物理(十五)
advertisement

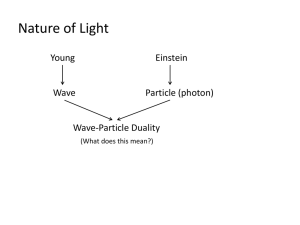
12 量子物理 Sections 1. 2. 3. 4. 5. 6. 7. 8. 9. Photon and Matter Waves Compton Effect Light as a Probability Wave Electrons and Matter Waves Schrodinger’s Equation Waves on Strings and Matter Waves Trapping an Electron Three Electron Traps The Hydrogen Atom 12-1 Photon and Matter Waves (光子和物質波) • Light Waves and Photons c f E hf ( photon energy ) h 6 . 63 10 34 J s The Photoelectric Effect 光電效應 The experiment • First Experiment (adjusting V)– the stopping potential Vstop K max eV stop 光電子的最大動能與 光強度無關 •Second Experiment (adjusting f)– the cutoff frequency f0 低於截止頻率時即使光再 強也不會有光電效應 The plot of Vstop against f h V stop ( ) f e e The Photoelectric Equation hf K max h V stop ( ) f e e h 6 . 6 10 34 J s Work function 12-2 Compton Effect p hf c h (photon momentum) 康 普 吞 效 應 實 驗 圖 表 康普吞效應圖示 Energy and momentum conservation hf h f K K mc ( 1) 2 2 hf h f mc ( 1) h h mc ( 1) pX h / p e mv Frequency shift h 0 h h cos mv cos sin mv sin h (1 cos ) mc Compton wavelength 12-3 Light as a Probability Wave The standard version The Single-Photon Version First by Taylor in 1909 The single-photon, double-slit experiment is a phenomenon which is impossible, absolutely impossible to explain in any classical way, and which has in it the heart of quantum mechanics - Richard Feynman The Single-Photon, WideAngle Version (1992) 50μm The postulate Light is generated in the source as photons Light is absorbed in the detector as photons Light travels between source and detector as a probability wave 12-4 Electrons and Matter Waves •The de Broglie wave length •Experimental verification in 1927 •Iodine molecule beam in 1994 h p 1989 double-slit experiment 7,100,3000, 20,000 and 70,000 electrons Experimental Verifications X-ray Electron beam 苯 環 的 中 子 繞 射 12-5 Schrodinger’s Equation • Matter waves and the wave function ( x, y , z , t ) ( x, y , z )e i t •The probability (per unit time) is 2 ie. * Complex conjugate 共軛複數 The Schrodinger Equation from A Simple Wave Function ( x, y , z , t ) ( x, y , z )e i t A sin( kx ) B cos( kx ) (1D) p h / k E p / 2m k / 2m 2 2 2 1D Time-independent SE A sin( kx ) B cos( kx ) 1 d 2 d / dx 2 E 2 2 k k 1 2 2 d 2 2 m dx d 2 2 2 2 m dx 2 E dx 2 3D Time-dependent SE 2 d 2 2 m dx 2 2 ( 2m E 2 x 2 2 i 2 2m 2 y 2 t V i 2m z 2 ) E 2 2 2 t 12-6 Waves on Strings and Matter Waves 駐波與量子化 Quantization 駐波: = 2L n f v n v n = 0 ,1 ,2 , 2L Confinement of a Wave leads to Quantization – discrete states and discrete energies 12-7 Trapping an Electron For a string: L n n 1, 2 , 3 , 2 y n A sin( n : quantum n ) x, L number n 1, 2 ,3 , Finding the Quantized Energies of an infinitely deep potential energy well h/ p h/ 2 mE , L n / 2 E n n h / 8 mL , 2 2 2 n 1, 2 ,3 , The Energy Levels 能階 The ground state and excited states The Zero-Point Energy n can’t be 0 The Wave Function and Probability Density For a string y n A sin( n A sin( 2 n n n 1, 2 , 3 , ) x, n 1, 2 ,3 , L n L A sin ( 2 ) x, 2 n L ) x , n 1, 2 , 3 , The Probability