Principles of Bioinorganic Chemistry
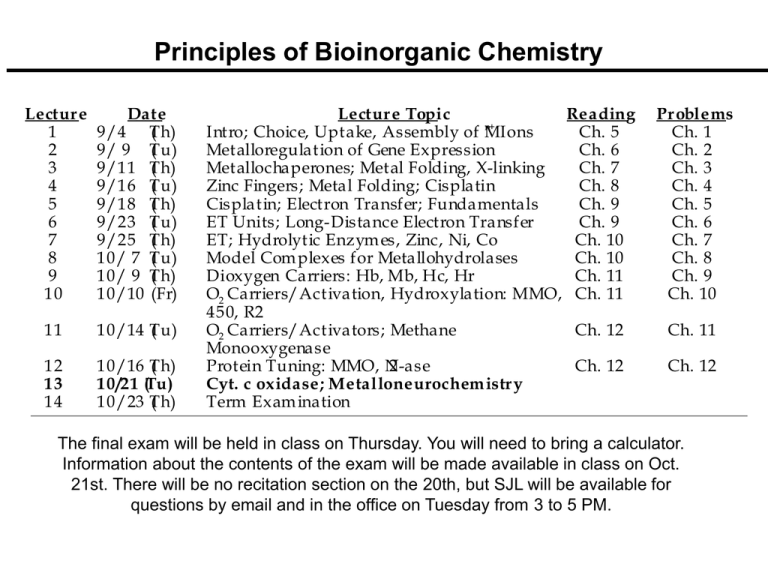
Lectur e
1
2
3
4
5
6
7
8
9
10
Date
9/4 T
( h)
9/ 9 T
( u)
9/11 T
( h)
9/16 T
( u)
9/18 T
( h)
9/23 T
( u)
9/25 T
( h)
10/ 7 T
( u)
10/ 9 T
( h)
10/10 (Fr)
11
10/14 T
( u)
12
13
14
10/16 T
( h)
10/21 (Tu)
10/23 T
( h)
Lectur e Topic
Reading
n+
Intro; Choice, Uptake, Assembly of MIons
Ch. 5
Metalloregulation of Gene Expression
Ch. 6
Metallochaperones; Metal Folding, X-linking
Ch. 7
Zinc Fingers; Metal Folding; Cisplatin
Ch. 8
Cisplatin; Electron Transfer; Fundamentals
Ch. 9
ET Units; Long-Distance Electron Transfer
Ch. 9
ET; Hydrolytic Enzym es, Zinc, Ni, Co
Ch. 10
Model Com plexes for Metallohydrolases
Ch. 10
Dioxygen Carriers: Hb, Mb, Hc, Hr
Ch. 11
O2 Carriers/Activation, Hydroxylation: MMO, PCh. 11
450, R2
O2 Carriers/Activators; Methane
Ch. 12
Monooxygenase
Protein Tuning: MMO, N
2-ase
Ch. 12
Cyt. c oxidase; Metalloneurochem istr y
Term Exam ination
Pr oblems
Ch. 1
Ch. 2
Ch. 3
Ch. 4
Ch. 5
Ch. 6
Ch. 7
Ch. 8
Ch. 9
Ch. 10
Ch. 11
Ch. 12
The final exam will be held in class on Thursday. You will need to bring a calculator.
Information about the contents of the exam will be made available in class on Oct.
21st. There will be no recitation section on the 20th, but SJL will be available for
questions by email and in the office on Tuesday from 3 to 5 PM.
Cytochrome c Oxidase
O2 binds and is reduced at the CuB-heme
Proposed O–O Bond Splitting Mechanism
O–O bond splitting mechanism in cytochrome oxidase
Margareta R. A. Blomberg, Per E. M. Siegbahn, Gerald T. Babcock and Mårten
Wikström
New Strategies and Tactics for Optical Imaging
of Zinc, Mercury, and NO in
Metalloneurochemistry
Metalloneurochemistry
Examples where metal ions and coordination
compounds play a key role in neurobiology:
Ion Channels and pumps: Na+, K+, Mg2+, Ca2+
Signaling at the synapse: Zn2+ (hippocampal CA3 cells),
NO (guanylyl cyclase), Ca2+ (synaptotagmin)
Metalloenzymes and neurotransmitters: dopamine
b-hydroxylase, a-amidating monooxygenase
Review: S. C. Burdette & S. J. Lippard, PNAS, 2002, 100, 3605-3610.
