Electron probe microanalysis EPMA
advertisement

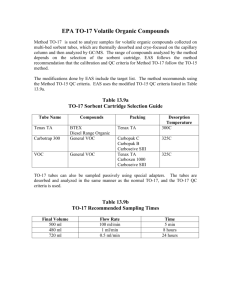
Electron probe microanalysis EPMA Trace Element Analysis Mod 11/8/10 What’s the point? What’s the minimum detection limit for a particular element – or said otherwise, at what point can be be sure that a small inflection above the surrounding background really is a peak? What kind of confidence level should be place on such a number? Definitions “Generally, WDS can achieve limits of detection of 100 ppm in favorable cases, with 10 ppm in ideal situations where there are no peak interferences and negligible matrix absorption.” (Goldstein et al., p. 341) “No” Zn ... but at what level of confidence? Major >10 wt% Minor 1-10 wt% Trace <1 wt% wt% ppm 100 1000000 10 100000 1 10000 0.1 1000 0.01 100 0.001 10 0.0001 1 Trace elements .... and trace elements In the real world, the definition of “trace element analysis” is sometimes broader than the strict quantitative analysis of ppm level elements in one microvolume (~micron interaction volume). Many individuals desire to use EPMA to tell them about the “distribution of trace elements” in their materials, e.g. where the 30 ppm of Pb is in a cast iron. There are two possibilities: the 30 ppm is spread uniformly throughout the material, or in fact most of the material has probably <1 ppm of Pb, but a small fraction of the volume has phases that have Pb at major element levels.The question is then, are they at least the size of the interaction volume and if so, where are they. Our discussion here will deal with all these aspects. A little background-1 Interest in trace elements dove tails with the develop of techniques that could achieve better/quicker/cheaper/more precise/small volumes of said elements. • From the 1960s on, geochemists and petrologists developed increasing interest in trace element partitioning between fluids/melts and minerals. The electron microprobe became the instrument of choice for characterizing the trace levels in doped experiments . • There has been an interest in “trace elements” in certain minerals to assist in the search for ore bodies that contain said elements. A related research field is locating the naturally-occurring minerals that are responsible for certain levels of groundwater contamination (e.g. As). A little background-2 Also… • In both material science and geology, diffusion processes are studied, and EPMA is a prime technique. As you go further and further from the boundary between the two phases, the elements drop to trace element levels. But where do they drop below detection? Or how do you set up the EPMA conditions to reach down to a desired very low level? How low can we go? The USNM olivine standard above (San Carlos, Mg.9 Fe.1SiO4) has a published Ca content of “<0.04 wt% (= 400 ppm). This scan was acquired at 20 keV, 30 nA, with 10 seconds per channel. Clearly there is a peak at the Ca Ka position (24 cps), somewhat above the background (~10 cps). At what point can we say with 99% confidence that there is a statistically significant peak? MDL Equations - 1 The key concept here is minimum detection limit (MDL), i.e., what is the lowest concentration of the element present that is statistically above the background continuum level by 3 sigma (commonly accepted level). There are (at least) two equations used to define the MDL: • the first* uses the Student t test values: CMDL a CS 21/ 2 (t 1 n1 )SC 1/ 2 N S NSB n where the detection limit CMDL is in wt%, CS=std wt %, bar NS=ave. peak cts on std, bar NSB=bkg cts on std, SC= std dev of measured values and n=number of data points • the second†, which is probably more wider used, was developed by Ziebold (1967): (3.29) a CMDL (nTP PB )1/ 2 where n=number of measurements, T=seconds per measurement, P=pure element count rate, P/B= for pure element, and a=matrix correction (a factor or ZAF). * Goldstein et al., p. 500, equation 9.84 † Goldstein et al., p. 500, equation 9.85 MDL Equations - 2 There are several points to be made about these two equations: • the first (Student t test) equation only works for the average of several measurements, since it uses SC, the average of several measurement. This calculation is useful in that special case. • however, as many times as not, a specific area or region is only measured once (e.g., a linear traverse across a zoned crystal), and the second equation is the appropriate one to use. • note in the second equation, the term P times P/B appears in the denominator. As P2/B increases, the