Polymers: Biomedical Applications
advertisement

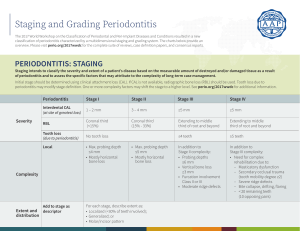
Polymers: Biomedical Applications Bryan Orellana September 16, 2010 Where can you find uses for polymers? • Everywhere! – There seems to be endless uses for polymers • Why? – – – – – – Easier to produce Biocompatibility Often cheaper Designed to mimic Replacement to old practices Designed to prevent additional surgery/trauma to patient http://www.sharecourseware.org/%28A%28a0C4BeKFywEkAAAAMWYxNTBiZjYtNTUwZS00N2MzLTlkNjQtY2NlOGM2Njc0MDBh8_JtaPeKutpFEQQVUOeBje 6glDI1%29%29/ShowLecture.aspx?ID=454&CatID=1&SubCatID=35 Polymers in Drug Delivery • Pill coatings • Rapid dissolving capsules – Fluid driven – pH driven • Degradable delivery vesicles in composite systems http://www.pharmainfo.net/raghanaveen/parenteral-controlled-drug-delivery-system http://www.flickr.com/photos/vancelucas/galleries/72157622318592067 A11/1.4 Poly β-Amino Ester Hydrogel • Purpose: – Used as a degradable carrying vessel for bioactive molecules • Macromer: – Synthesized by a condensation reaction between Diethylene Glycol Diacrylate and 3-Morpholinopropyl-amine at 85°C for 16hrs – Diacrylate : Amine ratio: 2.079gDiacrylate gAmine Hydrogel Polymerization • Hydrogel polymer produced from a mixture of macromer and DMPA (2,2-dimethoxy-2-phenyl acetophenone), a free radical initiator dissolved in ethanol • Solution sandwiched between glass plates (1.5mm thick) • Photo-polymerized with UV radiation for 5min • Polymerized gel is washed in ethanol and dried • Gel is grounded down to obtain particles (average diameters of 53 - 150 µm and 150-250 µm) Photo-polymerization • DMPA cleaves when exposed to UV energy producing free radical complexes • Excitation causes complexes to bombard with macromer • Double bonds break and binds with either another radical terminating the process or another polymer chain causing a further chain reaction http://www.usm.maine.edu/~newton/Chy251_253/Lectures/Free%20Radicals/RadicalPolymerization.GIF Composite Formation • Composite consisted of Calcium Sulfate (structural support) and varying wt. percentage (1% and 10%) of All/1.4 poly(β-amino ester) hydrogel particles (delivery component) • Mixtures are mixed with DI water and placed into a mold. • Samples are allowed to dry in a 43°C oven for 24hrs • Sample dimensions: – Diameter ~4.7mm – Height ~6.5mm μ-CT 3-D Image Construction • Images were contoured and analyzed to produce 3dimensional images • These images allowed for determination of hydrogel distribution The Problem: Oral Deficiencies • Oral bone deficiencies are a major Issue! – High prevalence of periodontitis – Injury/trauma to jaw bone or teeth – Other deficiencies from birth defects (i.e. cleft palate/lip) http://www.ohiohealth.com/mayo/images/image_popup/fl7_cleft_palate.jpg http://www.rad.washington.edu/staticpix/mskbook/MandibleFx.gif The Solution: Bone Augmentation • Performed to increase the amount of bone to allow for secure implant placement • Common procedures: 1) 2) Alveolar process augmentation of mandible and/or maxilla Maxillary sinus augmentation http://www.dr.agravat.com/images/bone_grafting02.jpg http://www.drleonedds.com/images/photos/bonegraft1.jpg http://davidhan.info/yahoo_site_admin/assets/images/bf-Picture4.242141428_std.jpg Problem with bone graft augmentation Donor site morbidity http://scottfross.com/wp-content/uploads/2009/12/bone-graft1.jpg Bone Augmenation Example Courtesy of Scott Price, D.M.D. Two weeks post-op Courtesy of Scott Price, D.M.D. 7 months post-op Courtesy of Scott Price, D.M.D. Conclusion • Seemingly endless possibilities with new advances in polymer chemistry and medicine • Being a patient conscience field, new polymer innovations have helped make these individuals’ lives easier What to Remember for Final Exam!! • What polymerization process was used to produce the Poly(β-amino ester) hydrogel? Give a brief explanation of how the process works. – Photo-polymerization • DMPA (Photo Initiator) cleaves when exposed to UV energy producing free radical complexes • Excitation causes complexes to bombard with macromer • Double bonds break and binds with either another radical terminating the process or another polymer chain causing a further chain reaction References • Borrell, Luisa N., and Natalie D. Crawford. "Social Disparities in Periodontitis Among United States Adults 1999-2004." Community Dent Oral Epidemiol 36 (2008): 383-91. • Chapple, Iain L.C., Mike R. Milward, and Thomas Dietrich. "The Prevalence of Inflammatory Periodontitis Is Negatively Associated with Serum Antioxidant Concentrations." Journal of Nutrition 137 (2007): 657-64. • Karadag, Erdener, and Dursun Saraydm. "Swelling Studies of Super Water Retainer Acrylamide/crotonic Acid Hydrogels Crosslinked by Trimethylolpropane Triacrylate and 1,4-butanediol Dimethacrylate." Polymer Bulletin 48 (2002): 299-307. • Tonetti, Maurizio S., and Andrea Mombelli. "Early-Onset Periodontitis." Ann Periodontol 4.1 (1999): 39-52.