Hist & Struct of Atom
advertisement

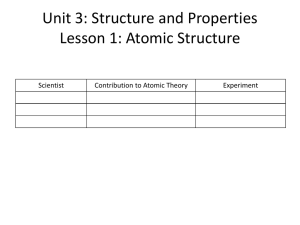
How Humans Learned about… In the late 1800s, scientists such as J.J. Thomson were using cathode ray tubes to study more about the nature of matter. 1897: J.J. Thomson experiments with a cathode ray tube. Thomson’s experimental results prompted him to propose a model of matter: Thomson proposed a model, sometimes called the "plum pudding" or "raisin cake" model, in which thousands of tiny, negatively charged spheres swarm inside a cloud of mass-less positive charge. Yes, on a microscopic level, all matter is like a raisin cake. Ernest Rutherford and His Experiment • In 1911 Ernest Rutherford thought it would prove interesting to bombard atoms with alpha rays, figuring that this experiment could yield information about the inside of the atom. He used radium as the source of the alpha particles and shined them onto the atoms in gold foil. Behind the foil sat a fluorescent screen for which he could observe the alpha particles impact. • Rutherford thought • he would see this: Instead, he saw this: Rutherford Discovers the Nucleus • Alpha particles that deflected right back must have hit something with a strong positive charge. Rutherford then theorized that there was something called a nucleus, which contained a high density of positively charged particles. In fact Rutherford speculated that the atom was like a miniature solar system, where the electrons orbited like little planets. The Mystery of Light Emission and How Niels Bohr Solved It Scientists of the 19th century discovered that when an electrical current passes through a small quantity of a gas in a glass tube, the atoms in the gas emit light. BUT this radiation occurs only at certain specific, or discrete, wavelengths, and different elements and compounds emit different wavelengths, depending on the element. Argon Xenon The lines are at specific wavelengths that are characteristic of that element. Niels Bohr, a young scientist working in Rutherford’s laboratory, set out to understand the emission of radiation at these wavelengths based on the nuclear model of the atom. The Bohr Atom Using Rutherford’s model of the atom as a miniature solar system, Bohr developed a theory by which he could predict the same wavelengths scientists had measured radiating from atoms with a single electron. He concluded that because atoms emit light only at discrete wavelengths, electrons could only orbit at certain designated radii, and light could be emitted only when an electron jumped from one of these designated orbits to another. The Bohr atom was an improvement over the Rutherford atom, and it most certainly was an improvement over JJ Thomson’s raisin cake model. However, some important and VERY odd phenomena still needed explaining, and Bohr’s model couldn’t do it. It seems that every object (including YOU) has a particle nature AND a wave nature. For very small objects such as the electron and the photon (photons are light packets) the wave nature is as significant as the particle nature. So how should the electron be described, as a wave OR a particle??? A new and RADICAL model of the atom was soon to come along That would take into account both the wave and particle nature of matter….. the The work of a number of scientists contributed to the development of the quantum mechanical model: Albert Einstein Louis de Broglie Wolfgang Pauli Max Planck Werner Heisenberg Enrico Fermi THE QUANTUM MECHANICAL MODEL OF THE ATOM The quantum mechanical view of atomic structure does keep some of Rutherford and Bohr’s ideas. The nucleus is still at the center of the atom and provides the electrical attraction that binds the electrons to the atom. And specific energy levels are associated with each electron shell. Contrary to Bohr’s theory, however, the electrons do not circulate in definite planet-like orbits. Rats.. Rats The quantum-mechanical approach to the atom provides the framework for viewing the electrons as… fuzzy clouds of negative charge. Electrons still have assigned states of motion, but these states of motion DO NOT correspond to fixed orbits. Instead, they tell us something about the geometry of the electron cloud—its size and shape and whether it is spherical or bunched in lobes like a figure eight. Physicists called these states of motion atomic orbitals. But how did we come to learn the shapes of the electron clouds, or orbitals? Enter… Erwin Schroedinger and His Famous Equation Schroedinger’s equation, when solved, gives us the geometry of the electron orbitals (clouds). Four types of orbitals, or electron cloud structures, exist in the atom: s orbitals p orbitals d orbitals f orbitals One final strange feature of the quantum mechanical model of the atom is that you can never know exactly where the particle version of the electron is inside its orbital. This strange feature is called the Heisenberg Uncertainty Principle. Cartoon about the Heisenberg Uncertainty Principle: Get it?