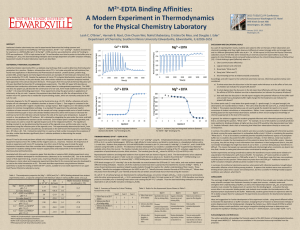
edta_dtpa_inactivation of AP
advertisement

Chelating Agents and Temperature Effects on Alkaline Phosphatase Activity Steven Fan, Stefanie Lopez, Carrie Pusec, Jun Shi Purpose The purpose of the experiment is to use activity assay to determine the effects of chelating agents (EDTA and DTPA) and the effects of temperature on Alkaline phosphatase. Alkaline Phosphatase and Metal ions in Active Site Zn2+ Mg2+ Metal ion roles in AP mechanism Zn2+ ions most significant for conformation stability and catalytic function roles in coordination of nucleophilic attack and facilitating departure of leaving groups Mg2+ unessential (Dieckmann) but contributes to greater stabilization and activity generates hydroxyde for serine deprotonation (Strec. et al) EDTA six coordination bonds with a metal ion general chelator DTPA eight coordination bonds with metal ion zinc specific chelator formation constant (equilibrium constant) 100 fold greater than EDTA relatively insoluble in water, <0.5 g/ 100mL, low pH ~ 2.5 Material and Methods Performed experimental trials to determine the [EDTA] and [DTPA] to use to make comparisons with the temperature For each activity assay, absorbance was measured at 410nm with a spectrometer with 30 second intervals with an extinction coefficient 0.0187 uM-1cm-1 Five activity assays were performed 1. 2. 3. 4. 5. AP with EDTA at RT AP with EDTA at 50°C AP with DTPA at RT with pH = 2.3 AP with DTPA at RT with pH = 8.4 AP with DTPA at 50°C pH = 2.3 Plotted V0 vs. Time to determine half-life based on its exponential decay curve Initial Conditions of AP/Chelator Solution EDTA DTPA Mixture Concentrati on mM Mixture Concentratio n mM EDTA 0.24 DTPA 0.005 Alkaline Phosphatase 0.0073 Alkaline Phosphatase 0.0073 Tris Buffer pH 7.4 10 Tris Buffer pH 10 7.4 Initial Conditions of the Activity Assays for EDTA and DTPA Assay Volume ( L) AP/Chelator Solution 25 PNPP (0.658 mM) 500 Buffer (200 mM Tris pH 8.1) 500 Total 1025 EDTA 9 8 7 6 Vo EDTA HIGH TEMP 5 EDTA ROOM TEMP 4 y = 6.9884e-0.041x 3 2 1 y = 5.8027e-0.272x 0 0 10 20 Time (min) 30 40 50 DTPA 8 DTPA HIGH TEMP 7 DTPA ROOM TEMP PH 8.4 6 Vo (uM/min) 5 DTPA ROOM TEMP - PH 2.3 4 3 2 y = 2.9531e-0.052x 1 0 0 10 20 Time (min) 30 40 50 Half-Life Values for each Condition Half-life (min) EDTA Room Temperature pH 8 11.23 EDTA Elevated Temperature 2.55 pH 8 DTPA Room Temperature pH 2.3 Inconclusive (~hours) DTPA Elevated Temperature 4.02 pH 2.3 General Conclusions Generally, EDTA and DTPA at 25°C and 50°C showed an exponential decay representing the enzyme activity with respect to time. EDTA at the higher temperature increased the enzyme activity and decreased the half life by a factor of 4.4. DTPA is more temperature dependent than EDTA because after heating the system, the time it took for the exponential curve, the half life of DTPA dropped from multiple hours to about one hour Conclusion Between Chelators 1. 2. 3. The DTPA chelator has a bigger effect on the inactivation of Alkaline Phosphatase - Based on the facts…. DTPA binds 100 fold more strongly to the metal ions than EDTA. DTPA is an octadentate and EDTA is a hexadentate Zinc metal is more essential for catalysis over magnesium (Diekmann 2004). The [DTPA] used was 50x less than the [EDTA]. The half life was DTPA at elevated temperature was 4.02 and the EDTA was 2.55, which is fairly close for such a great difference in concentration. Problems The pH of DTPA - pH influenced the solubility - pH adjustment from 2.3 to around neutral pH The concentration of EDTA was not equal to DTPA to make a direct comparison References Boguslaw Stec. 2000. A Revised Mechanism for the Alkaline Phosphatase Reaction Involving Three Metal Ions. J. Mol. Biol. 1303-1311. Coleman, JE. 1992. Structure and Mechanism of Alkaline Phosphatase. Annu. Rev. Biophys. Biomol. Struct. 21:441-83. Garen, A and C Levinthal. 1959. A Fine- Structure Genetic and Chemical Study of the Enzyme Alkaline Phosphatase of E. Coli. Biochem Biophys. Acta. 11;38:470-83. Plocke DJ, Levinathal C, and BV Vallee. 1962. Alkaline Phosphatase of Escherichia coli: Escherichia coli: A Zinc A Zinc Metalloenzyme Metalloenzyme. . Biochem Biochem.. 1;3:373 1;3:373—7 Plocke DJ, and BV Vallee. 1962. Interaction of Alkaline Phosphatseof E. coli with Metal Ions and Chelating Agents. Biochem. 1;6:1039-43 Meow its question time! ???