10_Proton_NMR
advertisement

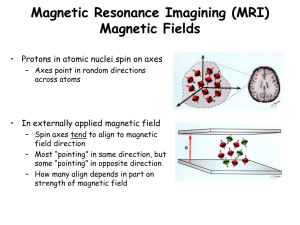
Proton Nuclear Magnetic Resonance (1H-NMR) Spectroscopy - Part 1 Lecture Supplement page 133 Organic Structure Analysis Crews, Rodriguez, Jaspers 1998 p.5-6 1H-NMR Spectroscopy Background and Theory Fundamental principle The energy required to cause nuclear spin flip is a function of the magnetic environment of the nucleus. •Protons, electrons, neutrons have “spin” (I) •Motion of charged particle creates magnetic field •In absence of external influence, magnetic poles (spin axis) randomly oriented •Add external magnetic field (Bo) spins align Bo add magnetic field No external magnetic field Spin alignment random With external magnetic field Spins aligned Background and Theory Nuclear Spin Flip •I = +1/2 parallel to Bo (lower energy); I = -1/2 antiparallel to Bo (higher energy) Increasing energy •Addition of energy results in nuclear spin flip I = -1/2 I = +1/2 Absorb energy (excitation) Release energy (relaxation) Ground state Nuclear spin parallel to Bo Lower energy DE ~ 0.02 cal mol-1 = radio wave photons Excited state Nuclear spin antiparallel to Bo Higher energy Contrast this with absorption of infrared light (p. 114 of lecture supplement) Background and Theory Magnetic Field Controls DE • DE influenced by magnetic field strength at nucleus Spin state energy I = -1/2 DE DE Small magnetic field small DE Large magnetic field large DE I = +1/2 Magnetic field strength at nucleus Energy required for spin flip (DE) Information about magnetic field strength at nucleus Information about chemical structure Background and Theory The NMR Spectrum •Spectrum = plot of photon energy versus photon quantity Intensity of signal (photon quantity) NMR signal Spin flip energy (photon energy) Background and Theory The NMR Spectrum Nuclear: Manipulation of nuclear spin Magnetic: Magnetic field strength influences DE Resonance: Tendency of a system to oscillate at maximum amplitude at a certain frequency NMR C 1H O X C O nucleus = a proton 1H-NMR = proton NMR Background and Theory Spectrum Structure How do we deduce structure from NMR spectrum? Information from NMR spectrum: •Number of signals number of nonequivalent proton groups in molecule •Position of signals (chemical shift) magnetic environment of protons •Relative intensity of signals (integration) ratio of equivalent proton types •Splitting of signals (spin-spin coupling) proton neighbors Number of Signals Proton Equivalency •NMR signal due to photon absorption •Photon energy controlled by magnetic environment of nucleus •Nuclei in same magnetic environment = equivalent •Multiple magnetic environments multiple signals •Number of signals = number of equivalent proton sets H O H H H Cl Protons equivalent One NMR signal CN Protons not equivalent Two NMR signals Number of Signals Proton Equivalency How to test for equivalency? •Equivalent = proton magnetic environments identical in every way •Nonequivalent = proton magnetic environments not identical in one or more ways •Easier to test for nonequivalency than for equivalency Useful vocabulary CH3 = methyl H H H Cl C C C H H Cl CH2 = methylene H H H H Cl C C C H H Cl H CH = methine H H H Cl C C C H H Cl H Number of Signals Proton Equivalency Proton equivalency examples Hb, Ha not equivalent Ha, Hc not equivalent H H C Cl H One signal H H H C C H H rapid equilibrium Cl Two signals ? Hb, Hc equivalent Ha, Hc equivalent •NMR = camera with slow shutter speed •NMR detects only average when rotation is fast •Thousands of 360o bond rotations per second •Therefore Ha, Hb, Hc appear equivalent Single bond rotation in acyclic