Toxic Effects of Metal Ions in Neurobiology
Metal ions have also been connected with neurological
disorders including:
Familial amyotrophic lateral sclerosis (FALS; Cu/Zn)
Alzheimer’s disease (AD; Fe, Cu and Zn)
Prion diseases such as Creutzfeldt-Jakob disease and
transmissible spongiform encephalopathies (Cu and Zn)
Parkinson’s and Huntington’s disease
Environmental contamination (Hg and Pb)
Research Objectives
Construct bright, fast-responding fluorescent sensors
for
zinc(II) and nitric oxide, and apply to understand
neurochemical signaling by these species.
Synthesize fluorescent, “turn-on” sensors for
mercury(II) ion and apply to detect environmental
mercury.
Ultimately develop “optical imaging” as a complement
to
MRI for connecting behavior with chemistry in
primates and humans.
Zinc and the Neurosciences
Neuronal Zn2+: Brain contains
highest Zn2+ concentrations in
body (mM).
Labile Zn2+: chelatable Zn2+ colocalized with Glu in vesicles of
hippocampus, which controls
learning and memory.
Mobile Zn2+: Up to 300 mM Zn2+
released into synaptic cleft of
dentate gyrus-CA3 mossy fiber
projections in hippocampus.
Adapted from http://www.ahaf.org/alzdis/about/brain_head.jpg
Proc. Natl. Acad. Sci. USA 2003, 100, 3605
Zn2+ and Signaling in Neurons
Presynaptic
Glutamate
Nerve Terminal
ZnT-3
NMDA R
Postsynaptic
Neuron
Adapted from Nature 2002, 415, 277.
• ZnT-3 is a Zn2+ transporter
that loads the vesicles in
presynaptic neurons (300
mM)
• Released Zn2+ binds to
extracellular side of
NMDA receptor
• Knockout mice lacking
ZnT-3 have few neurological symptoms and do
not get b-amyloid plaques
Uncontrolled Zn2+ Release and Neuronal Damage
Neurotoxicity: Uncontrolled Zn2+
release during seizures induces
acute neuronal death.
Neurodegenerative Diseases:
Disrupted Zn2+ release triggers
amyloid peptide aggregration and
the formation of crosslinked
extracellular plaques. Elevated
levels of Zn2+ observed in
Alzheimer’s patients. AD attacks
hippocampus in earliest stage.
www-medlib.med.utah.edu/WebPath/ORGAN.html
Choi and Koh, Annu. Rev. Neurosci. 1998, 21, 347
Defining the Complex Roles of Neuronal Zn2+
Physiology
Presynaptic
Glutamate
Nerve Terminal
ZnT-3
NMDA R
Postsynaptic
Neuron
• Detect Zn2+ release from presynaptic
terminal to the synapse, and onto and
into the postsynaptic neuron
• Correlate Zn2+ fluxes with synaptic
with
synaptic
strength;
simultaneously image Zn2+ fluxes and
measure activities of ligand-gated ion
channels (e.g., glutamate receptors).
Adapted from Nature 2002, 415, 277.
• Use to map neural networks
Pathology
• Map Zn2+ in living tissue during
plaque formation
www-medlib.med.utah.edu/WebPath/ORGAN.html
Requirements for Biological Sensors
1. Water soluble, bind analyte rapidly and reversibly, and have the
ability to tune the lipid solubility.
2. Excitation wavelengths > 340 nm for passage through glass and
minimization of UV-induced cell damage.
3. Emission wavelengths > 500 nm to avoid fluorescence from
native species in the cell. l ~ 700-900 nm for imaging applications.
4. Different emission wavelengths for bound and unbound
fluorophores, so that measurements of analyte concentrations can
be made with correctable background for unbound sensor.
5. Controlled diffusion across cell membrane for intracellular
retention and/or trapping.
6. Tunable dissociation constant (Kd) wrt analyte concentration.
Peptide-Based Zn2+ Sensors
H 3C
H3C
CH3
N
O
N
CH3
HO
O
CO2 H
SO3 O2 S
NH
O
O
N
O
ATK CPE CGKSFSQ C SDLVKHQRTHTG CO2Lissamine
(Donor)
Fluorescein
(Acceptor)
Godwin & Berg, J. Am. Chem. Soc., 1996, 118, 6514
CH3
N
CH3
O2S
O
NH
H3C
HN YQCQYCEKR N
ADSSNLKTHIKTKHS NH2
H O
Walkup & Imperiali, J. Am. Chem. Soc., 1996, 118, 3053.
