lecture 14 organic synthesis
advertisement

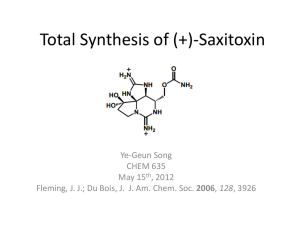
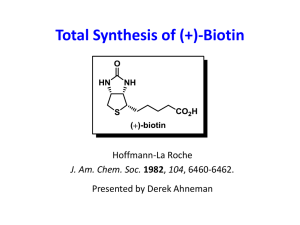
Lecture 14 APPLICATIONS IN ORGANIC SYNTHESIS Copyright ©The McGraw-Hill Companies, Inc. Permission required for reproduction or display. I. Enantioselective functional group interconversions ORGANOMET CHEM IN ORGANIC SYNTHESIS II. Carbon-carbon bond formation via nucleophilic attack on a ligand. ORGANOMET CHEM IN ORGANIC SYNTHESIS III. Carbon-carbon bond formation via carbonyl or alkene insertion. ORGANOMET CHEM IN ORGANIC SYNTHESIS IV. Carbon-carbon bond formation via transmetallation reactions. ORGANOMET CHEM IN ORGANIC SYNTHESIS V. Carbon-carbon bond formation through cyclization reactions. ORGANOMET CHEM IN ORGANIC SYNTHESIS The C=C and C=O undergoes transformations to variety of organic compounds (alcohols, alkyl halides, alkanes). The C=C and C=O are planar and achiral but in their reactions creates one or more stereogenic centers in the reaction product. Assymetric Hydrogenations Methods of producing an enantiomer of a chiral compound: Chemical resolution of a racemate Chiral chromatography Use of a chiral natural products as starting material Stoichiometric use of chiral auxilliaries Asymmetric catalysis Asymmetric Hydrogenations Chiral chromatography: - Use of chiral, enantioenriched groups to the solid support - In the chiral environment, the two enantiomers will have diastereomerically different interactions with the columns ORGANOMET CHEM IN ORGANIC SYNTHESIS Synthesis of biotin (involved in enzymatic transfer of CO2): ORGANOMET CHEM IN ORGANIC SYNTHESIS Use of chiral auxiliaries: ORGANOMET CHEM IN ORGANIC SYNTHESIS Asymmetric Catalysis: same approach as the use of chiral auxilliary except that the selectivity occurs catalytically The most environmentally benign approach to enantioselectivity. ORGANOMET CHEM IN ORGANIC SYNTHESIS Wilkinson’s catalyst: LnM+ (M = Rh or Ir) Assymetric Hydrogenations Chiral Diphosphine Ligands: Asymetric Hydrogenation using Rh Catalysts Mechanism: Assymetric Hydrogenation using Rh-CHIRAPHOS Assymetric Hydrogenation Assymetric Hydrogenation Assymetric Hydrogenation Assymetric Hydrogenation of C=C bonds using Ru(II) Noyori pioneered the development of Ru(II) catalysts showing enantioselective hydrogenation. ASYMMETRIC HYDROGENATION OF C=C BONDS ASYMMETRIC HYDROGENATION OF C=C BONDS ASYMMETRIC HYDROGENATION OF C=C BONDS Asymmetric Hydrogenation of C=O ASYMMETRIC HYDROGENATION OF C=O ASYMMETRIC HYDROGENATION OF C=O ORGANOMET CHEM IN ORGANIC SYNTHESIS ORGANOMET CHEM IN ORGANIC SYNTHESIS Transfer hydrogenation (TH) Asymmetric TH ASYMMETRIC HYDROGENATION OF C=O ASYMMETRIC HYDROGENATION OF C=O Assymetric Hydrogenation Using Ir(I) Catalysts ORGANOMET CHEM IN ORGANIC SYNTHESIS ORGANOMET CHEM IN ORGANIC SYNTHESIS ASYMMETRIC OXIDATION ORGANOMET CHEM IN ORGANIC SYNTHESIS Pd-Catalyzed Oxidation of Secondary Alcohols OXIDATION OF SECONDARY ALCOHOLS ORGANOMET CHEM IN ORGANIC SYNTHESIS CARBON – CARBON BOND FORMATION VIA NUCLEOPHILIC ATTACK ON AN 3 - ligand: THE TSUJI-TROST REACTION ORGANOMET CHEM IN ORGANIC SYNTHESIS Organic synthesis using allylic substrates: unpredictable stereochemistry poor control of regioselectivity possible carbon- skeleton rearrangement. Leaving groups for Tsuji-Trost Reaction TSUJI – TROST REACTION Tsuji-Trost Reaction: With hard nucleophiles (pKa of conjugate acid >25) results in an overall inversion of configuration at the allylic site. With