molecules often allows equivalency Number of Signals Proton Equivalency Proton equivalency examples Hb, Ha not equivalent Ha, Hc not equivalent H H C Cl H One signal H H H C C H H rapid equilibrium Cl Two signals ? Hb, Hc equivalent vs. Ha, Hc equivalent Number of Signals Proton Equivalency More proton equivalency examples H H H C C H H OH H Three signals H C F Cl H H C C C Four signals C H C mirror plane C C H H H C H H F C C F Cl One signal H C C H C H H C Cl Two signals H C F H C H One signal H Cl Number of Signals Proton Equivalency Sample spectra •Verify what we have learned about equivalent protons •Which spectrum belongs to this molecule? H3 C H3 C H C C H3 C H OH Three proton sets three signals O H3C H3C C H C H Number of Signals Proton Equivalency Which spectrum belongs to this molecule? H H HO OH H Two proton sets two signals H HO OH H3 C CH3 Proton Nuclear Magnetic Resonance (1H-NMR) Spectroscopy - Part 2 Lecture Supplement page 139 OCH3 H3C C CH3 OCH3 1H-NMR Spectroscopy Part 1 Summary •Atomic nucleus has spin, and therefore generates a magnetic field •Nuclear spin axis can be parallel or antiparallel to external magnetic field (Bo) •Spin parallel to Bo (I = +1/2) lower energy than spin antiparallel to Bo (I = -1/2) •Energy difference between spin states (DE) controlled by magnetic field at nucleus •Absorption of radio wave photon with energy = DE causes nuclear spin flip •NMR spectrum = plot of photon energy (spin flip energy) versus photon quantity Information from NMR spectrum •Number of signals reveals number of sets of equivalent protons Equivalency: Protons must be identical in all ways to be equivalent Nonequivalency: Protons can be different in just one way Example: 1H-NMR spectrum of CH3CH2OH has three signals •Position of signal (chemical shift) •Relative intensity of signals (integration) •Splitting of signals (spin-spin coupling) Vollhardt, 10-2 Position of Signals The Chemical Shift How does spin flip energy relate to molecular structure? •Spin flip energy depends on magnetic field strength: Spin state energy I = -1/2 DE DE Small magnetic field small DE Large magnetic field large DE I = +1/2 Magnetic field strength at nucleus •Magnetic field strength varies between NMR spectrometers •High magnetic field = higher spectral resolution (more spectral detail) •Need a spin flip energy scale that is independent of magnetic field strength •Chemical shift: Spin flip energy scale normalized to be independent of field strength Position of Signals The Chemical Shift How does molecular structure influence chemical shift? •Chemical shift controlled by DE which is controlled by magnetic field at nucleus What contributes to magnetic field at nucleus? Earth’s magnetic field Weak; 0.3-0.6 gauss Spectrometer’s magnetic field Strong; typically 94 kilogauss Other electrons and nuclei in the molecule •Electron cloud shields atomic nucleus from external magnetic fields Shielded: Nucleus feels weaker magnetic field Deshielded: Nucleus feels stronger magnetic field Position of Signals Intensity of signal (photon quantity) The Chemical Shift Reference point? 0.00 ppm (CH3)4Si Tetramethylsilane (TMS) 15 ppm Spin flip Chemical energy shift (photon scale energy) 0 ppm (ppm) Deshielded Shielded Low Magnetic Field Strength High Magnetic Field Strength Position of Signals The Chemical Shift •How does molecular structure influence chemical shift? 