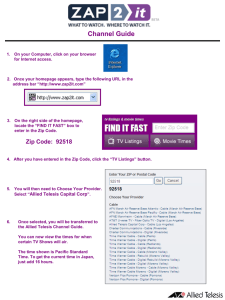
MALDI Clean-up - QB3
advertisement

Sample clean-up (MALDI) • Removal of buffer, salts, urea, guanidine, EDTA, glycerol, DMSO, detergents • Dilution • Washing • Drop Dialysis • Cation Exchange • Zip Tips Typical contaminants in protein/peptide samples • No interference: TFA, formic acid, acetic acid, mercaptoethanol, DTT, HCl, NH4OH. • Tolerable (<50 mM): HEPES, MOPS, Tris, NH4OAc. • Minimizing buffer concentrations improves performance. • Avoid: Glycerol, sodium azide, DMSO, SDS, phosphate, NaCl, Urea (> 2M), guanidine (>2M) Sample Dilution • The goal is to dilute the contaminants to the point where they no longer interfere with the analysis of the sample. • Requires high enough analyte concentration in the sample to provide acceptable data after dilution. On-Plate Washing Buffer and salt removal Dry sample and matrix Deposit 1 - 2 L cold 0.1% TFA Leave for 5 - 10 sec, then remove Detergent contamination use 5% isopropanol - cold Cell extract contamination Use 100% isopropanol- cold Drop Dialysis • • • • To remove low molecular weight contaminants. Fill a 250 mL container with ultra pure water. Float the membrane on the water (shiny side up). Place 10 - 25 L of sample solution on the membrane. • Add 1 L of AcN to sample spot to increase surface area, • Allow to sit for approximately 45 min. • Remove with pipette, add matrix, spot. Cation Exchange • • • • For removal of alkali metal ions. Prepare resin in NH4+ form according to instructions. Place app. 0.1 mg of beads on a clear piece of parafilm. Add 5 L of sample and 5 L matrix to beads to make a slurry (the slurry should be app. 50% beads). • Slowly mix up and down with a pipette 10 - 15 times. • Allow beads to settle for 15 - 30 sec. • Pipette supernatant onto sample plate. – Avoid getting beads onto plate. – Does not work for positively charged species. Solid Phase Extraction - Zip Tip • Zip tip - miniature column chromatography. • Standard C18 zip tips have 0.6 L bed volume. • Micro C18 zip tips have 0.2 L bed volume better for automation. • C4 zip tips for clean-up of protein samples. • Other types available, e.g. metalchelating for phosphor peptides and cation exchange. Procedure for Zip Tip Use • Condition the tip according to instructions. • Load the sample onto the tip by pipetting 5 - 10 L of sample up and down several times and discard the liquid. • Wash with 3 x 10 L 0.1% TFA to remove salts. • Elute the sample using 30 - 70% AcN or matrix.