Enzymes SL - HS Biology IB
advertisement
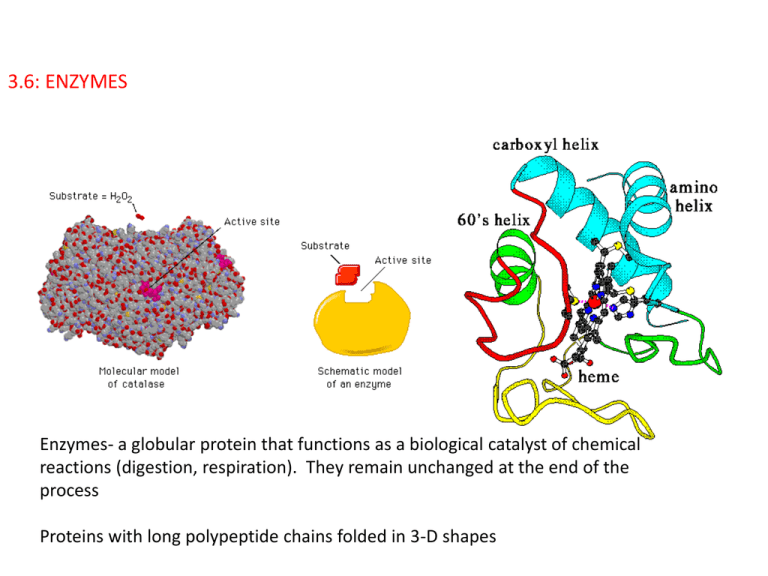
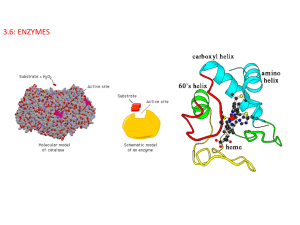
3.6: ENZYMES Enzymes- a globular protein that functions as a biological catalyst of chemical reactions (digestion, respiration). They remain unchanged at the end of the process Proteins with long polypeptide chains folded in 3-D shapes 3.6.4: Define denaturation. Denaturation is a structural change in a protein that results in the loss (usually permanent) of its biological properties. Refer only to heat and pH as agents. 3.6.1: Define enzyme and active site. Active site- specially shaped region of an enzyme where substrates are brought together Substrate- Chemicals involved in a chemical reaction 3.6.2: Explain enzyme–substrate specificity. • Lock and key model http://www.youtube.com/watch?v=CZD5xsOKres http://www.youtube.com/watch?v=V4OPO6JQLOE Shape of enzyme and substrates are complementary, meaning they fit together perfectly, like a lock and key. Lock and Key Hypothesis explains how each enzyme can be so specific. The substrates can be either broken apart, or put together IB Question: (a) Define active site. (b) Explain enzyme-substrate specificity. (a)Define active site. site on surface/portion of the enzyme/protein to which the substrate binds (b) Explain enzyme-substrate specificity. enzymes fit together with substrates similar to a lock and key; active site has shape that gives specificity; enzymes catalyze a reaction with a specific substrate; example of named enzyme and its substrate; substrate held precisely in (optimum) position to make/break bonds/carry out reaction / chemical interaction occurs between enzyme and substrate; [3 max] Accept these points shown in an annotated drawing. Factors Affecting Enzyme Reaction Temperature pH 3.6.3: Explain the effects of temperature, pH and substrate concentration on enzyme activity. pH 3.6.3 pH is the measure of the number of H+ ions and OH- ions Low pH (acidic) has a lot of H+ ions High pH (basic) has a lot of OH- ions pH 7 (neutral) has equal number of H+ and OH- ions pH and Enzyme Activity Enzyme action is influenced by pH because the amino acids that make up an enzyme contain many +/- regions, some around the active site. An excess of H+ ions in an acidic solution can lead to bonding between the H+ ions and the negative charges in the active site. (same with OH- in basic solutions where the OH- ions bond to the positive sites). This will inhibit the matching process between enzyme and the substrate, which will slow or prevent enzyme activity. Extreme ph may denature an enzyme • Most enzymes have an optimum pH such as pepsin in the stomach and trypsin in the small intestines. • Enzymes in the cytoplasm of body cells have optimum pH of 7 • However, not all have an optimum pH. Protease (digest proteins) in the stomach has an optimum pH of 2, but protease in the small intestines has a pH of 8. IB QUESTION: Explain the effects of pH on enzyme catalysed reactions. [3] enzymes have a pH optimum; active site works best at this pH; activity decreases above and below the optimum; by interfering with H-bonding/active site structure; denaturing by extremes of pH so enzyme activity/reaction stops; [3 max] IB Question: The enzyme pepsin, involved in protein digestion in the stomach, requires an acid pH to work properly. Explain the effect of pH on enzyme activity. [2] IB Question: The enzyme pepsin, involved in protein digestion in the stomach, requires an acid pH to work properly. Explain the effect of pH on enzyme activity. [2] each enzyme has an optimum pH where enzyme functions best / drawing of bell curve with optimal pH labelled; changes in pH (from optimum pH) decrease activity/effectiveness; pH slightly changes the shape of the active site / tertiary structure of protein changed / enzyme denatured; more difficult to form enzyme-substrate complex; [2 max] . 