Symptom Benefit ANZGOG-0701
advertisement

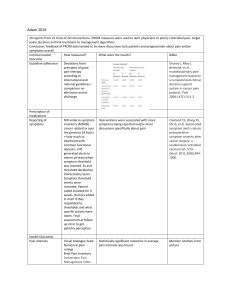
,START UP MEETING FOR STAGE 2 Response Rates in Phase 3 Trials Chemotherapy Liposomal doxorubicin Gemcitabine Gemcitabine + Pertuzumab Topotecan Response rates % 10-12 5-9 13 6 Trabectedin 7 Patupilone 15 Patients with good PS entered into clinical trials- 2nd Line Therapy Symptom Benefit Study Expectation is that Symptom Benefit > Response Rate ( otherwise why would we treat so many patients ) • How to best measure the impact of chemotherapy on symptom improvement ? • Can we use Symptom Benefit Measures as an alternative outcome measure • Can we identify patients most likely to benefit from palliative chemotherapy? Stage 1-Primary Aim • The symptoms and aspects of HRQL that are rated as most severe, troublesome and important by patients. • The optimal items and questionnaire/s for measuring these improvements. Study Schema Target Population • >18yrs •platinum resistant/ refractory epithelial ovarian cancer /> 3 lines of therapy •ECOG 0-3 •Able to commence treatment within 2wks of registration •Sufficient English to complete QoL forms independently R E G I S T E R Stage1 100 patients • Complete 7 QoL forms • 20 subjects will participate in additional QoL telephone interview Data Collection 4 Treatment cycles or Disease progression Stage 1 QoL Questionnaires • • • • • • • • Symptom Representation Questionnaire FACT-O (includes FOSI) EORTC QLQ-C30 EORTC QLQ-OV28 Patient Data Form Expected and Perceived Benefit Scale HAD Scale (Baseline & End of Treatment) Herth Hope Index (Baseline & End of Treatment only) Results • Majority Platinum Resistant • Compliance 96% All questionnaires were completed to a very high compliance rate with few or no missing data Reasons for Starting Chemotherapy REASONS FOR CHEMOTHERAPY Patients (N=89) Symptom control only 1 Rising CA125 only 1 Radiological evidence only 1 Symptom control + rising CA125 only Symptom control + radiological evidence only 15 4 Rising CA125 + radiological evidence only 19 Symptom control + rising CA125 + radiological evidence 48 Patients “3 most noticed symptoms” and clinician rated AE’s at baseline CAUSE BOTH TREATMENT & DISEASE PREDOMINANTLY DISEASE RELATED PREDOMINANTLY EMOTIONAL % Patients reported (N=83) % AE reported by clinician (N=92) FATIGUE 40% 55% PAIN (ABDOMINAL/ UNSPECIFIED) 40% 32% ABDOMINAL PROB/BLOATING 30% 17% NAUSEA/VOMITING 20% 33% BOWEL PROBLEMS 20% 27% SHORTNESS OF BREATH 12% 2% APPETITE LOSS 11% (Anorexia 22%) SLEEP DISTURBANCE 27% 4% DEPRESSION/MOOD PROBLEMS 12% 5% SYMPTOM Number of Lines of Prior Therapy Previous Lines 1 2 3 4 5 6 7 UK Patients 89 22 26 20 7 6 2 1 5 Improvement in quality of life (Prior to Cycle 3 N=72) Is your symptom improvement enough to affect your overall quality of life? Stage 1-outcome Results have led to development of MOST (Measure of Ovarian cancer Symptoms and Treatment concerns) Modification of Patient Data Form COVERS ALL SYMPTOMS AND ASPECTS OF QOL IDENTIFIED IN STAGE 1 MOST Measure of Ovarian cancer Symptoms and Treatment concerns Comprises of 35 individual items on a discrete scale of 0-10, where major symptomatic distress is represented by 10. The first 15 items refer to disease symptoms Items 16 and 17 refer to physical and emotional well-being Item 18 is a question referring to overall well-being Items 19-35 deals with side effects and other concerns The study will examine the extent of clinical improvement by examining changes in these items from baseline at each time point- to determine MCID Schema – Stage 2 Target Population •Informed consent R •≥18yrs E •Platinum Resistant/Refractory •ECOG 0-3 •Life expectancy > 3 months •Able to commence treatment within 2wks of registration •Able to complete questionnaires independently G I S T E R Data Collection • Baseline • Each treatment cycle • One month post completion of treatment or until disease progression Stage 2 Primary Objective To determine: The criteria for defining a clinically significant subjective improvement and the optimal instrument/s to measure benefit Secondary Objectives • The proportion of women benefiting from palliative chemotherapy • The time to symptom deterioration • The proportion of women who receive treatment because they are (a) symptomatic, (b) have rising tumor markers alone, or (c) have imaging evidence of disease progression • The percentage of patients who complete 4 or more cycles of treatment • The most common, most severe and most noticed symptoms as perceived by patients. • Develop a prognostic index Stage 2 • MOST • FACT-O • EORTC QLQ C30 • EORTC OV28 • Expected and Perceived Benefits These forms will be completed at Baseline and after each cycle until chemotherapy ceases. Prognostic Factors Recruitment The recruitment target is 600 evaluable patients (~780 enrolled patients) The estimated recruitment period is until December 2011 Currently there are 20 sites activated and 101 patients recruited with a further 40+ sites to open International participation Canada (additional sites) To be confirmed United Kingdom Japan Ireland Spain Germany Scandinavia Italy France USA- selected sites Central Study Contacts Study Chair: Professor Michael Friedlander ANZGOG Coordinating Centre: Study team: Symptom Benefit NHMRC Clinical Trials Centre Locked Bag 77 CAMPERDOWN NSW 1450 AUSTRALIA symptombenefit@ctc.usyd.edu.au Central Coordinator: Kim Gillies +61 2 9562 5032 kim.gillies@ctc.usyd.edu.au Data Manager: Lisa Martyn +61 2 9562 5394 lisa.martyn@ctc.usyd.edu.au Program Manager: Julie Martyn +61 2 9562 5092 julie.martyn@ctc.usyd.edu.au