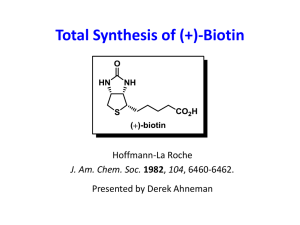
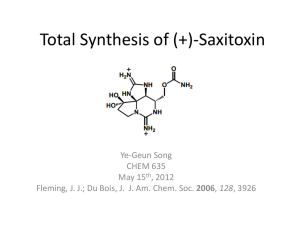
Table of content :
advertisement
جامعة النجاح الوطنية عمل الطالبة : روند فخري ابو الرب . إشراف الدكتور : فؤاد محمود . TABLE OF CONTENT : Objectives . The reaction mechanism . Application and recent literature . References . OBJECTIVE : I make this presentation to achieve the following : 1. To illustrate this interaction . 2. To show you the mechanism for this reaction . 3. Show you the application and the recent literature for this reaction . BACKGROUND : The Biginelli reaction is a multiplecomponent chemical reaction that creates 3,4-dihydropyrimidin-2(1H)-ones 4 from ethyl acetoacetate 1, an aryl aldehyde (such as benzaldehyde 2), and urea 3. It is named for the Italian chemist Pietro Biginelli. The Biginelli reaction : This reaction was developed by Pietro Biginelli in 1891. The reaction can be catalyzed by Bronsted acids and/or by Lewis acids such as boron trifluoride.Several solid-phase protocols utilizing different linker combinations have been published. Dihydropyrimidinones, the products of the Biginelli reaction, are widely used in the pharmaceutical industry as calcium channel blockers, antihypertensive agents, and alpha-1-aantagonists. REACTION MECHANISM : The first step in the mechanism is believed to be the condensation between the aldehyde and urea, with some similarities to the Mannich Condensation. The iminium intermediate generated acts as an electrophile for the nucleophilic addition of the ketoester enol, and the ketone carbonyl of the resulting adduct undergoes condensation with the urea NH2 to give the cyclized product. Application and recent literature: N-Substituted Ureas and Thioureas in Biginelli Reaction Promoted by Chlorotrimethylsilane: Convenient Synthesis of N1-Alkyl-, N1-Aryl-, and N1,N3-Dialkyl-3,4-Dihydropyrimidin-2(1H)(thi)ones Catalysis of the Biginelli Reaction by Ferric and Nickel Chloride Hexahydrates. One-Pot Synthesis of 3,4Dihydropyrimidin-2(1H)-ones Synthetic Manipulation • So, with the dihydropyrimidine in hand, whatcan be done? • Partial of full oxidation (not trivial) • Reduction of the ring to the hexahydropyrimidine • Alkylation and acylation of the heteroatoms • Manipulation of the ester at C(5) • Manipulation of the methyl group at C(6) (halogenation,nitration, etc.) • Ring condensing reactions to make bi,tri-cycles Biological The biological activity is what make thesepyrimdines such attractive targets • (A 1930 patent for use of a Biginelli cmpd for protection of wool from moths!?!) • Antiviral activity • Antibacterial activity • Antitumor • Antiinflammatiry)(مضاد التهابات • Analgesic ((مسكن • Blood palette aggregation inhibitor • Cardiovascular activity • Potent calcium channel blockers CONCLUSION : . Mechanistic insights have provided rational modifications to the experiment protocols, allowing dihydropyrimidines to be synthesized in high yield. The interesting and diverse biological activity of dihydropyrimidines has been explored through the generation of libraries of compounds via microwave, solid-phase, and fluorous-phase technologies. Most recently, asymmetric methods have been developed to give enantioenriched dihydropyrimidines. The frontier of the Biginelli reaction will continue to be developed as new asymmetric methods are reported and as the biological importance of this class of compounds is explored in greater detail. REFERENCES : S. V. Ryabukhin, A. S. Plaskon, E. N. Ostapchuk, D. M. Volochnyuk, A. A. Tolmachev, Synthesis, 2007, 417427. I. Cepanec, M. Litvić, A. Bartolinčić, M. Lovrić, Tetrahedron, 2005, 61, 4275-4280. J. H. Schauble, E. A. Trauffer, P. P. Deshpande, R. D. Evans, Synthesis, 2005, 1333-1339. H. Hazarkhani, B. , Synthesis, 2004, 1239-1242 . www.organic-chemistry.org . Thank you very much for your attention and I hope to be benefited from this presentation.