MDL decreases (the lower, the better!). This P2/B term is called the ‘figure of merit” for trace element work. • following some discussions with John Valley about the traditional (second) equation and how the peak and background used in it were from the pure element standard—not the unknown, I went back to first principles and derived the equation. Deriving the MDL equation-1 1. We need to determine the precision of the background value for the unknown, i.e. for a given background value, how big is the statistical error bar (counting statistic, 3 sigmas) above it. So here, is it large as on the right below, or is it small as on the LEFT? Deriving the MDL equation-2 2. Let us consider Ca Ka peak on our olivine. We measure the background and get 9 cts/sec. The 1 sigma value however is calculated from TOTAL counts, NOT count rate. So we must multiply 9 cts/sec by the time, 10 seconds, and we get 90 counts. 1s=Sq.Rt. of 90 =9.5 counts, so 3s =28.5. We’ll use 3 s for now, the 99.7% confidence level. Ergo, our MDL for Ca in the olivine is 29 counts above background, over 10 seconds (or if plotted on the wavescan where data are in cts/sec, it would be a value of 3 cps (the left purple marker). Note: we haven’t said one word about count rate on a standard, and we have figured out the minimum detection limit for Ca in our unknown -- though we don’t know what that mdl of 3 cts/sec translates to in ppm or wt%). Deriving the MDL equation-3 3. However, we usually want to “translate” those raw counts into a more usable number, i.e. so many ppm. For that, we need some reference intensity counts for Ca Ka. We then count Ca ka peak and backgrounds for the same time (10 sec) on CaSiO3 (38.6 wt% Ca) and find a count rate of 6415 cts/sec on the peak and 16 cts/sec on the background. 4. So what is 2.9 cts/sec equal to in elemental wt%? We create a pseudo kratio where we take the statistical uncertainty of the background counts (square root, i.e. 1 sigma) divided by the Peak-Bkg of the standard counts on the element peak of interest KCamdl unk bkgctsunk ZAFCa Ca st d 3 C st d st d Ca (pkcts bkgcts)Ca ZAFCa and multiply by 3 (for 3 sigma, 99% confidence) and the ZAF of each and then by the composition C of the standard.The mdl will be in whatever units C is in. Deriving the MDL equation-4 ZAFCaol 1.1087 ZAFCastd 1.080 MDL ol Ca 90 1.1087 3 38.6 .017 wt%Ca 66250 1.080 This is virtually the same result as the “single line” detection limit provided by Probe for Windows (0.015 wt%, shown on next slide), derived from the Ziebold equation. It would appear that the Ziebold equation is not exactly correct, for we must really be concerned with the background precision of the unknown, and the background level of the standard could be several times higher or lower. Going back and re-reading Ziebold, we find two interesting statements: that the equation “gives a measure of the detectability limit” and “there is more than one way to define a detectability limit”. Both are correct, and yes, the equation gives an approximation of the detection limit -- but not the limit per se. MDL in olivine - single line Good totals Excellent stoichometry These are the single line detection limits, calculated with Ziebold’s equation (Goldstein, eq 9.85, p. 500) MDL in olivine - average For a homogeneous sample, it is “legal” to add together all the counts, which gives greater precision and a lower detection limit, e.g. 110 ppm here for Ca at 99% ci. This is a handy chart that shows what kind of counting time would be required, under the same analytical conditions, to achieve a lower detection limit. For example, to get a mdl of 25 ppm, you’d need to count for ~5 minutes on the Ca peak and then background (for each of 10 spots). The current analysis here was a little over 1 minute per spot (thus, about 12 minutes for a mdl of 110 ppm. For 25 ppm, it would add an additional 100 minutes to the analysis time. “Figure of Merit” Variation of figure-of-merit (P2/F) with accelerating voltage for various elements in different matrices. Scaled so 15 kV=1, no ZAF corrections. Probers in Australia have much interest in pushing the lower limits of EPMA detection, for mineral exploration research.Utilizing extreme operating conditions (50 kV, 475 nA, 10 minute counts) they have achieved mdl’s below 5 ppm for some elements. Variation of figure-of-merit with accelerating voltage, each element relative to its level at 15 kV. Detection limits (3 sigma) calculated for 100 sec counts, 50 kV, 450 nA. Data