Density Correspondence principle (對應原理) At large enough quantum numbers, the predictions of quantum mechanics merge smoothly with those of classical physics •Normalization (歸一化) ( x )dx 1 A 2 n 2/L A Finite Well 有限位能井 d 2 dx 2 8 m 2 h 2 [ E E pot ( x )] 0 The probability densities and energy levels Barrier Tunneling 穿隧效應 •Transmission coefficient 8 m (U b E ) 2 T e 2 bL k h 2 STM 掃描式穿隧顯微鏡 Piezoelectricity of quartz 12-8 Three Electron Traps • Nanocrystallites 硒化鎘奈米晶粒 那種顏色的顆粒比較小 2 En t n h 2 8mL c ft 2 ch Et A Quantum Dot An Artificial Atom The number of electrons can be controlled Quantum Corral 量子圍欄 12-1.9 The Hydrogen Atom •The Energies U 1 q1 q 2 4 0 r En me 4 8 h 2 0 1 2 n 2 1 e 2 4 0 r 13 . 6 ev n 2 , n 1, 2 , 3 , The Bohr Model of the Hydrogen Atom Balmer’s empirical (based only on observation) formula on absorption/emission of visible light for H 1 1 R 2 2 , for n 3, 4, 5, and 6 n 2 1 Bohr’s assumptions to explain Balmer formula 1) Electron orbits nucleus 2) The magnitude of the electron’s angular momentum L is quantized Fig. 39-16 L n , for n 1, 2, 3, 39- 41 Orbital Radius is Quantized in the Bohr Model Coulomb force attracting electron toward nucleus F e 2 4 0 r 2 1 Quantize angular momentum l : F k q1 q 2 r v2 ma m r 2 rm v sin rm v n v n rm Substitute v into force equation: h 0 2 r me r an , for n 1, 2, 3, 2 n , for n 1, 2, 3, 2 2 Where the smallest possible orbital radius (n=1) is called the Bohr radius a: h 0 2 a me 2 5.291772 10 10 m 52.92 pm 39- 42 Orbital Energy is Quantized The total mechanical energy of the electron in H is: 2 1 e 2 1 E K U 2 mv 2 4 r 0 Solving the F=ma equation for mv2 and substituting into the energy equation above: E 1 e 2 8 0 r Substituting the quantized form for r: En 2.180 10 n 2 18 J = En 13.60 eV n 2 me 4 1 8 h n 2 0 2 2 for n 1, 2, 3, , for n 1, 2, 3, 39- 43 Energy Changes hf E E high E low Substituting f=c/ and using the energies En allowed for H: 1 1 2 2 3 2 8 0 h c n high n low 1 1 1 R 2 2 n n high low 1 me 4 Where the Rydberg constant R me 4 8 0 h c 2 3 1.097373 10 7 m -1 This is precisely the formula Balmer used to model experimental emission and absorption measurements in hydrogen! However, the premise that the electron orbits the nucleus is incorrect! Must treat electron as matter wave. 39- 44 氫 原 子 能 階 與 光 譜 線 Schrödinger’s Equation and the Hydrogen Atom Fig. 39-17 U r e 2 4 0 r The Ground State Wave Function 1 (r ) a h 0 3/2 e r /a 2 a me 2 5 . 29 pm (Bohr radius) Quantum Numbers for the Hydrogen Atom For ground state, since n=1→ l=0 and ml =0 The Ground State Dot Plot Wave Function of the Hydrogen Atom’s Ground State Probability of finding electron within a within a small distance from a given radius Probability of finding electron within a small volume at a given position P r 2 r Fig. 39-21 Fig. 39-20 39- 50 氫原子的量子數 N=2, l=0, ml=0 N=2, l=1 Hydrogen Atom States with n>>1 As the principal quantum number increases, electronic states appear more like classical orbits. Fig. 39-25 P r fo r n 4 5, n 1 44 39-54