Alcohols

Reactions of Alcohols
oxidation
tosylation and reactions of tosylates
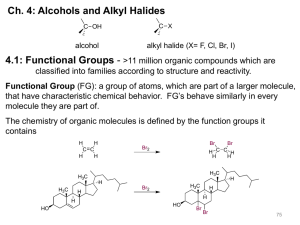
substitutions to form alkyl halides
dehydration to form alkenes and ethers
pinacol rearrangement
esterification
cleavage of glycols
ether synthesis
Classification of Reactions
Oxidations
addition of O or O
2
addition of X
2
loss of H
2
Reductions
loss of O or O
2
loss of X
2
addition of H
2 or H -
Classification of Reactions
Neither an oxidation nor a reduction
Addition or loss of H +
Addition or loss of OH -
Addition or loss of H
2
O
Addition or loss of HX
Classification of Reactions
Oxidations
count C-O bonds on a single C
the more C-O bonds, the more oxidized the C
H
OH
O increasing level of oxidation
OH
OH
O
OH
Reactions of Alcohols -
Oxidation
For alcohols, the oxidation comes from the loss of
H
2
.
Oxidation of a 2° alcohol gives a ketone.
Chromic acid reagent used in lab oxidations.
Na
2
CrO
Cr
3
2
O
7
+ H
+ H
2
SO
4
2
O (dil H
2
+ H
2
O 2H
2
SO
4
) H
2
CrO
4
CrO
4
+ 2NaHSO
4
Reactions of Alcohols -
Oxidation
Oxidation of a 1° alcohol gives
a carboxylic acid if chromic acid reagent is used.
an aldehyde if pyridinium chlorochromate
(PCC) is used.
Reactions of Alcohols -
Oxidation
Two other reagents behave like the chromic acid reagent:
KMnO
4
HNO
3
(will attack C=C, too)
These two oxidizing agents are so strong that C-C bonds may be cleaved.
Bleach (OCl ) also oxidizes alcohols.
Reactions of Alcohols – Swern
Oxidation
Uses dimethyl sulfoxide (DMSO), oxalyl chloride (COCl)
2 and a hindered base.
The reactive species is (CH
3
)
2
SCl + .
The result is a ketone or an aldehyde
(the same as for PCC).
Reactions of Alcohols – Swern
Oxidation
Uses dimethyl sulfoxide (DMSO), oxalyl chloride (COCl)
2 and a hindered base.
OH +
O
H
3
C S CH
3
+
O O
Cl C C Cl
(CH
3
CH
2
)
3
N
CH
2
Cl
2
-60°C
O
H
+ H
3
C S CH
3
+ CO
2
+ CO + 2HCl
Reactions of Alcohols –
Oxidation with DMP
Uses Dess-Martin periodinane (DMP).
Mild conditions: room temperature and neutral pH with excellent yields
The result is a ketone or an aldehyde
(the same as for PCC and the Swern oxidation).
Reactions of Alcohols –
Oxidation with DMP
Uses Dess-Martin periodinane (DMP).
OH +
AcO
OAc
OAc
..
I
O
O
O
H
+
OAc
..
I ..
O + 2HOAc
O
Reactions of Alcohols -
Biological Oxidation
Ethanol is the least toxic alcohol, but it is still toxic.
The body detoxifies ethanol with NAD catalyzed first by alcohol dehydrogenase (ADH) and second by aldehyde dehydrogenase (ALDH):
ethanol acetic acid
The reason methanol and ethylene glycol are so toxic to humans is that, when they react with
NAD/ADH/ALDH, the products are more toxic than the original alcohols.
methanol formic acid
ethylene glycol oxalic acid
Reactions of Alcohols -
Oxidation
3° alcohols will not oxidize , because there is no H on the carbinol C atom.
The chromic acid test capitalizes on this fact:
orange chromic acid reagent turns green or blue (due to Cr 3+ ) in the presence of 1° or 2° alcohols, but doesn’t change color in the presence of a 3° alcohol.
