Introduction to PALM and STORM
advertisement

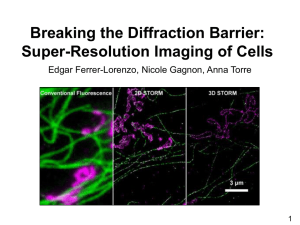
STORM and PALM How to get started. June 2014 Christa Walther 200 nm nm 1 μm approx. resolution 30 nm approx. resolution 250 nm STORM/ PALM workflow Planning time spent Preparation Imaging Reconstruction Analysis Publication Planning increase in time required to get results • single protein localisation – one colour experiment - difficult to relate to cell structure overall • two colour imaging - selection of compatible spectrally distinct labels/ appropriate activator combination • 2D or 3D imaging – 3D involves no scanning in z (no z-stack), additional calibration data needs to be acquired, z resolution will still be lower than x/y resolution • live cell imaging – possible but slower than conventional fluorescence microscopes Planning • examples from the literature: o organisms used o intracellular or extracellular staining o multicolour application o live or fixed cell work o for fluorescent proteins: label specific restrictions: multi-mer/ dimer/ temperature dependence => something that has been used only once before might be difficult to repeat, especially in a different system STORM imaging • using dyes/ labels such as Alexa 647, Cy5, Atto 680, Mitotracker Red – match label to available lasers/ filters • not all available dyes can be used for this, e.g. FITC/ Rhodamine/ DAPI do not work • can be targeted by antibodies or SNAP/CLICK tag labelling – intracellular labelling is not always possible • choice of targeting will influence achievable resolution PALM imaging • not all known fluorescent proteins will work – need to show switching behaviour: o photoactivation/ photoswitching: on/off o photoconversion: wavelength switching o e.g. GFP does not work • uses fluorescent proteins such as mEos, PAmCherry – match label to available lasers/ filters, e.g. eYFP needs a 514 nm laser • targeting by genetic modification – expression needs to be tested Preparation • chosen labelling needs to work in your system using diffraction limited imaging (deconvolution/ confocal) • acquire images of good quality with your protocol – spend some time optimising your signal • dual colour imaging should show similar fluorescence intensities => remember: higher resolution can only give meaningful information when the labelling is sufficient Imaging - optimisation • controls to show that localisation imaged is not negatively influenced by labels chosen (e.g. unlabelled, sec. antibody only) • imaging in super resolution requires the use of specific mounting buffers: STORM, oxygen scavenger based, reducing agent – multitude of recipes PALM, usually PBS (plus mounting reagent), not as complex as STORM Imaging - optimisation • different labels/ label combinations require mounting buffer optimisation: time consuming trial and error – can only be done on the N-STORM system • choice of label - sequence of imaging o PALM: image higher wave-length emission first to minimise crosstalk but activation laser will always act on both o combination of PALM and STORM is possible Imaging – sample mounting • sample needs to be properly sealed for buffer to work • inclusion of fiducial markers in sample preparation to allow for o correction of drift (either during or after the experiment) o autofocusing during experiment o channel alignment in multi-colour experiments • fiducial markers: fluorescent beads or gold nanoparticles o need to be immobilised on slide to be of use Image reconstruction N-STORM • Nikon image format • analysis computer/ software will be available • Nikon software can be used for image reconstruction o multitude of open software options offering different levels of analysis Image analysis • localisation / resolution of structure analysed • co-localisation – strongly depends on label density • consistency of results with other imaging techniques • pattern recognition/ statistics • confirmation of results by variation of approaches: o switch of labels o drug treatment that should eliminate structure Ordered by acquisition time Ordered by Z-position In summary: PALM/ STORM Analysis Publication ! Publication Any publications using images from the SIM or N-STORM system needs to include: The super resolution facility at the LMF is funded by MRC grant No MR/K015753/1.