are all k-ratios, not ZAF corrected. They utilize a “figure of merit” of P2/B as a measure of how to achieve lower mdl (the higher the P2/B ). From our first principles derivation, we can see that the P comes from the standard, the B from the unknown. From Advances in Electron Microprobe Trace-Element Analysis by B.W. Robinson and J. Graham, 1992, ACEM-12 Keys to low detection levels • Maximize counts by utilizing • Highest currents feasible (concern: beam damage) • Highest E0 as feasible (concern: increased penetration/range) • Longer count times • Correctly determine background locations • Correct for unavoidable on-peak interferences within the matrix correction* *Donovan, Snyder and Rivers, 1993, An improved interference correction for trace element analysis, Microbeam Analysis, 2, 23-28. Backgrounds: traces can overlap traces Correct locating of background positions is particularly important in trace element work, as both first order and higher order peaks can cause incorrect assessment of background level. Here, scans of the 3 Caltech/MAS trace element glass standards are overlain. (Xe L edge present as a Xe gas sealed counter used.) From Carpenter, Counce, Kluk, and Nabelek, Characterization of Corning Standard Glasses 95IRV, 95IRW and 95IRX: NIST/MAS Workshop, April 2002. Backgrounds: Pb Ma in Monazite Here the Th Mz1 and 2nd order La La1 peaks fall close to potential backgrounds for Pb Ma. Monazite (Ce,La,REE,Th)PO4 has been used for age dating, using U, Th and Pb concentrations. Backgrounds ... holes Au La Probers in Australia, interested in detecting trace levels of gold in certain minerals, discovered a “hole in the background” about 200 sin theta units below the Au La position. (This scan was on SrTiO3, on the LIF crystal). Trace elements as fingerprints: apatite in bentonites Crystals were separated from clay; mixed population (zircons, white; apatites, yellow in false color mosaic BSE image) mounts in 4 mm plug (above). (Research of Norlene Emerson.) A range of trace elements were analyzed in bentonites (very old volcanic ash), in order to verify common stratigraphic horizons in Ordovician sediments. 40-60 ppm mdls were achieved with 20 second counts and 60 nA currents. Where is the ...Arsenic? Some groundwaters in northeast Wisconsin have elevated Arsenic (8 mg/L), and EPMA is being used to help understand the source. Aquifer strata contain mineralized zones (500-80 ppm whole rock), mainly marcasite (FeS2) and quartz. X-ray maps (PfW-MAN) were acquired overnight for Fe, S, Si, O and As. They showed that As is located on the edge of some quartz grains. Here, a rectangular area was mapped at 10 mm intervals. (Research of Toni Simo,Katie Thornberg, Selena Mederos) Pb in Cast Iron C Ka Fe Ka (Research of Jun Park, Carl Loper and John Fournelle.) Pb Ma This cast iron has 100 ppm of Pb in the bulk analysis, and the question was which phase did it reside in. The working hypothesis was that it was associated with graphite dendrites. A full quantitative X-ray map (backgrounds acquired) was acquired overnight (conditions 15 keV, 300 nA, 150 seconds each on Pb peak and bkg). The mdl for Pb is 200 ppm (.02 wt%). X-ray mapping of irregularly positioned/shaped zircon grains • Mounted in epoxy: need to avoid melting epoxy with high currents! • Define polygon boundary • Select point spacing interval • Fully automated quantitative EPMA • Software contouring or 3D surface mapping (“Surfer”) CL Huckleberry Ridge Tuff Zircon grain A BSE BSE (2 Ma, 2500 km3, normal d18O) U wt% Th wt% Th and U Ma (PET) • 18 keV, 400 nA • 94 points • 10 elements • 11 mm spacing • 50 sec on peak + 50 sec on bkgs = 8 hours total time • mdl = 130 ppm (.013 wt%) (Research of Ilya Bindeman, John Valley and John Fournelle) Standards: validating trace element procedure • There is an issue of trace element accuracy on unknowns, where the standard for the element of interest was at a high level. Such a standard should be used for peaking the spectrometer and acquiring standard counts, but it is recommended that a secondary trace level standard be also analyzed to validate the procedure. • Such secondary standards could be • Synthetic glasses such as the Caltech/MAS 95IRV,W and X glasses; NIST glasses and metals; Ni-Cr diopside glass, etc. • Minerals and glasses analyzed by ion probe Comparison: Trace elements by WDS vs EDS WDS is clearly the better method for acquiring trace element data, by an order of magnitude or so compared to EDS. Goldstein et al, 1992, p. 501