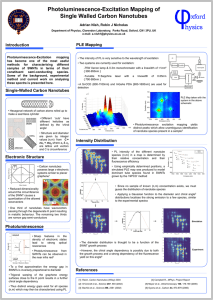
Wai-Leung Yim, XG Gong, and Zhi-Feng LiuJ. Phys. Chem. B 2003

Nanotube as a gas Sensor
Outline
• What are Carbon nanotubes?
– Types
– Properties
– Applications
• Motivation
• Approach
• Results
– Preliminary results
• Symmetry and
• Basis set Effect
– NO
2
+CNT
• Future Work
• Conclusion
• Acknowledgements
• References
What are carbon nanotube
•Carbon nanotubes, long, thin cylinders of carbon, were discovered in 1991 by S. Iijima
• They can be considered as rolled up graphene tubes of carbon
• There are two types: SWNT and MWNT.
•It has very strong C-C chemical bonding.
Types of SWNT
A chiral vector C h characterizes the nanotubes C h
=na
1
+na
2
, where a
1 and a
2 are lattice vectors of the 2D hexagonal lattice, and n and m are integers
Properties of nanotubes
• They can be either metals or semiconductors with different size energy gaps, depending diameter and helicity of the tubes, on the indices (n,m)
• A SWNT is considered metallic if the value n - m is divisible by three. Otherwise, the nanotube is semi conducting
.
• Ultra-small SWNTs (diameter 4Å) exhibit
Superconductivity below 20K.
• Nanotubes are very strong with very high Youngs Modulus and extremely flexible
• High thermal conductivity.
• High sensitivity to gas adsorption
Applications
• Micro-electronics / semiconductors
• Controlled Drug Delivery/release
• Field Effect transistors and Single electron transistors
• Nano electronics
• Nanogear
• Hydrogen Storag e
Motivation
• Use of carbon nanotubes as chemical sensors for gases like NH
3 and NO
2 was first demonstrated by Kong et al.
• Electrical conductance of an SWNTs increases by three orders of magnitude when exposed to NO
2 and to decrease by 2 orders of magnitude in the presence of Ammonia.
• In general all the papers have predicted physisorption between NO
2
, followed by charge transfer from tube to molecule
• There is very little or no interaction between NH
3
SWNT . The interaction between NH
3 and and SWNT was studied by photoemission spectroscopy and it was found out that the tube is sensitive to NH
3 sensitivity is lesser as cmpd to NO2 even though the
Approach
• The motivation for out study was to study the nanotube gas interaction by approximating the nanotube as a molecule of specific length and using semi empirical method (PM3) to predict the change in properties.
• A 10, 0 semiconducting nanotube was used for our study.
• To study the change in the electronic structure of the tube
– when NO2 physisorbs
– NO2 chemisorbs
– PES with varying C-N bond length
– Rotational PES for NO
2 physisorbed well in chemisorbed well and
– Effect of adsorption of 2 NO
2 molecules
D10d symmetry
Effect of symmetry
D10h symmetry
•The symmetry of the nanotube depends on the number of hexagons along the tube axis and along the circumference.
•They to, 2 different types of point groups, Dnh (with horizontal mirror planes) and Dnd (with dihedral mirror planes)
Effect of symmetry on the LUMO and
HOMO
Single point calculations using DFT(B3PW91 ) and HF
Effect of Symmetry
LUMO and HOMO of D10h tube
Band gap and dipole moment
Basis set effect
•The DFT and HF calculations done using the STO-
3G basis set concentrate the frontier orbitals of the carbon nanotube on the edge carbon atoms While the
Hf / 3-21G split valence basis set distributes the orbitals along the axis of the nanotubes resulting in delocalized HOMO- LUMO orbitals.
Length effect
HOMO of (10,0 )N=5 and N= 7 energy band gap of N= 5 tube = 0.255
LUMO 10,0 N=5 ,and 7
HOMO and LUMO ,CNT +NO
2
HOMO and LUMO orbital for NO
2 at a dist of 1.8 and 2.61A
PES wrt C-N
PES of Rotation
Pes for rotation in physisorbwell
1.6384
1.6382
1.638
1.6378
1.6376
1.6374
1.6372
0 50 100 150
Angle between No2 and CNT z axis(deg)
200
Series1
PES of rotation in Chemisorbed well
1.646
1.645
1.644
1.643
1.642
1.641
1.64
0 50 100
Angle (deg)
150 200
Series1
C-N = 1.8A
C-N = 2.61A
NO
2
in
Type plain CNT
Energy
2.2320
Physisorbed 1.6374
Chemisorbed 1.6481
No2in 1.6105
band gap
0.045
0.255
0.261
0.272
Model Chemistry
Type
CNT
CNT+NO
2
(PM3)
CNT+NO
2
(ROHF/3-21g
CNT +NO
2 in (PM3)
CNT +NO
2 in (ROHF/3-21g)
2NO
2
(PM3)
Band gap
0.045
0.255
0.016
0.272
0.011
0.06
Binding energy
BE = (E molecule+CNT
– E
CNT
–E
NO2
)
Type
Physisorbed
Chemisorbed
NO
2 in
2NO
2 binding energy
-0.5916
-0.5787
-0.618
0.0326
Future Work
• To evaluate the binding energies of the structures using ROHF/3-21G level of theory.
• To determine the amt and type of charge transfer in the system
• To study the method for regeneration process
– Either by the route of Chemical reaction or
– By investigating the Energy difference for 2 NO2 molecules on the surface
• To study the behavior of the tube in the presence of the electric field
• To compare the more favourable position for NO2 inside or outside the nanotube
Conclusion
• Symmetry of the nanotube fragment affects the nature of the frontier orbitals
• As the length of the nanotube increases , the orbitals are less delocalized
• PM3 introduces a spin contamination which can be potentially solved by doing a single point at higher level of theory
• From the current calculation NO2 prefers to be inside the Nanotube
• Between chemisorbed and Physisorbed region, physisorption seems to be the preferred state.
Acknowledgment
• Dr Schlegel
• Dr Goldfield
• Dr Hratchian
• Dr Anand
• Dr Knox
• Jie Lie
• Stan Smith
• Barbara Munk
References
• http://www.pa.msu.edu/cmp/csc/ntproperties/
• http://dagotto.phys.utk.edu/condensed/noppi.carbon.2.pdf
• http://physicsweb.org/articles/world/11/1/9
• http://www.e-nanoscience.com/application.html
• Teri Wang Odom; Jin-Lin Huang; Philip Kim; Charles M. Lieber,
J.
Phys. Chem. B , 2000, 104, 2794-2809.
• L.G. Bulusheva; A.V. Okotrub; D.A. Romanov; D. Tomanek, J. Phys.
Chem.A, 1998, 102, 975-981.
• M.J.Frisch et al., GAUSSIAN 03, Revision B.05, Gaussian Inc.,
Pittsburgh PA, 2003.
• Shu Peng a,, Kyeongjae Cho a, Pengfei Qi , Hongjie Dai, Chemical
Physics Letters 387 (2004) 271–276
• Wai-Leung Yim, X. G. Gong, and Zhi-Feng LiuJ. Phys. Chem. B
2003, 107, 9363-9369