Reactions of Alcohols -
Tosylation
In order to perform an S
N
2 reaction on an alcohol, i.e., with the alcohol as the substrate, the -OH group must leave the alcohol:
R-OH + Nuc: R-Nuc + OH -
OH is a poor leaving group
H
2
O is a better leaving group, but this requires protonation of the alcohol which, in turn, requires an acidic solution. Most nucleophiles are strong bases and cannot exist in acidic solutions.
We need to convert the alcohol to an electrophile that is compatible with basic nucleophiles.
Reactions of Alcohols -
Tosylation
Converting the alcohol to an alkyl halide (already discussed) or an alkyl tosylate lets it act as an electrophile.
Stereochemical configuration of alcohol is retained.
A Tosylate Ion is an
EXCELLENT LEAVING GROUP
As good as or better than a halide.
A Tosylate Ion is an
EXCELLENT LEAVING GROUP
As such, tosylates (just like halides) are candidates for
S
N
2 reactions
E2 reactions
S
N
1 reactions
E1 reactions
Just like the halides
S
N
2 Reactions of Tosylates
R-OTs + OH ROH (alcohol) + OTs
R-OTs + CN RCN (nitrile) + OTs
R-OTs + Br RBr (alkyl halide) + OTs
R-OTs + R’O ROR’ (ether) + OTs
R-OTs + NH
3
RNH
3
+ OTs (amine salt)
R-OTs + LiAlH
4
RH (alkane) + OTs
S
N
2 Reactions of Tosylates -
Mechanism
Single step
Inversion of configuration
Alcohols to Alkyl Halides: Hydrohalic
Acids (HX)
Hydrohalic acids are strong acids, existing in aqueous solution as H + and X .
Recognize a hydrohalic acid: NaBr/H a good leaving group (H
2
O).
2
SO
4
The H + is need to convert the -OH of the alcohol into
The reaction mechanism, S
N structure of the alcohol.
1 or S
N
2, depends on the
Alcohols to Alkyl Halides: Hydrohalic
Acids (HX)
The structure of the alcohol dictates whether the mechanism is S
N
1 or S
N
2.
Alcohols to Alkyl Chlorides:
The Lucas Reagent
Cl is a weaker nucleophile than Br .
ZnCl
2 coordinates with the -OH of the alcohol (like H + does) to form a better leaving group (HOZnCl
2
) than water.
ZnCl
2 is a better Lewis acid than H + .
This promotes the S
N
1 reaction between
HCl and 2° and 3° alcohols.
HCl/ZnCl
2 is called the Lucas reagent.
Alcohols to Alkyl Chlorides:
The Lucas Test
Add the Lucas reagent to a solution of the unknown alcohol and time the formation of a second phase.
3° alcohols react immediately.
2° alcohols take 1-5 minutes.
1° alcohols take >6 minutes.
Alcohols to Alkyl Halides:
Limitations of Using HX
This reaction does not always give good yields of RX.
1° and 2° alcohols react slowly with
HCl, even with ZnCl added.
2
Heating an alcohol with HCl or HBr can give the elimination product, an alkene.
Rearrangements can occur with S
N
(this is not necessarily bad).
1
HI does not give good yields of alkyl iodides, a valuable class of reagents.
Alcohols to Alkyl Halides: PBr
3 and P/I
2
Can give good yields of 1° and 2° alkyl bromides and iodides without the acidic conditions that go with HX.
3 R-OH + PBr
3
PBr
3 and P/I alcohols.
2
3RBr + P(OH)
3
PI
3 is unstable and must be made in situ:
6 R-OH + 2P + 3I
2
6RI + 2P(OH)
3 do NOT work well with 3°
Alcohols to Alkyl Halides: PBr
3
Mechanism
A double S
N
2 mechanism, which is why it does not work on 3° alcohols.
