectives: Antimicrobial resistance drives the increasing need for discovery and development of new antibiotics. Novel pentacyclic tetracycline analogs containing an isoindoline moiety, accessible by total synthesis, were designed to explore their potential to achieve antibacterial potency and overcome tetracycline resistance while retaining oral bioavailability and other desirable physicochemical properties of the tetracycline class. Method: Pentacyclic tetracycline analogs containing an isoindoline as the DEring moiety were synthesized from a DE-ring and an AB-ring precursor via a tandem Michael-Dieckmann annulation. These new isoindoline analogs were evaluated for in vitro antibacterial activity by susceptibility testing according to CLSI guidance against a panel of bacteria strains including organisms expressing tetracycline-resistant ribosomal protection tet(M) or efflux tet(K) or tet(A). In vivo efficacy was assessed in a mouse septicemia model against Staphylococcus aureus ATCC 13709. Results: Antibacterial activity of representative isoindoline analogs are shown in the table below. Additional studies showed that compound TP-834 had 48.3% oral bioavailability in rats and ED50 values of 1.5 mg/kg (IV) and 6.2 mg/kg (PO) in the mouse septicemia model. Table 1. In vitro antibacterial activity of isoindoline pentacyclines with various C7-substituents Survival (%) IV PO 3 mg/kg 30 mg/kg SA101 SA161 SA158 29213 MRSA,tet(M) tet (K) MIC (µg/mL) SE164 EF159 SP106 12228 tet (M) 49619 SP160 tet (M) HI 33929 MC205 8176 TP-834 0.25 2 0.25 0.0313 1 0.0156 0.0625 1 0.25 100% 83% TP-6860 0.25 2 0.0625 ≤0.0156 2 ≤0.0156 0.5 1 0.125 50% 50% TP-9215 0.25 1 0.125 n/t 1 0.0156 0.125 n/t n/t 50% 0% TP-5648 0.5 2 0.125 0.0625 2 ≤0.0156 0.0625 0.5 0.125 100% 33% TP-3114 1 2 0.25 0.0625 2 ≤0.0156 0.25 2 0.5 100% 20% TP-4045 0.5 2 0.125 0.0625 2 ≤0.0156 0.125 1 0.25 100% 100% TP Number Contact: Leland Webster Tetraphase Pharmaceuticals lwebster@tphase.com MIC (µg/mL)(b) EF159 SP106 tet (M) ATCC49619 TP Number R7 TP-2516(a) H 1 4 0.5 2 TP-5984 N(CH3)2 1 4 0.25 TP-6515 F 0.5 2 TP-9229 OCH3 0.5 TP-4404 Cl TP-6017 CF3 SA101 SA161 ATCC29213 MRSA,tet (M) SP160 tet (M) EC107 ATCC25922 EC155 tet (A) 0.25 0.25 2 4 4 ≤0.0156 0.25 1 8 0.125 2 0.0156 0.25 1 4 4 0.0625 4 ≤0.0156 0.125 1 8 2 4 0.5 4 0.25 1 4 16 0.5 2 0.25 2 ≤0.0156 0.25 4 8 1 32 >32 >32 0.0625 >32 1 >32 SP160 tet (M) EC107 ATCC25922 EC155 tet (A) Tetracycline SA158 tet (K) (a) 9R = 1,1-dimethylpropyl. (b) SA: S. aureus; EF: E. faecalis; SP: S. pneumoniae; EC: E. coli. Table 2. In vitro antibacterial activity of isoindoline pentacyclines with various N9-substituents SA: S. aureus; SE: S. epidermidis; EF: E. faecalis; SP: S. pneumoniae; HI: H. influenzae; MC: M. catarrhalis; n/t: not tested. TP Number Conclusion: Novel isoindoline-containing pentacyclines have potent in vitro activities against tetracycline-resistant, Gram-positive and Gram-negative bacterial strains, especially pathogens commonly implicated in communityacquired bacterial pneumonia (CABP). A number of the new analogs showed excellent IV and oral in vivo efficacy in a mouse septicemia model of infection. Compound TP-834 demonstrated promising oral bioavailability (%F = 48.3%, rats) and IV/oral efficacy (ED50 = 1.5 mg/kg IV, 6.2 mg/kg PO, mouse septicemia) and was selected for further pre-clinical development. R9 SA101 SA161 ATCC29213 MRSA,tet (M) TP-1489 H TP-9131 H3 C TP-1689 Introduction F TP-7113 Tetracyclines are a class of broad spectrum antibiotics first discovered in the mid-1940s. However, decades of widespread use of tetracyclines have led to significant bacterial resistance and have drastically decreased these agents’ efficacy. Besides natural tetracyclines, a number of non-natural tetracycline antibiotics have been developed to combat tetracycline resistance. These semisynthetic tetracyclines include