Lecture-11
advertisement

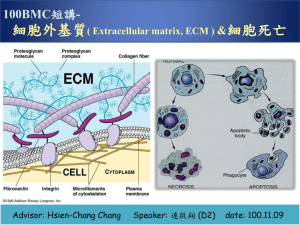
Prof. Ishtiaq Mahmud ishtiaq51@yahoo.com March 11, 2013 Lecture 10 ADVANCES IN CELL BIOLOGY Lecture 10: The Extracellular matrix (ECM) 1. Introduction 2. Collagen, the most abundant protein 3. Post-transcriptional modification 4. Genetic diseases related to collagen synthesis 5. Elastin, Fibrillin, Laminin 6. Proteoglycans and Glycosaminoglycans 7. Functions of proteoglycans 8. Bone, a mineralized connective tissue 9. Cartilage Introduction 1. Tissues are not made up solely of cells. A substantial part of their volume is extracellular space which is largely filled by an intricate network of macromolecules constituting the extracellular matrix (ECM) (fig 10-31, P -972, Edn 3rd) Figure 1 9-53 The connective tissue underlying an epithelium. This tissue contains a variety of cells and extracellular matrix components. The predominant cell type is the fibroblast, which secretes abundant extracellular matrix. 2. The ECM is composed of a variety of versatile proteins and polysaccharides that are secreted locally and assembled into an organized meshwork in close association with the surface of the cell that produce them. 3. Connective tissues from the architectural framework of the vertebrate body. In these tissues the matrix is frequently more plentiful than the cells that it surrounds and it determines the tissues physical properties. (Fig 19-32, P-973, Edn 3rd) Figure 19-54 Fibroblasts in connective tissue. This scanning electron micrograph shows tissue from the cornea of a rat. The extracellular matrix surrounding the fibroblasts is here composed largely of collagen fibrils. The glycoproteins, hyaluronan, and proteoglycans, which normally form a hydrated gel filling the interstices of the fibrous network, have been removed by enzyme and acid treatment. 4. Variations in the relative amounts of the different types of matrix macromolecules and the way they are organized in the ECM give rise to an amazing diversity of forms, each adapted to the functional requirements of particular tissue. The matrix can become calcified to form the rock-hard structures of bone or teeth or it can form the transparent matrix of cornea. At the interface between an epithelium and connective tissue, the matrix forms the basal lamina, a thin but tough mat that plays important part in controlling cell behavior. 5. (a) Until recently the vertebrate ECM was thought to serve as inert scaffolding for stabilizing physical structure of tissues but it is now clear that ECM plays far more active and complex role regulating the behavior of cells that contact it, influencing their development, migration, proliferation, shape and function correspondingly ECM has complex molecular composition (b) Two main classes of extracellular macromolecules that make up the matrix are glycosaminoglycans (GAGs) which are linked to protein to form proteoglycans and fibrous proteins of two functional types: Structural (collagen and elastin) and mainly adhesive (fibronectin and laminin). (c) The principal receptors on animal cells for binding most ECM proteins are the integrins, a large family of homologous transmembrane linker proteins. Extracellular Matrix 6. The macromolecules that constitute the extracellular matrix are mainly produced locally by cells in the matrix. In connective tissue fibroblast secretes the macromolecules. ECM contains three major classes of biomolecules (i) The structual proteins, collagen, elastin and fibrillin (ii) Certain specialized proteins such as fibrillin, Fibronectin and laminin (iii) proteoglycans. 7. ECM has been found to be involved in many normal and pathologic processes i.e. inflammatory states, spread of cancer cells, rheumatoid arthritis and osteoarthritis. Collagen 8. It is the major component of most connective tissues, constitute approximately 25% of protein of mammals and it is the most abundant protein in the animal world. 9. It provides extracellular framework for all animals and exists in every animal tissues. At least 25 distinct types of collagen made up of over 30 distinct polypeptide chains have been identified in human tissues. (Table 47-1, P-546, Harper 27th Edn) 10. A number of proteins that are not classified as collagens have collagen-like domains in their structures there proteins are sometimes referred to as ‘non-collagen collagen’. 11. Types of collagen are subdivided into a number of classes based primarily on the structures they form. (Fig 19-41, P979, Edn 3rd) Figure 19-63 A fibroblast surrounded by collagen fibrils in the connective tissue of embryonic chick skin' In this electron micrograph, the fibrils are organized into bundles that run approximately at right angles to one another. Therefore, some bundles are oriented longitudinally, whereas others are seen in cross section. The collagen fibrils are produced by the fibroblasts, which contain abundant endoplasmic reticulum, where secreted proteins such as collagen are synthesized. Structure of collagen 12. All collagen types have a triple helical structure. Mature collagen type 1, containing approximately 1000 amino acids. Each polypeptide subunit or alpha chain is twisted into a left handed helix of three residues/ turn (Fig 47-1, P-547, Harper 27th Edn). Structure of collagen 13. 