5.0 4.0 2.0 1.0 2.0 3.0 4.0 O 3.5 F 4.0 Cl 3.0 Br 2.8 I 2.5 C 2.5 0.0 ppm (CH3)4Si 0.00 ppm CH3CH3 0.86 ppm CH3Br 2.68 ppm CH3Cl 3.05 ppm CH3OH 3.42 ppm 1.0 EN of X in CH3-X 3.0 Chemical shifts for H3C-X: Si H 1.8 2.1 Conclusion: EN of atoms near H chemical shift •Electron cloud shields atomic nucleus from external magnetic fields Shielded: Nucleus feels weaker magnetic field Deshielded: Nucleus feels stronger magnetic field deshielded Vollhardt, Fig 10-9 shielded Position of Signals The Chemical Shift How does electronegativity influence chemical shift? •Chemical shift related to magnetic field strength at nucleus •Electron cloud shields nucleus from effects of Bo H H I Iodine has low EN Br H Cl H F Fluorine has high EN Electron density at H is high Electron density at H is low H more shielded H is less shielded H has lower chemical shift H has higher chemical shift Metaphor: Ozone layer shields Earth Position of Signals Do not memorize chemical shifts. Table given on exams. Position of Signals Notes On Characteristic Chemical Shifts Table •Characteristic shifts are typical proton averages. Actual shifts may lie outside given range. O O CH3 Typically 2.0-2.6 ppm Useful chemical shift trends •RCH3 < RCH2R < R3CH 0.23 ppm CH3OH 3.39 ppm CH3 2.01 ppm CH3O CH3 2.59 ppm EN of C (in R) > EN of H •EN effects decrease with distance: CH4 O 3.59 ppm CH3CH2OH 1.18 ppm 1.53 ppm CH3CH2CH2OH 0.93 ppm 3.49 ppm Position of Signals •Example: Avoid this common misconception: “NMR peaks can be assigned based on chemical shift alone” 3.8 ppm not always ROCH3 2.3 ppm not always ArCH3 Common exception 6.5-8.0 ppm usually benzene ring protons C=O stretch Relative Intensity of Peaks Integration Information from NMR spectrum •Number of signals number of nonequivalent proton groups in molecule •Position of signals (chemical shift) magnetic environment of protons •Relative intensity of signals (integration) ratio of equivalent proton types •Splitting of signals (spin-spin coupling) proton neighbors Relative Intensity of Peaks Integration •Beer’s Law: Amount of energy absorbed or released proportional to moles of stuff present •NMR: Amount of radio wave energy proportional to peak area •Measurement of peak areas = integration Peak height Peak area ∫ir I∫aac Newton Gottfried Leibniz Inventor∫ of calculu∫ •Relative intensities of NMR signals proportional to relative number of equivalent protons •Integrals do not always correspond to exact number of protons Example: Integrals of 2:1 might be 2H:1H or 4H:2H or... Sample Spectra •Verify what we have learned about equivalent protons, chemical shifts, and integration •Assign peaks to corresponding hydrogens: H H C H OH 4.19 ppm: integral = 1.0 (1 H) 3.41 ppm: integral = 3.0 (3 H) CH3OH has 4 H Sample Spectra Assign peaks to corresponding hydrogens: OCH3 H3C C CH3 OCH3 3.19 ppm: integral = 1.0 (6 H) 1.33 ppm: integral = 1.0 (6 H) C5H12O2 has 12 H Two equal integrals Two groups of equivalent H Smallest integral often set = 1 Integration gives proton ratio Sample Spectra Assign peaks to corresponding hydrogens: CH3OCH2CH2OCH3 3.55 ppm: integral = 1.0 (4 H) 3.39 ppm: integral = 1.5 (6 H) Two groups of equivalent H Two unequal integrals C4H10O2 has 10 H 10 H / (1.0 + 1.5) = 4 H per unit OCH3 H3C OCH3 CH3 1 1 2 CH3OCH2CH2OCH3 O H3C C H C H H3C HO 2 H3 C 3 C OH 2 2 H3 C H C C H3 C H H3 C CH3 CH3CH2Br OH 2 2 Lecture Supplement p. 138, 139, 146 Sample Spectra •Assign peaks to corresponding hydrogens: CH3CH2Br Three peaks! Four peaks! •Why the extra peaks? Hint: Think about spin and magnetic fields Proton Nuclear Magnetic Resonance (1H-NMR) Spectroscopy - Part 3 Lecture Supplement page 147 CH3CH2Br 1H-NMR Spectroscopy Part 2 Summary Information from NMR Spectrum 1. Number of signals how many sets of equivalent protons 2. Position of signals (chemical shift) magnetic environment of nucleus Deshielding by electronegative atoms higher chemical shift 3. Relative intensity of signals (integration) how many hydrogens per signal Integrals give proton ratio; not always equal to absolute proton count (i.e., 1.5:1) 4. Splitting of signals (spin-spin coupling) Example: 3.55 ppm: integral = 1.0 4 H CH3OCH2CH2OCH3 3.39 ppm: integral = 1.5 6 H Two groups of equivalent H Two unequal integrals C4H10O2 has 10 H 10 H / (1.0 + 1.5) = 4 H per unit Signal Splitting 1H-NMR spectrum of CH3CH2Br has more details... 