Designing a Fluorescent Sensor for Zn2+
1)
Selectivity for species of interest (Zn2+ over K+, Na+, Ca2+, Mg2+)
2)
Sensing mechanism: discernable change in emission/excitation
intensity (turn-on) or color (ratiometric) with analyte binding
Photoinduced Electron Transfer (PET) Strategy
Free (OFF)
Bound (ON)
Guest
Host
LUMO
LUMO
HOMO
HOMO
Fluorophore-Receptor
Fluorophore-Receptor
Quinoline-Based Sensors for Intracellular Zn2+
H 3CO
EtO2C
HN
Frederickson, C. J. et al.
J. Neurosci. Meth., 1987,
20, 91-103
N
SO2
O
H 3CO
HN
SO2
CH3
HN
Zalewski, P. D. et al.
Biochem. J., 1993,
296, 403-408
CH3
CH3
TSQ
N
N
CH3
SO2
Kay, A. R. et al.
Neuroscience, 1997,
79, 347-358
CO2H
Zinquin
TFLZn
Properties of Zinquin:
Me
Me
Kd < 1 nM
Detection limit between ~4 pM and 100 nM
Brightness (e F) = 1.6 103 M-1 cm-1
MeO
Excitation/Emission lmax = 350/490 nm
O’Halloran, et al., J. Am. Chem. Soc., 1999, 121, 11448;
J. Biol. Inorg. Chem., 1999, 4, 775.
O
O S
N
O
Zn
N
Me
Me
S
N
N
O
OMe
Synthesis of Fluorescein-based Zn2+ Sensors
O
HO
O
CH3
HO
OH ZnCl2
CH3
O
CH3
OH
O
O
CH3
CH3
O
O
O
Bz2O
pyridine
O
O
O
O
hydantoin
AcOH, PhCl
O
N
Br
O
O
Br
O
O H H O
O
O
N
O
DMSO
NaHCO3
O
HO
O
OH
O
DPA
ClCH2 CH2 Cl
NaBH(OAc)3
N
N
HO
N
O
CO2H
O
N
HO
Cl
O
O
O
N
N
Cl DPA, CH3CN
CO2H (CH2O)n, H2O
N
N
HO
Cl
Zinpyr-2
N
O
O
Cl
CO2H
N
Zinpyr-1
Burdette, Walkup, Spingler, Tsien, and Lippard, J. Am. Chem. Soc., 2001, 123, 7831.
Zn2+-Binding Titration of Zinpyr Sensors
Titration with Zinpyr-2
Hill plot
Fluorescence response to Zn2+ from dual-metal
single-ligand buffer system. Varying [Ca(EDTA)]2and [Zn(EDTA)]2- give free Zn2+ concentrations of
0, 0.17, 0.42, 0.79, 1.32, 2.11, 3.3, 5.6, 10.2 and
24.1 nM. Final spectrum obtained at ~25 mM.
Buffer: PIPES 50 mM, 100 mM KCl, pH 7
Zinpyr-1
Zinpyr-2
Kd
0.7 ± 0.1 nM
0.5 ± 0.1 nM
Response fits a Hill coefficient of
1 indicating a 1/1 Zinpyr:Zn2+
complex is responsible for
the fluorescence enhancement
lex
inc. in integrated emission
507 nm
3.3 fold
490 nm
6.0 fold
Zn2+-Induced Fluorescence Enhancement
Quantum Yields:
Fluorescein
F = 0.95
Zinpyr-1 F = 0.39
Zinpyr-1 + Zn2+ F = 0.87
Zinpyr-2 F = 0.25
Zinpyr-2 + Zn2+ F = 0.92
50 mM PIPES, 100 mM KCl
pH 7
N
Zinpyr-2
Brightness (e F)
25 mM Zn2+, 1 mM Zinpyr
Zinpyr-1 : 85 103 M-1 cm-1
Zinpyr-2 : 45 103 M-1 cm-1
N
N
N
HO
X
N
O
O
X
CO 2H
N
Metal Ion Selectivity of Fluorescence
Response
Zinpyr-1
Zinpyr-2
50 mM PIPES, 100 mM KCl, 10 mM EDTA, pH 7
20 mM M2+; neither 1 mM Mg2+ nor 1 mM Ca2+ interfere
Fluorescence enhancement by closed shell metal ions is
indicative of a PET quenching mechanism of the unbound fluorophore
Behavior of Zinpyr in Aqueous Solution
+
N
N
NHN
O
NHN
O
O
X
CO2-
X
N
+ Zn2 +
- Zn2 +
Kd(1) = 0.5 - 0.7 nM
N
N Zn N
H2 O
O
X
2+
N Zn N
H2O O
X
N
N
O
O
O
N ZnN
OH2
O
X
Crystallization
CH3 CN
O
CO2
- Zn 2+
N
NHN
O
N Zn N
H 2O O
X
-
+ Zn 2+
H2 O
X
Kd(2) = 75 mM
N
Zn
N
N
O
O
CO2
-
X
2+
X-ray Crystal Structure of Zinpyr-1 Complex
O
N
Cl
2.04 2.09
N
1.94
O
Zn
2.18 2.07
N
OH2
NMR studies show free ligand and formation of 1:1 and 2:1 complexes. The 1’ and
8’ protons on fluorescein ring are indicative of the structure. The lactone ring
forms as a result of crystallization; in solution, the complex is in the open,
fluorescent form.