3 of these α-chains are then wounded into a right handed super helix forming a rod-like molecule of 1.4 nm in diameter and about 300 nm long. A striking characteristic of collagen is the occurrence of glycine residues at every third position of the triple helical portion of the alpha chain. 14. Glycine is the only amino acid small enough to be accommodated in the limited space available down the central core of the triple helix. This repeating structure represented as (Gly-x-y) n is an absolute requirement for the formation of triple helix. While x and y can be any other amino acid, about 100 of the X positions are proline and 100 of the Y positions are hydroxy proline. These two amino acids provide rigidity on the collagen molecule 15. Collagen fibers are further stabilized by the formation of covalent cross-links. These cross links form through the action of lysyl oxidase, a copper dependant enzyme. 16. Type IV collagen the best characterized example of a colagen is an important component of basement membranes where it forms a mesh like network. . Collagen undergoes modification extensive post-translational 17. Collagen is synthesized on ribosome in a precursor form, Preprocollagen, which contains a leader or signal sequence that directs the polypeptide chain into the lumen of ER, where the leader sequence is removed, Hydroxilation of proline and lysine residues and glycolysation of hydroxylysine in the procollagen molecules also take place at this site (Fig 19-43, P-981, Edn 3rd) Collagen undergoes modification extensive post-translational 18. The procollagen molecule contains extension peptide of 2035 kDa at both its ends. Following secretion from golgi apparatus the extension peptides are removed and enzymes. Then the triple helix spontaneously assembles into collagen fibers. These are further stabilized by the formation of inter and intrachain cross links through the action of lysyl oxidase. 19. Some cells that secrete collagen also secrete fibronectin, a large gylcoprotein present on cell surfaces. Fibronectin binds to aggregating procollagen fibers and alters the kinetics of fiber formation in the matrix. Associated with fibronectin and procollagen in this matrix are the proteoglycans heparan sulfate and chondroitan sulfate. Once formed collagen is relatively metabolically stable. However, breakdown is increased during starvation and inflammation. A number of genetic diseases result from the abnormalities in the synthesis of collagen 20. About 30 genes encode collagen and atleast 8 enzymes catalyzed posttranslational steps. Number of diseases occur due to mutation in the collagen genes. (Table 47-4, P-548) 21. Elastin It is a connective tissue protein that is responsible for properties of extensibiity and elastic recoil in tissues. Although not as widespread as collagen, elastin is present in large amounts, particularly in tissues that require these physical properties e.g. lung, blood vessels and elastic ligaments and small amounts in ear and skin. (Fig 19-49, P985, edn 3rd) Figure 1 9-70 Elastic fibers. These scanning electron micrographs show (A) a lowpower view of a segment of a dog's aorta and (B) a high-power view of the dense network of longitudinally oriented elastic fibers in the outer layer of thesame blood vessel. All the other components have been digested away with enzymes and formic acid. 22. It is synthesized as soluble monomer of 70kDa called tropoelastin, deletion of elastin gene have been found in 90% of the subjects with Williams syndrome, a developmental disorder affecting connective tissue and the central nervous system (Table 47-5, P-549, Harper 27th edn). A number of skin diseases are associated with accumulation of elastin. Alternatively, a decrease of elastin is found in – conditions such as pulmonary emphysema and aging of the skin Fibrillin 23. A large glycoprotein (about 350 KDa) that is a structural component of microfibrils 10-12 nm fibers found in many tissues. It is secreted into ECM by fibroblast and becomes incorporated into the insoluble microfibrils which appear to provide a scaffold for deposition of elastin. 