3.43 ppm: integral = 1.0 2 H 1.68 ppm: integral = 1.5 3 H Two unequal integrals 5 H / (1.0 + 1.5) = 2 H per unit CH3CH2Br Three lines A triplet Four lines A quartet Signals are split Signal Splitting What is the origin of signal splitting? Each line in signal... ...has slightly different chemical shift ...represents slightly different spin flip energy ...represents nucleus with slightly different magnetic environment A nucleus with only one magnetic environment causes a singlet A nucleus with two magnetic environments causes a doublet Signal Splitting How can one nucleus have different magnetic environments? •Caused by spin direction of adjacent nuclei Ha Bo C C Hb Ha feels Bo + Hb Larger DE Ha feels Bo - Hb Smaller DE NMR signal for Ha •Ha feels two different magnetic environments •Ha has two different spin flip DE •Ha has two different (but very similar) chemical shifts •Ha signal is split into a doublet Signal Splitting Some Useful Terms Spin-spin coupling: One nuclear spin influences spin of another nucleus Splitting: Effect on NMR signal caused by spin-spin coupling J Coupling constant (J): Spacing between lines in a splitting pattern Signal Splitting More Than One Neighbor What is splitting when there is more than one neighbor? Ha C C Hc Hb Ha feels Bo + Hb + Hc Ha feels Bo - Hb + Hc = Bo Bo equal energy } H feels B + H - H = B a o b c o Ha feels Bo - Hb - Hc •Ha has three different (but very similar) chemical shifts •Ha signal is split into a triplet NMR signal for Ha 1:2:1 because of energy state population probabilities Signal Splitting Rules and Restrictions General rule: The signal for a proton with n neighbors is split into n+1 lines Rules and Restrictions for Proton-Proton Spin-Spin Coupling 1. Only nonequivalent protons couple. X X Ha Hb Hc Hd H C C C C H H H H •Hb couples with Hc H •Hb and Ha do not couple because they are equivalent •Hc and Hd do not couple because they are equivalent Signal Splitting Rules and Restrictions 2. Protons separated by more than three single bonds usually do not couple. Hb Hc •H and H _____ 2 bonds apart Can couple * Cannot couple a b C Ha X *Assuming Ha and Hb are not equivalent C C •Ha and Hc _____ 3 bonds apart Can couple Cannot couple •Ha and Hd _____ 4 bonds apart Can couple Cannot couple Hd Pi bonds do not count toward this bond limit, but J may be too small to observe. Free spacer Hb Hc C 2 (+1) bonds apart Can couple Cannot couple •Ha and Hc _____ C Ha C Hd •Ha and Hb _____ 2 bonds apart Can couple Cannot couple 3 (+1) bonds apart Can couple* Cannot couple •Ha and Hd _____ *But Jad may be very small Signal Splitting Rules and Restrictions 2. Protons separated by more than three single bonds usually do not couple. Hd •Benzene ring = one big free spacer Hc F Hb Cl Ha •All benzene ring protons may couple with each other but J may be small •Ha, Hb, Hc, and Hd all couple with each other •Jad may be too small to observe Benzene ring is a “gated community”; it blocks some coupling that we expect to observe. H X CH3 X CH2CH3 Signal Splitting Rules and Restrictions 3. Signals for O-H and N-H are usually singlets singlet H2N triplet H H