Note possible coordination site on zinc for external ligand.
Fluorescence Response of Zinpyr-1 in COS-7
Cells
Zinpyr-1 (5 mM)
ON+
After addition of Zn2+ (50 mM)
and pyrithione (20 mM)
SH
pyrithione
Zinpyr Localizes in the Golgi or a Golgi-Associated Vesicle
Zinpyr-1
GT-ECFP
Overlay
GT-ECFP lex = 440, lem = 480
Zinpyr-1 lex = 490, lem = 535
GT-ECFP - galactosyl transferase-enhanced cyan fluorescent protein fusion
Walkup, Burdette, Lippard, & Tsien, J. Am. Chem. Soc., 2000, 122, 5644.
Burdette, Walkup, Spingler, Tsien, and Lippard, J. Am. Chem. Soc., 2001, 123, 7831.
Brief Introduction to Two-Photon Microscopy (TPM)
Jablonski Diagrams of the absorption-emission
process
One Photon
Two Photon
Comparison of imaging methods
OPE
TPE
TPM - 3D imaging technology based on nonlinear excitation of fluorophores
TPM has 4 unique advantages:
1. Significantly reduces photodamage, facilitating imaging of living species
2. Permits sub-mm resolution imaging of specimens at depths of hundreds of mm
3. Highly sensitive since the emission signal is not contaminated by excitation light
4. Initiate photochemical reactions in subfemtoliter volumes inside tissues and cells
Two-Photon Microscopy of Zinpyr Sensors
1. MCF-7 cells w/Zinpyr-1
0
2. Zn2+/pyrithione
750
3. TPEN
TPM collaboration with M. Previte and P.T.C So, MIT
Zinpyr-1 Staining of Zinc-Rich Mossy Fibers in a 200 m
Thick Rat Hippocampal Brain Slice*
4 X Dry
60 X Oil
Granule Neurons
Mossy Fibers
About 1 mm
*Courtesy of Dr. C. J. Frederickson, U. Texas
Fluorinated ZP with Enhanced Dynamic Range
ZP1
ZP2
ZP3
ZPF1
ZPCl1
ZPBr1
ZPF3
X/Y
Cl/H
H/H
F/H
Cl/F
Cl/Cl
Cl/Br
F/F
pKa
8.4
9.4
6.8
6.9
7.0
7.3
6.7
Emission
1
0.8
0.6
0.4
0.2
0
2
4
6
8
pH
10
12
F(free)
0.38
0.25
0.15
0.11
0.22
0.25
0.14
Fluorescence Response of Electronegative ZP Probes to Zn2+
ZP1
ZP2
ZP3
ZPF1
ZPCl1
ZPBr1
ZPF3
X/Y
Cl/H
H/H
F/H
Cl/F
Cl/Cl
Cl/Br
F/F
pKa
8.4
9.4
6.8
6.9
7.0
7.3
6.7
F(free)
0.38
0.25
0.15
0.11
0.22
0.25
0.14
F(Zn2+)
0.87
0.92
0.92
0.55
0.50
0.36
0.60
Kd / nM
0.7
0.5
0.7
0.9
1.1
0.9
0.8
Intracellular Staining of Zn2+ in Live Hippocampal Neurons
ZP3 tracks intracellular Zn2+ reversibly
ZP3 (10 mM)
+ Zn(pyrithione)2 (50 mM)
+ TPEN (50 mM)
embryonic rat hippocampal neurons, DIV
18
Chang and Lippard, unpublished
ZP3 Localizes in a Golgi or Golgi-Associated Compartment
ZP3 co-stains with Golgi marker
ZP3 (10 mM)
GT-DsRed
Overlay
embryonic rat hippocampal neurons, DIV 18
Time-Resolved Detection of Zn2+ Entry into Live Neurons
ZP3 can respond to Zn2+ fluxes on the ms to s timescale
Zn2+ (50 mM)
0s
250 ms
500 ms
1s
2s
5s
10 s
30 s
TPEN (50 mM)
embryonic rat hippocampal neurons, DIV 18
Imaging Endogenous Zn2+ in Live Brain Tissue
ZP3 can probe endogenous Zn2+ in intact tissue
ZP3 (10 mM)
TPEN (50 mM)
CA1
mossy fibers
CA3
dentate gyrus
Acute rat hippocampal slices, 90 day-old adults
Synthesis of Trappable Zinpyr-1 Sensors
Woodroofe & Lippard, 2003
ZP1T, R = Et
Metabolite, R = H
Physical Constants and Cell Permeability of ZP1T
Negative control
ZP1T, R = Et
Metabolite,
R=H
HeLa cells were incubated 30 min at RT with the indicated
dye, washed, and treated with 20 mM Zn-pyrithione for 10
min at RT. Image exposure time was 20 sec.