24. Marfan Syndrome, It is a relatively prevalent inherited disease affecting connective tissues. It is inherited as an autosomal dominant trait. It affects the eyes, the skeletal system and hyper-extensibility of the joints and cardiovascular system. (Abraham lincolin may have had this condition). This syndrome is due to mutations in the gene for fibrilin. Lamininn 25. It is a protein of about 850 kDa and 70 nm long, consists of 3 polypeptide chain linked together to form an elongated cruciform shape. It has binding sites for type IV collagen, heparin and integrins. It helps in the anchoring of the lamina to the cells. Fibronectin 26. It is a major glycoprotein of ECM, also found in soluble form in plasma. It contains an Arg-Gly-Asp (RGD) sequence that binds to the receptor. The protein is involved in the adhesion of cells to ECM and cell migration. The amount of fibronectin around many transformed cells are sharply reduced partly explaining the faulty interaction with the ECM Proteoglycan and Glycosaminoglycans (GAG) 27. Proteoglycans are proteins that contain covalently linked glycosaminoglycans. At least 30 have been characterized and given names such as syndecan, serglycin. They vary in tissue distribution. The protein bound covalently to glycosaminoglycan is called core proteins. The amount of carbohydrate in a proteoglycan is usually much greater than is found in a glycoprotein and many comprise up to 95% of this weight. 28. There are at least seven glycosaminoglycans (GAGs), Hyaluronic acid, chondoitin sulfate, Keratin sulfate and chondroitan sulfate I and II, heparin, heparan sulfate and dermatan sulfate. (Fig 19-33, P-973, Edn 3rd) Figure I 9-55 The repeating disaccharide sequence of a heparan sulfate glycosaminoglycan (GAG) chain. These chains can consist of as many as 200 disaccharide units, but are typically less than half that size. There is a high density of negative charges along the chain due to the presence of both carboxyl and sulfate groups. The proteoglycans of the basal lamina-perlecan, dystroglycan, and collagen XVlll-all carry heparan sulfate GAGs. The molecule is shown here with its maximaf number of sulfate groups. ln vivo, the proportion of sulfated and nonsulfated groups is variable. Heparin typically has >70o/o sulfation, while 29. A GAG is an unbranched polysaccharide made up of repeating disaccharides. One component of which is always an amino sugar (hence the name GAGs), either D-glucosamine or D-galactosamine. The other component of the repeating disaccharide is an uronic acid. With the exception of hyaluronic acid, all the GAGs contain sulfate groups. GAGs are highly negatively charged. They occupy large amount of space and form hydrated gels Proteoglycans have numerous functions 30. Proteoglycans have numerous functions and found in every tissue of the body mainly in ECM or ‘ground substance’ where they are associated with each other and also with the other major structural components of the matrix, collagen and elastin. These interactions are important in determining the structural organization of the matrix. The GAGs bind polycations and cations such as Na+ and K+, Polycations can also act as sieves restricting the passage of large growth factors such as TGF-β, modulating their effects on cells. 31. Some functions of specific GAGs and proteoglycans (i) Hyaluronic acid, is in high conc. in embryonic tissues and is thought to play an important role in permitting cell migration during morphogenesis and wound repair. 32. Chondroitin sulfate, it is located at sites of calcification in endochondral bone and is also found in cartilage, located also in certain neurons providing endoskeletal structure to maintain their shape. (Table 47-8, P557, Harper) 33. Keratin sulfate I and dermatan sulfate are present in the cornea, plays important role in corneal transparency. 34. Heparin is an important anticoagulant, binds with factors IX and XI, but it’s most important interaction is with plasma antithrombin. It can also bind to lipoprotein lipase present in capillary walls, causing a release of this enzyme into the circulation. Proteoglycans are also found in intracellular locations such as nucleus Bone is a mineralized connective tissue 35. It contains both organic and inorganic, organic material is mainly protein, Bone is a dynamic structure that undergoes continuous cycle of remodeling, consisting of resorption followed by deposition of new bone tissue. The major cell types are osteoclasts and osteoblasts. Osteoclast is multinucleated cells derived from pluripotent hematopoietic stem cells. Osteoblasts control mineralization. 36. Metabolic and genetic disorder involves bone. Osteogenesis imperfecta (brittle bones) is characterized by abnormal fragility of bone. 37. Osteopetrosis (marble bone disease) characterized by increased bone density is due to inability to resolve bone. 38. Osteoporosis is a generalized progressice reduction in bone tissue mass per unit volume causing skeletal weakness (Table 4710. P-561) Cartilage 39.Collagen is the principal protein. Elastic cartilage contains elastin and fibroelastic cartilage contains type 1 collagen. Cartilage contains a number of proteoglycans which play important role in its compressibilty.