soft nucleophile (pKa of conjugate acid < 25) react to give retention of configuaration. TSUJI – TROST REACTION TSUJI – TROST REACTION TSUJI – TROST REACTION - EXAMPLE TSUJI – TROST REACTION Several points in catalytic cycle where asymmetric reaction could occur: a) enantiomeric faces of the alkene b) enantiomeric leaving groups c) enantioface exchange in the 3 allyl complex d) attack at enantiotopic termini of the 3 ally ligand e) Attack by different enantifaces of prochiral nucleophiles. ASSYMETRIC TSUJI – TROST REACTION TSUJI-TROST REACTION TSUJI_TROST REACTION Assymetric Quat center Tsuji-Trost Reaction – Quat Center EXAMPLE: Tsuji-Trost Reaction ORGANOMET CHEM IN ORGANIC SYNTHESIS Tsuji Trost Reaction: C-C Bond formation via CO and alkene insertion CARBONYLATION INSERTIONS CARBONYL INSERTIONS EXAMPLE CARBONYL INSERTIONS C-C Double bond Insertion: The Heck Reaction Step a ) OA b) alkene coordination c) migratory insertion of C=C d) -elimination Insertion is key step R = aryl, alkyl, benzyl or allyl X = Cl, Br, I, OTf Heck Reaction – migratory C=C insertion Rate of reaction and regioselectivity are sensitive to steric hindrance about the C=C bond. Rate of reaction varies according to: Heck Reaction: Example: Heck Reaction Heck Reaction Also know as Cross Coupling Reaction: C-C Bond Bond formation via Transmetallation Reactions Transmetallation Reaction – a method for introducing a -bonded hydrocarbon ligands Into the coordination sphere transition metals. The equilibrium is thermodynamically favorable from left to right if the electronegativity of M is greater than that of M’. Transmetallation Reaction TRANSMETALLATION REACTIONS Via a concerted -bond metathesis --------transfer of R to M with retention of configuration. TRANSMETALLATION REACTION MECHANISM TRANSMETALLATION REACTIONS 4-TYPES GENERAL REACTION MECHANISM CROSS-COUPLING REACTION - GENERAL CROSS-COUPLING REACTION The use of organotin compound have the advantage that one group will preferentially transfer over the other: CROSS-COUPLING REACTION Example: Propose a catalytic cycle for the cross coupling plus carbonylation reaction below CROSS-COUPLING REACTION Mechanism: CROSS-COUPLING REACTION - STILLE Synthesis Application Example: CROSS-COUPLING REACTION - STILLE Sample Problem: CROSS-COUPLING REACTION - STILLE Transmetalating Agent is R-B(R’)2 but similar in scope as the Stille. CROSS-COUPLING REACTION - SUZUKI Reaction Pathway: CROSS-COUPLING REACTION - SUZUKI Synthesis Application: The chemo-, regio-, and stereoselectivity similar to those with Stille. Suzuki more widely used for aryl-aryl coupling. CROSS-COUPLING REACTION - SUZUKI Cross coupling between alkynyl and aryl : - Requires high loadings of Cu and Pd catalysts, relativelly hight temperatures - Cu-alkynes are formed in situ and then the alkyne is transferred to Pd. CROSS-COUPLING REACTION - Sonogashira Mechanism: CROSS-COUPLING REACTION - Mechanism: CROSS-COUPLING REACTION - Sonogashira Synthesis Applications: CROSS-COUPLING REACTION - Sonogashira Method of choice for syhthesis of acrylic, di- and triterpenoid systems. Organozinc are often used. CROSS-COUPLING REACTION - Negishi Reaction mechanism: CROSS-COUPLING REACTION - Negishi Synthesis Applications: CROSS-COUPLING REACTION – Negishi Mechanism: Dotz Arene Synthesis C-C Bond formation: Cyclizations Cyclization involving Palladium Mechanism: CYCLIZATION Pd Cyclization – Oppolzer’s Cyclization – Pauson - Kand CROSS-COUPLING REACTION