Ff ree
FZn
Kd (nM)
R=H
0.21
0.63
0.2
R = Et
0.13
0.67
0.4
Conclusion: the ethyl ester
enters cells, becomes
hydrolyzed to the acid. This
anion is trapped in the cell and
can sense zinc influx.
Woodroofe & Lippard, 2003
Extracellular Zinpyr Probes - ZP4
HOOC
O
OH
Cl
HO
CH3
O
CH3
HO
OH ZnCl2
O
TBS-Cl, DMF H C Si
3
t-Bu
Cl
imidazole
OH
O
OH
H3C
CH3
O
O
O
NH2
H3C
O
hydantoin H C Si
3
AcOH, PhCl
t-Bu
O
O
X
N
N
N
HN
O
H3C
H3C Si
t-Bu
N
O
CH3
Si CH
3
Cl t-Bu
O
O
O
N
N
HN
HO
X
N
CH3
X
Si CH
3
Cl t-Bu AgNO3 , CH3 CN
pyidine
O
O
TBAF
THF
O
N
Br
CH3
Si CH
3
Cl t-Bu
O
O
N
O
Cl
CO2 H
Zinpyr-4 will carry a charge of -1 at
neutral pH and thus not have the
cell penetrating properties of
Zinpyr-1 and Zinpyr-2.
Burdette & Lippard, 2002
Fluorescence Properties of Zinpyr-4
Kd = 0.65 ± 0.10 nM; lex = 500 nm
inc. integrated emission ~ 5-fold
lex (max)
F/Brightness
Zinpyr-4
506
0.06/2.9 103 M-1 cm-1
Zinpyr-4/Zn2+ 495
0.34/19.2 103 M-1 cm-1
50 mM PIPES, 100 mM KCl, pH 7
Zinpyr-4 Stains Zinc-Injured Neurons, but
Not Zinc-Filled Vesicles (Neuropil)
Epileptic seizure was
drug-induced in rats.
Zinc floods are released
from synaptic terminals.
Zinc enters vulnerable
neurons. Zinpyr-4, being
charged, cannot
penetrate vesicles and
thus images zinc only in
the damaged neurons.
The images are seen
after slicing in the
microtome. A significant
improvement over TSQ,
which images all zinc,
being lipophilic.
Hippocampal Neurons Damaged After Epileptic Seizure
Burdette, Frederickson, Bu, & Lippard, J. Am. Chem. Soc. 2003, 125, 1778.
Comparison of ZP4 and TSQ Sensors
N
H 3C
O
N
N
N
HN
HO
O 2S
O
NH
O
Cl
CO 2H
ZP4
CH3
TSQ
Hippocampal Pyramidal Neurons Injured By
Zinc-Influx During Epileptic Seizure
10 m
Zinpyr-4
Four Neurons Stained with ZP4
Note Intense Staining of Nuclei
Synthesis of Coumazin-1 - a Dual Fluorophore Sensor
HO2 C
CO2H HO
OH MeSO H HO
3
Cl
Cl
CO2H
O
O
Cl
CO2H
Ac2 O
pyridine
AcO
Cl
HO2 C
O
OAc
O
Cl
O
HO2 C
1. (COCl)2, DMF
AcO
OH
Cl
H
N
2. H2N
HO
O
OAc
O
O
N
O
O
O
O
N
O
O
N
Cl
CO2H
H
N
O
N
O O
Cl
H
N
O
OAc
Coumazin-1
Cl
O
O
N
N
Cl
O
O
N
HO
O
O
O
N
N
PPh 3, DIAD
Cl
O
O
DPA, CH3CN
(CH2 O)n , H2 O
AcO
Essentially nonfluorescent in linked
form; F < 0.04
Membrane permeable
Woodroofe & Lippard, 2003
O
Esterase Treatment of Coumazin-1
Michaelis-Menten kinetics of Coumazin-1
N N
HO
N
N
N N
O
O
Cl
Cl
CO2H
O
HN
O
O
O O
Cell
permeable
N
Esterase
N N
HO
N
Cl
O
N N
O
h = 445 nm
Cl
CO2H
O
HN
kcat = 0.023 mmol-1 min-1; kcat/Km = 0.37 min-1
N
h = 488 nm
+
OH
O
-O
O O
N
h = 505 nm h = 525 nm
Treatment of CZ-1 with commercial pig liver esterase yields parent fluorophores. Coumarin
343 fluorescence (lex 445 nm, lem 488 nm) indicates ester hydrolysis obeys MichaelisMenten kinetics. Cell studies are in progress (Woodroofe & Lippard, J. Am. Chem. Soc.,
2003).
