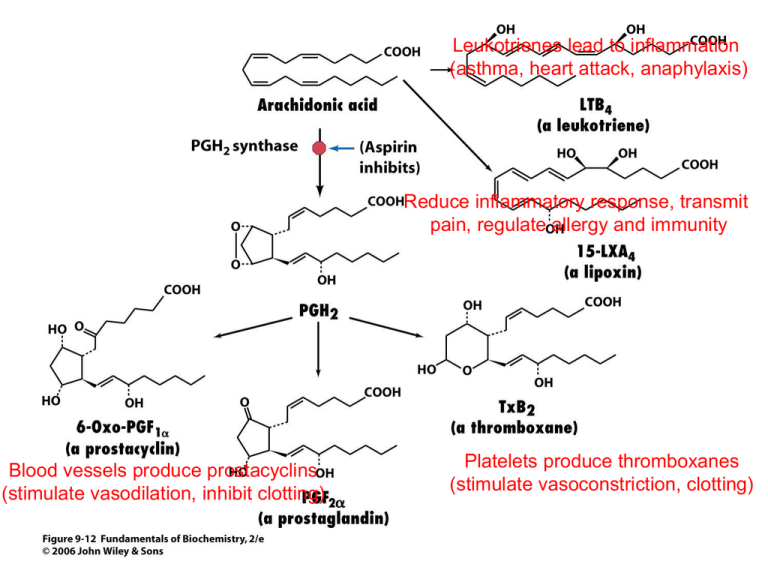
Leukotrienes lead to inflammation
(asthma, heart attack, anaphylaxis)
Reduce inflammatory response, transmit
pain, regulate allergy and immunity
Blood vessels produce prostacyclins
(stimulate vasodilation, inhibit clotting)
Platelets produce thromboxanes
(stimulate vasoconstriction, clotting)
1
Arachidonic acid
• Eicosanoid (C20) precursor from dietary
essential polyunsaturated fatty acids (linoleic
acid)
• Stored as C2-ester of phospholipids (released
by phospholipase A2)
• Hormone-like molecules, decompose within
seconds or minutes (have very local effects)
• Pain, fever, coagulation, blood pressure, and
reproduction
2
Prostaglandins
• Prostaglandin H2 synthase forms a
cyclopentane ring in linear arachidonic
acid
• Enzyme has 2 catalytic activities:
– Cyclooxygenase (adds two O2)
– Peroxidase (converts OOH group to OH)
• Commonly called COX
• Aspirin is an irreversible inhibitor of COX
(inactivates enzyme by acetylating active
site Ser and blocking active site from
reacting with substrate
Ser-ÖH
– Analgesic, antipyretic, anti-inflammatory
3
COX
• NSAIDs (NonSteroidal Anti-Inflammatory
Drugs)
• Acetaminophen and ibuprofen are
noncovalent inhibitors of COX
• COX-1 and COX-2 isoforms (60% identical)
• COX-2 inhibitors lack side effects of other NSAIDS
– COX-1 expressed ubiquitously, maintains homeostasis
– COX-2 expressed in certain tissues during inflammation
response and is responsible for elevated prostaglandin levels
• Aspirin and ibuprofen are nonspecific NSAIDs, have side
4
effects (gastrointestinal ulceration)
COX-2
•Structure-based drug design of selective COX-2 inhibitors
•COX-2 active site ~20% larger than COX-1 (make a bigger
inhibitor that cannot fit into COX-1 site)
COX
inhibitor
•Rofecoxib (Vioxx) popular and effective, but withdrawn due to
unanticipated cardiac side effects (mechanism may involve inhibition of
prostacyclin synthesis (leaving thromboxane synthesis less affected)
•Acetaminophen effective, but low affinity for COX-1,2 (binds
COX-3, which is expressed in the central nervous system)
5
Drug Design
• Drug Discovery: How?
Screening large numbers of compounds for
inhibition of enzymatic activity or receptor
signaling
Measure KI(or KI’)
Good lead compound has KI < 1mM
Why is high affinity necessary?
-specificity!!!!!
-dose!!!!!!!
Design related compounds using combinatorial
chemical techniques
6
Structure-based
design
• X-ray, NMR
• What does the active site
look like empty and with
the substrate in it?
• Look at structural and electrostatic properties of
active site and try to better fit/fill it.
For a candidate…
• Quantum mechanical calculation of charge
distribution
• Docking simulations
• Determine structure of complex, revise inhibitor
structure and re-assay KI
7
Bioavailability & Toxicity
To cause desired response:
– Drug must arrive at high enough concentration
– Drug must arrive to the location of the target protein
Oral drugs (cheapest)
– Acid-stable (stomach)
– Membrane-permeable (gut-blood transfer, so can’t be highly charged))
– Don’t bind tightly to other things (lipophilic drugs sequestered in
membrane/adipocytes)
– Survive detoxifying enzymes in the liver (portal vein drains intestine
directly to liver)
– Avoid rapid excretion by kidneys
– Must pass from capillaries to tissues
– (for brain) must pass blood-brain barrier, which blocks polar substances
– (for intracellular protein) must pass plasma (and other) membrane(s)
Protein drugs poor oral drugs (acid, pepsin, trypsin, immune system,
etc.)
8
Bioavailability & Toxicity
How drug interacts with barriers is
pharmacokinetics (Absorption, Distribution,
Metabolism, Excretion measurements)
Bioavailability (extent to which it reaches proper
site) depends on dose and pharmacokinetics
Best drugs a compromise: not too polar or
lipophilic, neutral at pH 6-8 (pass through
membrane in uncharged state)
Drugs with low KI for target are likely to be more
specific and have fewer side effects
9
Clinical trials
After in vitro (test tube) and in vivo (animal) studies:
Phase I: test safety, dosage range and method (20-100
healthy volunteers, or if toxic drug then test very sick
people)
Phase II: efficacy against target disease (100-500 volunteer
patients). Refine dosage, check for side effects. Singleblind tests (docs know, patients don’t)
Control substance is a placebo (ethical caveat)
Phase III: Monitor adverse effects from long-term use,
confirm efficacy (1000-5000 patients) through statistical
analysis of double-blind, placebo-controlled tests
(double blind removes bias from subjective judgments of
investigators…you see what you want to see…)
10
Clinical trials
Few drug candidates survive preclinical testing
(~5/5000, ~3 years)
Clinical trials 7-10 years, most fail in Phase II
~$500 million to bring drug to market!!!
Most difficult issue is identifying rare side effects
(1/10000) and long-term effects
Out of Control, by Celia Farber (HIV clinical trial
article… holiday reading)
http://www.harpers.org/archive/2006/03/0080961
11
P450s
12
Cytochromes P450
Well-tolerated drugs can be dangerous for
others…
Genetic differences among individuals
Different disease state
Other drugs
Sex
Age
Environmental factors
13
Cytochromes P450
Detoxify xenobiotics (embedded in ER membrane)
Superfamily of heme-containing enzymes in nearly all living
organisms
Fe(II), CO-bound enzyme absorbs at 450nm
Humans have ~100 isozymes (isoforms)
Monooxygenases (Fe undergoes reversible redox-state
change during catalytic cycle)
RH + O2 + 2H+ + 2e- ROH + H2O
e- from NADPH to the P450 heme via cytochrome P450
reductase
Oxidize lipophilic compounds for conjugation to glucuronic
acid or sulfate.
14