Inversion of configuration, but no rearrangements.
Alcohols to Alkyl Halides: Thionyl
Chloride, SOCl
2
Often the best way to make an alkyl chloride from an alcohol.
ROH + SOCl
2 heat
RCl + HCl(g) + SO
2 dioxane
(g)
Gaseous by-products keep the equilibrium well to the right.
Alcohols to Alkyl Halides:
Best Reagents
Alcohol
1°
Alkyl chloride
SOCl
2
Alkyl bromide
PBr
3
Alkyl iodide
P/I
2
2°
3°
SOCl
2
HCl
PBr
3
HBr
(P/I
2
)
(HI)
Alcohols to Alkenes:
Acid-Catalyzed Dehydration
We studied this in the formation of alkenes.
E1 elimination of a protonated alcohol
Best for 3° and 2° alcohols
Rearrangements common for 1° alcohols due to the carbocation intermediate
Zaitsev product predominates.
Alcohols to Alkenes:
Acid-Catalyzed Dehydration
Step 1: protonation of the alcohol
Fast equilibrium
Converts OH to a good leaving group
Alcohols to Alkenes:
Acid-Catalyzed Dehydration
Step 2: ionization to a carbocation
slow, rate-limiting
leaving group is H
2
O
Alcohols to Alkenes:
Acid-Catalyzed Dehydration
Step 3: deprotonation to give alkene
fast
The carbocation is a strong acid: a weak base like water or bisulfate can abstract the proton.
Alcohols to Symmetric Ethers:
Bimolecular Dehydration
Competes with alkene formation.
Lower temperatures favor ether formation, a ΔS thing.
After protonation, the alcohol can undergo an S
N
2 attack by another alcohol molecule to form a symmetric ether.
3° Vicinal Diols to Ketones:
The Pinacol Rearrangement
Acid-catalyzed dehydration of a 3° vicinal diol to form a ketone.
Involves a methyl migration, ~CH
3
3° Vicinal Diols to Ketones:
The Pinacol Rearrangement
3° carbocation
resonance-stabilized carbocation
3° Vicinal Diols to Ketones:
The Pinacol Rearrangement
Vicinal Diols to Carbonyls:
Periodic Acid Cleavage of Glycols
Periodic acid is HIO
4
.
Products are aldehydes and ketones.
Products the same as for ozonolysis.
HIO
4
Alcohols to Esters: Acids
When the acid is a carboxylic acid, the reaction is called Fischer esterification.
This is an equilibrium, and it does not always favor the ester.
Alcohols to Esters: Acids
When the acid is sulfuric acid, the product is a sulfate ester.
Alcohols to Esters: Acids
When the acid is nitric, and propane-
1,2,3-triol (glycerine) is the alcohol, what is the product?
When the acid is phosphoric acid, the product is a phosphate ester.
Phosphate esters are the links between nucleotides in RNA and
DNA.
DNA
image from Wikipedia
Oxidation or Reduction?
O O
HO OH
OH
O
C
OH
CH
3
H
2
C
OH O
C
OH
Predict the Product
CH
2
OH
H
2
SO
4
, heat
OH
OH
Na
2
Cr
2
O
7
H
2
SO
4
SOCl
2
Predict the Product
OH
1.
TsCl/pyridine
2.
NaCN
OH
OH
1.
TsCl/pyridine
2.
NaOCH
3
/CH
3
OH
1.
TsCl / pyridine
2.
NaI / acetone
Predict the Product
CH
3
CH
2
OH
H
2
SO
4
140 °C
As opposed to 180°C.
OH
P/I
2
Predict the Product
O Cl
C
OH
+
O
C
OH
+
OH
H
+
Conversions
Br
O
C
H
Br
H
3
C
C
CH
3
OH
CH
3
Br
CH
3
Conversions
OH HO
CH
3
CH
2
OH CO
2
CH
2
CH
3