Ratiometric Properties of Coumazin-1
Results:
Emission (arbitrary)
l534: l488 = 0.5 (no Zn2+)
lex = 505 nm
l534: l488 = 4.0 (xs Zn2+)
lex = 445 nm
+ Zn2+
Coumarin fluorescence
is unaffected, whereas
Zinpyr fluorescence
increases in response to
added Zn2+
Wavelength (nm)
A 2 mM solution of Coumazin-1 in HEPES buffer (pH 7.5)
was treated with pig liver esterase (Sigma) overnight.
Zn2+ was titrated into a 2 mL aliquot and the
fluorescence spectrum was recorded with excitation at
both 445 nm and 488 nm.
Woodroofe & Lippard
J. Am. Chem. Soc., 2003.
Imaging Zinc in HeLa Cells with Coumazin-1
No Zn, top; Zn pyrithione, bottom
Phase contrast
l(ex) 400-440 nm
l(ex) 460-500 nm
Implications and Future Work
• The Zinpyr family of intracellular sensors are excellent for use in two-
photon microscopy and have been optimized in second generation
synthetic studies to reduce background in the unbound sensor.
• A trappable Zinpyr sensor is available.
•Zinpyr sensors image Zn2+-containing synaptic vesicles in brain
slices, as well as Zn2+ exogenously applied to living cells and in injured
neurons.
• The extracellular sensor ZP4 has identified previously unseen, highly
fluorescent cells that become more abundant in pups and following
trauma.
• Coumazin, a dual fluorophore sensor, is ratiometric; cell studies are
in progress.
Acknowledgements
Coworkers:
Shawn Burdette, Chris Chang, Liz Nolan, and
Carolyn Woodroofe
Collaborators:
Morgan Sheng, Jacek Jaworski, MIT, cell imaging
Grant Walkup, Roger Tsien, UCSD, zinc sensors
Peter So, Michael Previte, MIT, two photon work
Chris Frederickson, NeuroBioTech, neuronal imaging
Support:
National Institute of General Medical Sciences
McKnight Foundation for the Neurosciences
MIT
Shawn Burdette
Carolyn Woodroofe
Chris Chang
Liz Nolan
Mercury in the Environment
human
consumptio
n
marine
environment
Hg2Cl2, Hg(II), Hg(0)
“inorganic mercury”
bacteri
a
food chain
(neurotoxic!)
methylmercury
Second Generation Hg(II) Sensor Synthesis
NO2
Br
Cl
HN
Cl
EtSH / Na
NO2
S
HN
EtOH, reflux
S
S
N
S
K2CO3
MeCN, rt
NH2
Pd black
H2 (1 atm)
S
N
S
MeOH
Tanaka, M. et. al. J. Org. Chem. 2001, 66, 7008-7012
S
S
N
NH2
H
HO
O
S
N
O
S
O
Cl
CO2H
1. EtOAc, rt
2. DCE, NaB(OAc)3H, rt
Nolan & Lippard, submitted (2003)
NH
HO
O
O
Cl
CO2H
Photophysical Characterization
Integrated Emission
1.2
1.0
pKa = 7.1
0.8
S
pKa = 4.8
S
0.6
N
0.4
NH
0.2
0
HO
2
4
6
8
10
12
Fluorescence Intensity
pH
30
O
O
Cl
CO2H
pH 7
25
20
+ Hg(II)
15
pH 7: ~500% increase in intensity w/ Hg(II)
10
5
ffree= 0.04 (e = 61,300 M-1cm-1)
0
fHg= 0.11 (e = 73,200 M-1cm-1)
480 500 520 540 560 580 600 620
Wavelength (nm)
Mercury Binding Properties
35
5
Intensity Change
Fluorescence Intensity
pH 7
30
25
20
S
4
N
3
NH
2
HO
0
O
O
+ TPEN
1
Cl
CO2H
free sensor
15
S
+ Hg(II)
1
2
3
4
5
Number of Cycles
10
1:1complex
5
0
480
500
520
540
560
580
Wavelength (nm)
600
620
640
Fluorescence enhancement
EC50 = 410 nM
A 2-ppb level of Hg(II) gives a 11.3± 3.1% fluorescence increase.
