Chapter 3 HK - Chemistry Department
advertisement

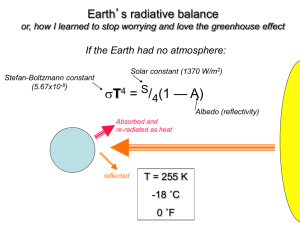
Chapter 3 The Chemistry of Global Warming Chemistry 1010 Dr. Harris Suggested Problems Emphasizing Essentials (pgs. 144 and 145): 2, 7, 9, 16, and 21. Concentrating on Concepts (pg. 146): 33. Exploring Extensions (pg. 147): 46. 3.1 In the Greenhouse Jean-Baptiste Joseph Fourier (1768-1830) John Tyndall (1820-1893) French mathematician and physicist Atmosphere and the glass of a “hothouse” or greenhouse. CO2 and H2O vapor absorb heat Today CO2 absorbs heat Atmospheric CO2 concentration has increased over the past 150 years Earth’s avg. temp has not remained constant 3.2 The Testimony of Time Photosynthesis chlorophyll 6 CO2 + 6 H2O 100 million years ago C6H12O6 + 6 O2 glucose Estimated avg. temp 10-15ºC warmer than today CO2 concentration assumed to be considerably higher Figure 3.3 Notice the correlation between carbon dioxide and temp. change. 3.3 The Earth’s Energy Balance Figure 3.4 – Look closely at the width of each arrow. 3.3 The Earth’s Energy Balance Global Greenhouse Effect Return of 84% of the energy radiated from the surface of the Earth Enhanced Greenhouse Effect Energy return of greater than 84% 3.4 Molecules: How They Shape Up CO2, H2O, and CH4 are greenhouse gases. N2 and O2 are not greenhouse gases. Why? Answer comes from a closer look at the molecular structure and shape. 3.4 Molecules: How They Shape Up Review of Lewis structures for N2 and O2 How about methane’s structure? Let’s look at four different molecular structures (methane, ammonia, water, and carbon dioxide). Determine # of outer electrons associated with each atom in the molecule. Remember the octet rule when drawing the molecule. Most stable molecular shape – bonding and nonbonding electrons are as far from each other as possible. Remember structure names and bond angles. Question 11 3.5 Vibrating Molecules and the Greenhouse Effect Figure 3.13 – Molecular vibrations in carbon dioxide. 3.5 Vibrating Molecules and the Greenhouse Effect Figure 3.16 – Molecular response to types of radiation. 3.5 Vibrating Molecules and the Greenhouse Effect Figure 3.14 Question 12 3.6 The Carbon Cycle: Contributions from Nature and Humans 6.0-8.2 Gt – amount of carbon dioxide released by human activities (deforestation/burning fossil fuels) per year. About ½ of this carbon dioxide is recycled into the oceans and biosphere. 3.1-3.5 Gt increase of carbon per year. Question 23 - Interesting 3.7 Weighing the Unweighable Atomic mass – average mass of an atom of that element as compared to an atomic mass of exactly 12 amu for carbon-12. 1 amu = 1.66 x 10-24 g. Notice that the mass number for carbon is 12.01. The “.01” comes from the contribution of the carbon-13 and carbon-14. Question 18 3.7 Weighing the Unweighable Let’s take a closer look at a laboratory scale. Can only read to 0.1 mg. Atomic Mass – can also be defined as the mass (g) of the same number of atoms that are found in exactly 12 g of carbon-12. # carbon-12 atoms in exactly 12 g = 6.02 x 1023 (Avogadro’s #) One Avogadro’s # of oxygen atoms = 16.00 g. 3.8 Of Molecules and Moles Chemists usually don’t work with single atoms or molecules. Mole – Avogadro’s number of anything. We need a chemist’s dozen. You can think of the “mole” as the chemist’s dozen. C + O2 CO2 Molar mass – mass of one Avogadro’s number of specified particles. Molar mass of carbon atoms = 12.0 g. 3.8 Of Molecules and Moles Molar mass CO2 = (1 x molar mass C) + (2 x molar mass O) = 44.0 g/mole 3.9 Manipulating Moles and Mass with Math Question – what mass of carbon dioxide contains 3.3 Gt of carbon? What mass units do you want to use? Let’s work this out to the mass units that you as a class select. 3.12 Has the Greenhouse Effect Already Started? Figure 3.24 3.12 Has the Greenhouse Effect Already Started? Status of text knowledge Guided by 2001 report from the Intergovernmental Panel on Climate Change (IPCC). Definitely true: CO2 contributes to an elevated global temperature. CO2 concentration in the atmosphere has been increasing over the past century. 3.12 Has the Greenhouse Effect Already Started? Very likely: Virtually certain: The increase in CO2 concentration in the atmosphere over the past century is a consequence of human activity. Average global temperature has increased during the past century. May be true: CO2 and other gases generated by human activity are responsible for this temperature increase. 3.12 Has the Greenhouse Effect Already Started? Uncertain: The average global temperature will continue to increase as anthropogenic emissions of greenhouse gases increase. 3.15 Global Warming and Ozone Depletion End of Chapter 3