3.6.3: Effect of substrate concentration If there is a set concentration of enzymes present in a reaction mixture, and the concentration of the substrate increases the rate of production of the product will increase. This is because there is a greater chance of collisions between the substrate and enzyme. If concentration of substrate increases too much, it will exceed the maximum rate at which enzymes can work. All active sites are occupied by the substrate. IB Question: Explain why enzymes are substrate specific and why their activity is affected by substrate concentration. [8] IB Question: Explain why enzymes are substrate specific and why their activity is affected by substrate concentration. [8] specificity: (active site works as a) lock and (substrate as a) key; (enzyme has) a specific shape; active site; (substrate has) a specific/complementary shape; (active site) fits substrate molecule/part of molecule / enzyme-substrate complex formed; activation energy lowered; substrate concentration: lower/medium concentration activity increases; directly proportional to concentration of substrate; random collisions more frequent; activity levels off / plateau; high concentration no change in activity; as all active sites fully utilised; [8 max] Award [6 max] if only specificity or substrate concentration aspects addressed. Lactose Intolerance and Enzyme Lactase All young mammals are fed milk for the early part of their lives Milk contains the sugar lactose (disaccharide), which is digested in the intestines by an enzyme lactase. 3.6.5 Explain the use of lactase in the production of lactose-free milk. During the normal digestion of milk, the enzyme lactase (made by yeast cells found in milk) breaks lactose into its two monomers: glucose and galactose. Both monomers are small enough to pass through the lining of the small intestine to be absorbed by the bloodstream. Lactose, however, is too large to pass through the intestinal lining. Some individuals are lactose-intolerant; as lactose passes through their digestive tracts it can cause irritation and diarrhea. Lactose intolerance is common in Southeast Asia where people evolved without milk in their diets. In Europe and the Middle East, by contrast, human populations have consumed milk for millennia and have therefore evolved the ability to tolerate lactose. Lactose-intolerant people can drink milk that has had lactase added to remove the lactose. The lactase added to “lactose-free” milk is obtained from a type of yeast called Kluveromyces lactis. Kluveromyces lactis is cultured by biotechnology companies that extract and purify the lactase. Benefits of milk containing glucose and galactose (lactose free) -Makes milk drinkable by lactose- intolerant people -Speeds up fermentation of yoghurt -Sweetens milk products so that less sugar needs to be added -Produce smoother textures (ice cream) IB Question: Describe the use of biotechnology in the production of lactose-free milk. [6] IB Question: Describe the use of biotechnology in the production of lactose-free milk. [6] it allows people who are lactose intolerant/have difficulty digesting lactose to consume milk (products); galactose and glucose taste sweeter than lactose reducing need for additional sweetener (in flavoured milk products); galactose and glucose are more soluble than lactose / gives smoother texture / reduces crystalization in ice cream; (bacteria) ferment glucose and galactose more rapidly (than lactose) shortening production time (of yogurt/cottage cheese); IB Question: Lactase is widely used in food processing. Explain three reasons for converting lactose to glucose and galactose during food processing. [3] IB Question: Lactase is widely used in food processing. Explain three reasons for converting lactose to glucose and galactose during food processing. [3] it allows people who are lactose intolerant/have difficulty digesting lactose to consume milk (products); galactose and glucose taste sweeter than lactose reducing need for additional sweetener (in flavoured milk products); galactose and glucose are more soluble than lactose / gives smoother texture / reduces crystalization in ice cream; (bacteria) ferment glucose and galactose more rapidly (than lactose) shortening production time (of yogurt/cottage cheese); Catalase Catalase enzyme is found in all living things that are exposed to oxygen Catalyzes the decomposition of hydrogen peroxide (H2O2 ) to H20 and O2 2H2O2 2H2O + O2 Hydrogen peroxide is a harmful by product of many metabolic reactions 1 catalase molecule can breakdown millions of hydrogen peroxide molecules in 1 second.