Selectivity for Mercuric Ion
6
pH 7
5
S
S
F / Fo
4
N
3
NH
2
HO
1
0
6
O
Cl
CO2H
1 2 3 4 5 6 7 8 9 101112 1314151617
pH 7
5
F / Fo
O
Cations of interest:
4
1, Li(I); 2, Na(1); 3, Rb(I); 4, Mg(II);
3
5, Ca(II); 6, Sr(II); 7, Ba(II); 8, Cr(III);
2
9, Mn(II); 10, Fe(II); 11, Co(II); 12, Ni(II);
1
13, Cu(II); 14, Zn(II); 15, Cd(II); 16, Hg(II);
0
1 2 3 4 5 6 7 8 9 101112 1314151617
17, Pb(II)
Summary
We have developed fluorescein-based sensors
for Hg(II) with desirable characteristics,
including:
Fluorescence “turn-on”
Water solubility
Selectivity for Hg(II)
Reversible binding
Immediate response
Detection of environmentally relevant [Hg2+]
Work of Liz Nolan
Nitric Oxide and the Neurosciences
NO and brain function (positive aspects):
Neuronal NO synthase (nNOS) is expressed in
postsynaptic terminal of neurons in the brain. Proposed
to act as a retrograde neurotransmitter in the
hippocampus during memory formation.
NO and brain damage (negative aspects):
Forms reactive nitric oxide species (RNOS) such as
NO2 and NO-, as well as ONOO-, peroxynitrite. All are
potentially neurotoxic and implicated in disorders including
HD, ALS, AD, MS, & stroke.
Goal: Obtain an in vivo sensor for NO, which can have a
physiological lifetime of ≤ 10 min and diffuse 100-200 mm.
NO in the Brain
NO acts as a neurotransmitter by passive
diffusion from its point of synthesis to
the target neuron
Presynaptic
neuron
cGMP
Stimulation of the postsynaptic
neuron by NO results in synthesis of
cGMP by soluble guanylate cyclase
(sGC)
sGC
NO
NOS
Current research relies on use of NOS
inhibitors and NO donors to elucidate
neuronal functions of NO
Postsynaptic
neuron
Existing NO Detectors in Biology
Griess assay for nitrite; electrochemical microsensors;
fiber optic fluorescent sensors: all have liabilities.
Soluble fluorescent sensors are desirable.
R
R
C6H5
R
C6H5
.
CO2H NO
C6H5
CO2H
reduction
.
NO
CO2H
NOH
CO2H
C6H5
C6H5
1
Non-fluorescent
CO2H
CO2H
C6H5
2
3
Weakly fluorescent
Fluorescent
1a, 2a, 3a
1b, 2b, 3b
R= H
R = N(CH 3) 2
Known as FNOCTs, fluorescent NO chelotropic traps,
these non-coordination compound sensors are valuable.
Problem: requires a reductant.
Other NO Detection Strategies
Quinoline-pendant cyclam
Sensor; light turns off
Katayama, et al., Anal. Chim. Acta
(1998) 365, 159-167
R
N
N
N
Fe2+
N
ON
Fluorescent
R
NH
NO.
X
O
N
N
N
Non-fluores cent
N
CO 2
O-
N
N
2+
Fe
N N
NH 2
X
NO
N
.
O
Non-fluor escent
O2
X
Diaminofluoresceins
require N2O3
O
Kojima, et al., Anal. Chem.
(1999) 39, 3209-3212
-
CO 2
X
O-
O
Fluor escent
Synthesis of Co(i-PrDATI)2
O
OTs PMB
Et3N
O HN
R N HN
1. Me3 OBF4
2. RNH 2, CH2Cl2
EtOH,
R N
TFA
1. KH
2. CoCl2, THF
R = i-Pr
OCH3
OCH3
N(CH 3)2
NH 2
1. NaH
2. DsCl, THF
Co( i-PrDATI)
R N HN SO2
R = i-Pr
H(i-PrDATI)
2
Franz, Singh, Spingler, Lippard,
Inorg. Chem., 2000, 39, 4081-4092
Reaction of NO with Co(i-PrDATI)2
Co(
i-Pr
DATI)2
NO
Co(i-PrDATI)(NO)2 +H(i-PrDATI)
1760
Infrared spectra reveal {Co(NO)2}10 unit
1837
Abs
x 10-3
8.0
2. 0
6.0
1. 5
4.0
2.0
1. 0
0.0
0. 5
2100
2000
1900 1800
cm-1
1700
1600
Time
(h)
NMR studies demonstrate ligand release.