doxycycline, minocycline, and tigecycline, all derived from natural tetracycline intermediates by semisynthetic approaches. The recently developed total synthesis of tetracyclines(1) can access novel tetracycline analogs that are inaccessible by traditional semisynthetic methods and has the potential to uncover new tetracycline analogs to overcome bacterial resistance. Novel pentacyclic tetracycline analogs (pentacyclines) containing an isoindoline moiety as the D-E ring, accessible by this total synthetic approach, were designed to explore their potential to achieve antibacterial potency and overcome tetracycline resistance while retaining oral bioavailability and other desirable physicochemical properties of the tetracycline class. H3CO TP-317 H 3C H 3C CH3 TP-135 H TP-8601 O H C H 3C 3 H3 C H 3C TP-6861 H3 C TP-9394 Methods 0.5 16 4 16 0.125 2 1 >32 0.5 2 0.25 2 0.0156 0.25 0.5 4 0.25 2 0.25 4 0.0156 1 2 32 0.5 8 0.5 8 0.0625 0.5 2 32 0.5 1 1 1 4 8 8 16 0.5 1 0.125 2 0.0625 0.25 1 4 0.5 8 0.25 8 0.0625 2 4 >32 1 1 1 0.5 8 8 4 >32 2 4 0.5 2 0.0625 0.25 4 16 1 4 0.25 2 0.0313 0.25 1 >32 0.5 1 0.25 1 0.125 0.5 2 32 0.5 2 0.125 8 0.0156 0.125 1 >32 CH 3 CH 3 N H3 C TP-8078 CH 3 HO Bacterial Strains. Strains with defined tetracycline resistance mechanisms were obtained from M. Roberts (University of Washington, Seattle, WA). Other strains were from the American Type Culture Collection (ATCC) , Micromyx (Kalamazoo, MI; S. aureus SA161), or Clinical Microbiology Institute (Wilsonville, OR). SA158 tet (K) MIC (µg/mL) EF159 SP106 tet (M) ATCC49619 O TP-8500 N O TP-9506 N F In vitro Susceptibility. Compounds were dissolved in water and assayed in microtiter plates according to CLSI methodologies.(2) Mouse Systemic Infection Studies. Preliminary assessment of in vivo efficacy was performed in a mouse septicemia model against Staphylococcus aureus ATCC 13709. Mice (6 per group) received treatment via intravenous (IV) injection (3 mg/kg) or oral gavage (30 mg/kg) 1 hour post-intraperitoneal (IP) infection. Percent survival was calculated at the termination of study (48 h post-dose). SA: S. aureus; EF: E. faecalis; SP: S. pneumoniae; EC: E. coli. Table 3. In vivo activity of isoindoline pentacyclines Time Kill Assays Materials. Isoindoline pentacyclines 11 were synthesized from the bicyclic D-E ring precursor 8 and the enone 9(1) via a Michael-Dieckmann annulation according to Scheme 1. Survival (%) MIC TP SA101 IV PO Number µg/mL 3 mg/kg 30 mg/kg Survival (%) MIC SA101 IV PO µg/mL 3 mg/kg 30 mg/kg TP Number R7 TP-9206 F 0.5 83% 100% TP-9571 Cl 0.25 17% 0% TP-6860 F 0.25 50% 50% TP-4609 CH3O 0.5 83% 75% TP-6472 F 0.5 50% 0% TP-5754 CF3 0.25 83% 100% 0.5 83% 0% TP-7525 (CH3)2N 0.5 100% 100% R9 R7 R9 Scheme 1. Synthesis of isoindoline pentacyclines O TP-9506 F N F SA: S. aureus. Conclusions A series of novel pentacycline analogs with an isoindoline moiety as the D-E ring were prepared using Tetraphase’s total synthesis approach These new analogs displayed potent activity against a range of tetracycline–resistant pathogens especially Gram-positive organisms A variety of substituents are tolerated at the C7 position, while small alkyl groups are preferred at the N9 position A number of the pentacyclines also showed promising oral activity in a mouse septicemia infection model A lead compound from this series has been profiled in additional in vitro and in vivo studies (TP-834, see posters P 1427, 1428, and 1452) References 1) 2) 3) M.G. Charest, C.D. Lerner, J.D. Brubaker, D.R. Siegel, A.G. Myers, Science, 308, 395 (2005). Clinical and Laboratory Standards Institute. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically, 8th Ed, 29 (2009). X.-Y. Xiao, D. K. Hunt, J. Zhou, R. B. Clark, N. D. Dunwoody, C. Fyfe, T. H. Grossman, W. H. O’Brien, L. Plamondon, M. Ronn, C. Sun, W.-Y. Zhang, J. A. Sutcliffe, J. Med. Chem., 55, 597 (2012).