C C H H OH singlet triplet •Splitting of O-H or N-H protons may be observed in rare circumstances Sample Spectra •Verify what we have learned about equivalency, chemical shifts, integration, and splitting •Assign peaks to corresponding hydrogens in structure BrCH2CH2CH3 3.39 ppm (triplet; integral = 1.0) 2 H 1.87 ppm (sextet; integral = 1.0) 2 H 1.03 ppm (triplet; integral = 1.5) 3 H 7 H / (1.0 + 1.0 + 1.5) = 2 H per unit Sample Spectra •Assign peaks to corresponding hydrogens in structure Br H3C C CH3 H 3.78 ppm (septet; integral = 1.0) 1 H 1.31 ppm (doublet; integral = 6.0) 6 H 7 H / (1.0 + 6.0) = 1 H per unit OCH3 H3C C OCH3 CH3 CH3OCH2CH2OCH3 O H3C C H H3C H3 C H C H3 C H C C H3 C H HO H3 C OH CH3 CH3CH2Br OH Lecture Supplement p. 138, 139, 146 Sample Spectra For next lecture: •Assign peaks to corresponding hydrogens in structure •Explain splitting patterns H 5.66 ppm (multiplet; integral = 1.0) H C H H2C C H2C 1.61 ppm (multiplet; integral = 2.0) C C H H H 1.98 ppm (multiplet; integral = 2.0) Proton Nuclear Magnetic Resonance (1H-NMR) Spectroscopy - Part 4 Lecture Supplement page 154 CH3 1H-NMR Spectroscopy Part 3 Summary Information from NMR Spectrum •Number of signals how many sets of equivalent protons •Position of signals (chemical shift) magnetic environment of nucleus Deshielding by electronegative atoms higher chemical shift •Relative intensity of signals (integration) how many hydrogens per signal Integrals give proton ratio; not always equal to absolute proton count (i.e., 1.5:1) •Splitting of signals (spin-spin coupling) The signal of a proton with n neighbors is split into n+1 lines (first order coupling) Example: CH3CH2Br CH3 is a triplet, CH2 is a quartet More complex patterns (non first-order coupling) are common 1H-NMR Spectroscopy Part 3 Summary Splitting rules •Only nonequivalent hydrogens couple with each other OH •Hydrogens can be at most three single bonds distant •Pi bonds and benzene rings are “free spacers” H C H H C H H C H H H H H •Benzene ring “gated community” •OH, NH usually do not couple, and usually are not split H Sample Spectra •Assign peaks to corresponding hydrogens in structure Multiplet: A splitting pattern that is too complex to decipher H 5.66 ppm (multiplet; integral = 1.0) 2 H H C H H2C C H2C 1.98 ppm (multiplet; integral = 2.0) 4 H 1.61 ppm (multiplet; integral = 2.0) 4 H C C H H H 10 H / (1.0 + 2.0 + 2.0) = 2 H per unit Non-First Order Splitting Why Is Cyclohexene Splitting Not Simple? First order splitting: All J values in a splitting pattern are equal •n+1 rule obeyed; “normal” doublets, triplets, etc. result Non-first order splitting: J values in a splitting pattern are unequal •More complex splitting patterns result Example: Hb Ha C C Hc Jab = Jac “normal” triplet Jab ≠ Jac doublet of doublets For Chem 14C we predict J values equal and “normal” coupling results. Exceptions are plentiful. Sample Spectra Effects of Pi Electron Clouds: Magnetic Induction •Vinyl protons deshielded by magnetic induction H H H H C H H2C C H2C C C H C Bo H Deshielded H •May also cause shielding Sample Spectra Benzene Ring Protons H 10.00 ppm (singlet) H 7.87-7.56 ppm (multiplet) H C H O H 9.5-11 ppm H 6.5-8.0 ppm •Magnetic induction causes benzene ring proton chemical shifts ~6.5-8.0 ppm •Magnetic induction by C=O causes aldehyde proton chemical shift ~9.5-11 ppm •Due to long range coupling, benzene ring proton signals often multiplets Sample Spectra Benzene Ring Protons Benzene ring proton signals can be deceptively simple... CH3 •7.2 ppm = singlet? •Benzene ring protons not equivalent When chemical shifts very similar J 0; splitting disappears