Fluorescence spectra are consistent
Suggests a new strategy for NO sensing;
Franz, Singh, Spingler, Lippard,
Inorg. Chem., 2000, 39, 4081-4092
fluorescence intensity
2200
400
440
480
520
nm
560
600
640
Design of a Novel Fluorescent Sensor
For NO Based on Cobalt(II) Coordination Chemistry
Me2N
S
O
O
N
H
H
N
S
O
N
The Co(II) complex of this ligand reacts
with NO but not O2 , as judged by fluorescence
spectral changes
N
a
(CH2) 4
H2DAT I-4
[Co(CH3CN)4](PF6)2
base
fluorescence intensity
O
NMe2
400
6h
b
+ NO
+ air
3 min
480
nm
560
640
400
480
560
nm
640
Franz, Singh, Lippard, Angew. Chem. Int. Ed., 2000, 39, 21212122
Interpretation of Fluorescence Changes when
NO Reacts with Co(i-PrDATI-4)
h =
350 nm
N
N
N
h =
350 nm
X
SO2
SO2
N
N
Co
N
h =
505 nm
SO2
NO
N
N
NO
Co
N
N
Franz, Singh, Lippard
Angew. Chem. Int. Ed., 2000, 39, 2121-2122
N
NO
HN
SO2
Synthesis and Structure of [Rh2(m-O2CCH3)4(Ds-R)2]
N
[Rh2(OAc)4]
+
[Rh2(OAc)4(Ds-R)2]
O S O
R
Ds = dansyl
N
R =
N
N
N
H
Ds-im
N3
O5A
O6A
Ds-pip
O4A
O1A
O3
S1 N2
N1
N1A
Rh1A
O2
Rh1
S1A
Selected crystallographic data
for [Rh2(OAc)4(Ds-im)2]:
O2A
O3A
O1
O6
N2A
Rh1-Rh1A 2.3906(7) Å
O4
O5
N3A
Rh1-N1
2.237(3) Å
Rh1-Oav
2.038 Å
Fluorescence Emission Spectra of Rh2(OAc)4(Ds-Im)2]
in DCE with Alternating 100 equiv NO/Ar Purges
100
+NO
Normalized
emission
80
60
40
20
Ar sweep
0
500
550
600
Wavelength (nm)
650
Reactivity of Rh2(OAc)4(Ds-Im)2]
in the Presence of Nitric Oxide
lex =
365 nm
lex =
365 nm
Ds-im
Ds-im
NO
lem =
560 nm
100 equiv NO
1,2-dichloroethane
Hilderbrand & Lippard, submitted
NO Sensors - Summary of Progress
Desirable Properties of a NO sensor:
Selective for NO over O2
Direct detection of NO
Sensitive
Simple instrumentation
Spatial resolution
Temporal resolution (<1 ms)
• Water solubility
Semiporous Membrane - An Approach to
the Water Solubility Problem
Aqueous NO at 1.9 (left) and 0 (right) mM in contact with 20 µM [Ru2(OAc)4]:DsPIP in a 2 :1 ratio. The two solutions are separated with a silicone polymer membrane
and irradiated with a hand-held illuminator, l 365 nm (Lim and Lippard,
unpublished).
Implications and Future Work
•A strategy has been designed to use coordination chemistry to build NO
sensors. Ligand dissociation upon NO binding allows fluorescence to
increase significantly.
•This strategy was tactically applied to provide the first reversible NO sensor
based on a ligand-tethered fluorophore bound to (m-acetato)-dirhodium(II).
Dissociation of the fluorophore in organic solvents following NO binding
yields bright fluorescence.
•Introduction of an aqueous NO solution through a semi-permeable
membrane provides a route to fashion fiber optical NO sensing devices for
biological applications.
•Needed improvements sensing NO in vivo include: water solubility; better
quantum yields and longer wavelength excitation; greater fluorescence
enhancement; ratioability; additional biological compatibility.
Acknowledgements
Coworkers:
Katherine Franz, Scott Hilderbrand, Mi Hee Lim
Support:
National Science Foundation
5.062, 2002
Finé!








