6.3 Electrolysing brine
advertisement

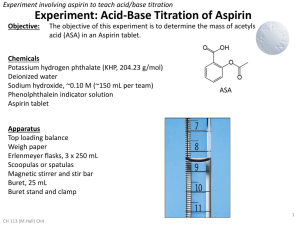
6.3 Electrolysing brine Brine is salty water. It’s chemical name is sodium chloride, NaClaq. Both elements are useful in thousands of products. 13 April 2015 6.3 Electrolysing brine 13 April 2015 • Brine can be electrolysed into – Sodium Hydroxide (NaOH) – Chlorine (Cl) – Hydrogen (H2). • Some of the uses of the products of the electrolysis of brine are; – – – – Soap (NaOH) PVC (Cl) Bleach (NaOH + Cl) Margerine (H2) Diaphragm cell What type of ion does chlorine form? What type of ion for hydrogen? Sodium is more reactive than Hydrogen so does not gather at the cathode. Wear Eye Protection. Practical Caution: Sodium Hydroxide Chlorine Method 1. Set up the apparatus as shown. 2. Write down your observations as shown below. Diagram 6V Results Electrode Anode (+) Cathode (-) Observations Beaker with saturated brine and Universal Indicator Questions 1. Name the gas given off at each electrode. 2. What are the four ions present in the brine? 3. Which ions remain in the solution? 4. What compound would they form? Answers 1. Name the gas given off at each electrode. • Anode = Chlorine, Cathode = Hydrogen 2. What are the four ions present in the brine? • Sodium+, Chloride-, Oxide2-, Hydrogen+ 3. Which ions remain in the solution? • Sodium+, Oxide2-, Hydrogen+ 4. What compound would they form? • Sodium Hydroxide (NaOH) Uses of electrolysed brine Brine produces 3 main products. Which products are used for; NaOH H Cl 1. 2. 3. 4. 5. 6. 7. 8. 9. Margarine PVC Bleach Soap Paper Rayon fibres Detergents Purification of aluminium ore Hydrochloric acid manufacture 2 Uses of electrolysed brine 1. 2. 3. 4. 5. 6. 7. 8. 9. Margarine [hydrogen] PVC [chlorine] Bleach [sodium hydroxide, chlorine] Soap [sodium hydroxide] Paper [chlorine] Rayon fibres [sodium hydroxide] Detergents [sodium hydroxide, chlorine] Purification of aluminium ore [sodium hydroxide] Hydrochloric acid manufacture [chlorine, hydrogen] 6.3 Electrolysing brine 13 April 2015 • Brine can be electrolysed into – Sodium Hydroxide (NaOH) – Chlorine (Cl) – Hydrogen (H2). • Some of the uses of the products of the electrolysis of brine are; – – – – Soap (NaOH) PVC (Cl) Bleach (NaOH + Cl) Margerine (H2)